Plasma‑Enhanced ALD of Co₃O₄ on TiO₂ Boosts UV Photocatalytic Degradation of Methylene Blue
Abstract
Co₃O₄‑coated commercial TiO₂ (P25) powders were fabricated by plasma‑enhanced atomic layer deposition (PEALD). X‑ray diffraction, transmission electron microscopy, and X‑ray photoelectron spectroscopy confirmed that the anatase TiO₂ lattice and crystallite size remain unchanged, while trace Co₃O₄ nanoparticles are anchored to the surface. Under UV irradiation, the 100‑cycle and 200‑cycle Co₃O₄‑coated samples achieved nearly 100 % degradation of methylene blue (MB) in 1.5 h, compared with 80 % for bare P25. Mott‑Schottky analysis demonstrates the formation of a p‑n heterojunction, facilitating efficient electron–hole separation. The Co₃O₄ coating also enhances MB adsorption due to a higher isoelectric point. These findings highlight PEALD as a precise method to construct effective p‑n junction photocatalysts.
Background
Industrial effluents increasingly contaminate water resources with persistent organic dyes, which not only pose health risks but also diminish light penetration in aquatic systems [1, 2]. Conventional treatment methods often fail to fully oxidise such recalcitrant compounds [3]. TiO₂‑based photocatalysts, prized for their low toxicity, chemical robustness, and broad‑spectrum activity, remain a leading solution for dye removal [4‑7]. Nevertheless, TiO₂ suffers from a rapid recombination of photogenerated charge carriers and a wide bandgap that restricts absorption to the UV region (≈ 4 % of solar irradiance) [8‑10]. To overcome these limitations, strategies such as metal/non‑metal doping, dye sensitisation, and heterojunction engineering have been pursued [11‑15]. In particular, p‑n junctions between n‑type TiO₂ and p‑type semiconductors (e.g., NiO, Ag₂O) create an internal electric field that promotes charge separation and extends light absorption [16‑20]. Co₃O₄, a p‑type transition‑metal oxide with a narrower bandgap (≈ 2.1 eV) and superior chemical stability, has shown promise in dye degradation, gas sensing, and energy storage [23‑28]. Co₃O₄/TiO₂ heterojunctions have already demonstrated enhanced photocatalytic performance compared with their individual components [29, 30]. While various synthesis routes (CVD, plasma spray, PECVD, impregnation–deposition–decomposition) exist for Co₃O₄/TiO₂ composites, they often involve multi‑step processes and high‑temperature calcination, potentially generating hazardous emissions. Atomic layer deposition (ALD) offers a single‑step, low‑temperature, conformal coating approach with precise thickness control [38‑42]. Plasma‑enhanced ALD (PEALD) further expands the technique’s versatility by allowing substrate temperatures well below 200 °C [41]. In this work, we employ PEALD to deposit trace Co₃O₄ layers onto P25 TiO₂, forming a p‑n junction that markedly enhances UV photocatalytic activity.
Methods
Commercial P25 TiO₂ powders were loaded into a porous carrier and transferred to a SUNALE R‑200 PEALD chamber (Picosun). The Co precursor, dicarbonyl cyclopentadienyl cobalt (CoCp(CO)₂, 96 % purity), was maintained at 45 °C, while a 2500 W O₂ plasma (160 sccm O₂, 99.999 % purity) served as the oxidant. PEALD cycles (200 °C) consisted of 0.2 s CoCp(CO)₂ exposure, 6 s N₂ purge, 21.5 s O₂ plasma, and 6 s N₂ purge. 100‑ and 200‑cycle Co₃O₄ coatings were produced by PEALD; a 600‑cycle sample was fabricated by thermal ALD using O₂ gas (130 sccm) in place of plasma. Structural analysis employed XRD (Rigaku‑D/max 2000, Cu Kα, 10°–80°) and BET surface area measurement (Micromeritics Tristar‑3000). Morphology and particle size were examined by FE‑SEM (Ultra 55) and TEM (FEI Tecnai G² F20). Chemical composition was assessed via XPS (Thermo ESCALAB, Al Kα) and ICP‑MS (Thermo X Series 2). Photocatalytic tests involved 50 mg of catalyst in 50 mL of 37.4 mg/L MB solution, illuminated by a 100 W UV LED (15 cm distance). Degradation was monitored by UV‑vis absorption at 664 nm. Visible‑light activity was evaluated using a 300 W Xe solar simulator (420‑nm cut‑off) on methyl orange (MO) solutions. Mott‑Schottky plots were recorded in 1 M NaOH using a CHI760E electrochemical station, with photocatalyst slurries electroplated onto FTO substrates. Zeta potential measurements (Malvern Zetasizer Nano ZS 90) determined the isoelectric points (IEPs) of MB, P25, and Co₃O₄‑coated P25.
