Low‑Temperature Plasma‑Enhanced ALD of Cobalt Films with Bis‑Ethylcyclopentadienyl Cobalt (Co(EtCp)₂) as Precursor
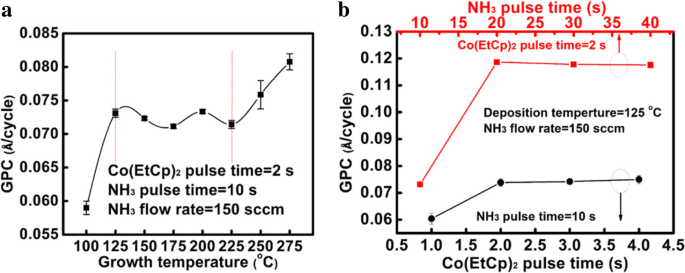