Results and Discussion
XRD and Structural Integrity
XRD patterns of bare P25 and 200‑cycle Co₃O₄‑coated P25 exhibit identical anatase peaks (JCPDS 21‑1272), confirming that the TiO₂ lattice and crystallite size (~20 ± 2 nm) remain unaltered after PEALD.
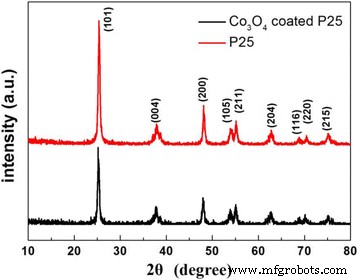
XRD patterns of pure P25 and 200‑cycle Co₃O₄‑coated P25 powders.
Morphology and Particle Size
SEM and HRTEM images show that both samples retain a 15–30 nm nanoparticle morphology. TEM reveals ~2–3 nm amorphous Co₃O₄ nanoparticles densely anchored to TiO₂ surfaces in the 200‑cycle sample, indicative of an island growth mode. The average particle sizes from image analysis are 25.8 nm (P25) and 26.2 nm (Co₃O₄‑coated), with minimal BET surface area reduction (112.6 → 104.0 m² g⁻¹).
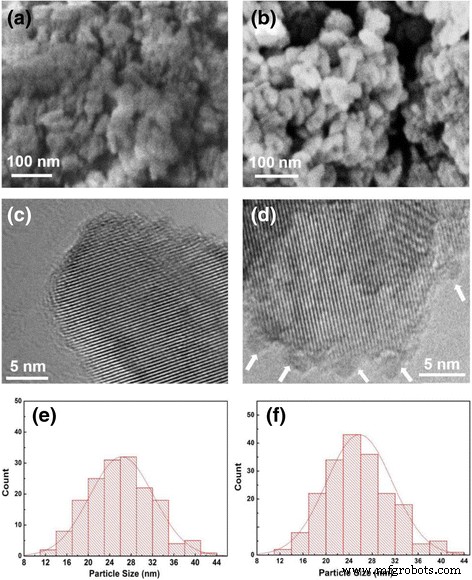
SEM (a, b) and HRTEM (c, d) images of pure P25 and 200‑cycle Co₃O₄‑coated P25 powders along with the particle size distribution (e, f). a, c, e Pure P25 powders. b, d, f 200‑cycle Co₃O₄‑coated P25 powders.
XPS and Composition
Ti 2p and O 1s spectra confirm TiO₂ bonding. Co signals are below XPS detection in 100‑cycle samples, but ICP‑MS quantifies ~3.63 ppm Co, confirming trace Co₃O₄ deposition. Thermal ALD (600 cycles) yields ~0.6 % Co, detectable by XPS.
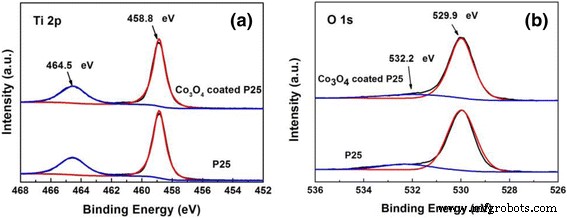
XPS spectra of 100‑cycle Co₃O₄‑coated P25 powders and pure P25 powders. a Ti 2p. b O 1s.
Optical Properties
UV‑vis diffuse reflectance shows negligible change for 200‑cycle samples; 600‑cycle thermal ALD samples exhibit enhanced visible‑light absorption due to Co‑related d‑d transitions. Bandgap analysis yields 3.41 ± 0.02 eV for 200‑cycle Co₃O₄‑coated P25 (essentially TiO₂) and a secondary bandgap of 2.47 ± 0.03 eV for 600‑cycle samples, reflecting Co₃O₄ contributions.
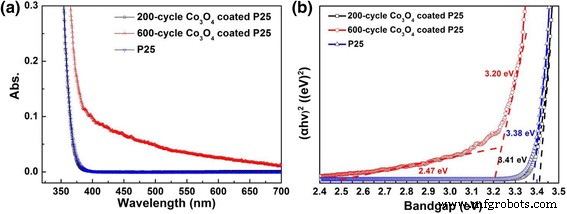
UV‑vis diffuse reflectance spectra (a) and bandgap determination (b) of pure P25, 200‑cycle, and 600‑cycle Co₃O₄‑coated P25 powders.
Photocatalytic Activity
Under UV illumination, 100‑ and 200‑cycle Co₃O₄‑coated P25 achieve ~100 % MB degradation in 1.5 h, surpassing the 80 % rate of bare P25. No degradation occurs in the absence of a catalyst, confirming that UV alone does not degrade MB.
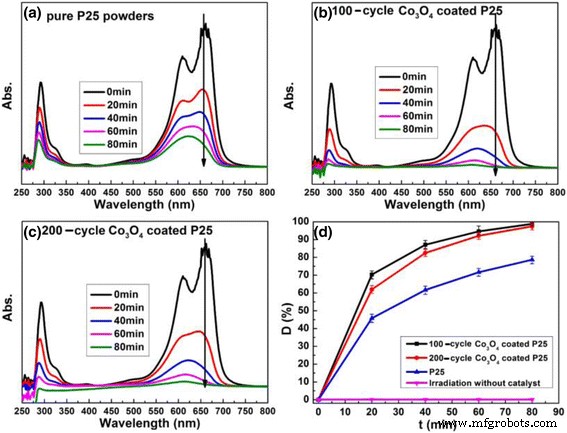
UV‑vis absorption (a) and degradation curves (d) of MB solution with various catalysts. b 100‑cycle Co₃O₄. c 200‑cycle Co₃O₄‑coated P25.
Recycling tests confirm that the 200‑cycle Co₃O₄‑coated P25 retains its activity after three consecutive runs.
Heterojunction Confirmation
Mott‑Schottky plots reveal a positive slope for bare P25 (n‑type) and a negative slope for Co₃O₄‑coated samples, confirming the formation of a p‑n junction that facilitates charge separation.
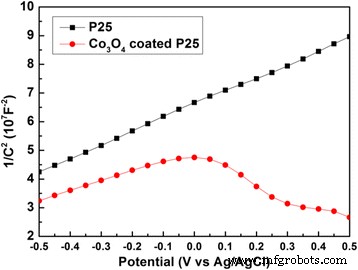
Mott‑Schottky plots of pure P25 and Co₃O₄‑coated P25 in 1 M NaOH at 1 kHz and 2 kHz.
Charge‑Transfer Mechanism
A schematic of the energy band alignment illustrates that photogenerated electrons transfer from Co₃O₄ to TiO₂ while holes move oppositely, creating an internal electric field that suppresses recombination and enhances surface reactions.
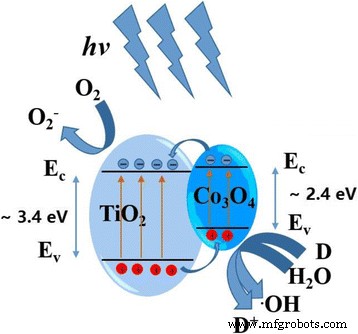
Schematic of energy level and electron‑hole movement in Co₃O₄–TiO₂ p‑n junction under UV irradiation.
Adsorption Enhancement
Zeta potential analysis shows IEPs of MB (5.37), P25 (6.74), and Co₃O₄‑coated P25 (7.42). At the experimental pH (~6.7), MB is negatively charged while both catalysts are positively charged; the higher positive surface charge of Co₃O₄‑coated P25 promotes stronger dye adsorption, contributing to its superior photocatalytic performance.
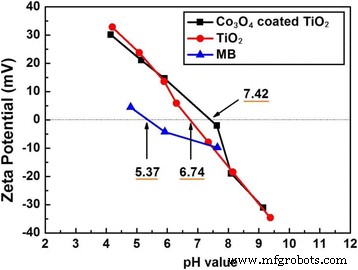
Zeta potential of MB, P25, and Co₃O₄‑coated P25 versus pH.
Visible‑Light Activity
The trace Co₃O₄ coating in 200‑cycle samples does not activate under visible light. However, the 600‑cycle thermal ALD sample, with higher Co loading, achieves ~26 % MO degradation in 120 min, demonstrating that Co₃O₄ nanoparticles provide visible‑light absorption.
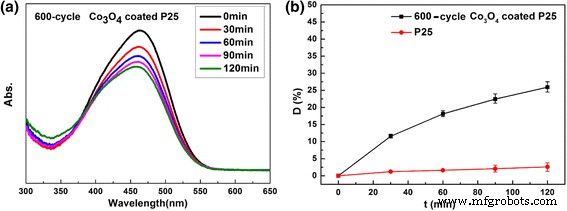
Visible‑light photocatalytic activity of 600‑cycle Co₃O₄‑coated P25 powders. a UV‑vis absorption; b degradation curve of MO under visible light.
Conclusions
Co₃O₄ layers deposited by PEALD form a p‑n heterojunction with TiO₂ without disturbing its anatase structure. The resulting Co₃O₄/TiO₂ nanocomposites exhibit a dramatic rise in UV photocatalytic efficiency, achieving near‑complete methylene blue degradation in 1.5 h. Mott‑Schottky and zeta‑potential measurements confirm enhanced charge separation and dye adsorption. The study validates PEALD as a scalable, low‑temperature route to engineer high‑performance p‑n junction photocatalysts.
Nanomaterials
- Harnessing Atomic Layer Deposition for Next‑Generation Micro‑LEDs and VCSELs
- Optimized HfO2/TiO2/HfO2 Trilayer RRAM: Electrode-Dependent Bipolar Switching and Enhanced Uniformity via Atomic Layer Deposition
- Optimizing HfO₂/Ge Gate Dielectrics: In Situ SiO₂ Interlayers via PEALD Enhance Interface, Electrical, and Band Alignment
- Supercycled ALD with In‑Situ O₂ Plasma Tunes ZnO Film Resistivity and Carrier Concentration Across Five Orders
- Impact of AlN Layer Thickness on Interface and Electrical Performance of ALD‑AlN on c‑Plane GaN
- Low‑Temperature CO₂‑Based PEALD of SiO₂ for Moisture‑Sensitive Applications
- Ultra‑Thin TiO₂ Nanomembranes via Atomic Layer Deposition Deliver Record‑High Capacitance for Pseudocapacitor Electrodes
- Low‑Temperature Plasma‑Enhanced ALD of Cobalt Films with Bis‑Ethylcyclopentadienyl Cobalt (Co(EtCp)₂) as Precursor
- Al₂O₃‑Coated Microchannel Plates via Atomic Layer Deposition Deliver Five‑Fold Gain and Extended Lifetime
- Co‑Sputtering and ALD Fabricated Ag Nanoparticle SERS Substrate: High Sensitivity and 30‑Day Stability for Glycerol Detection



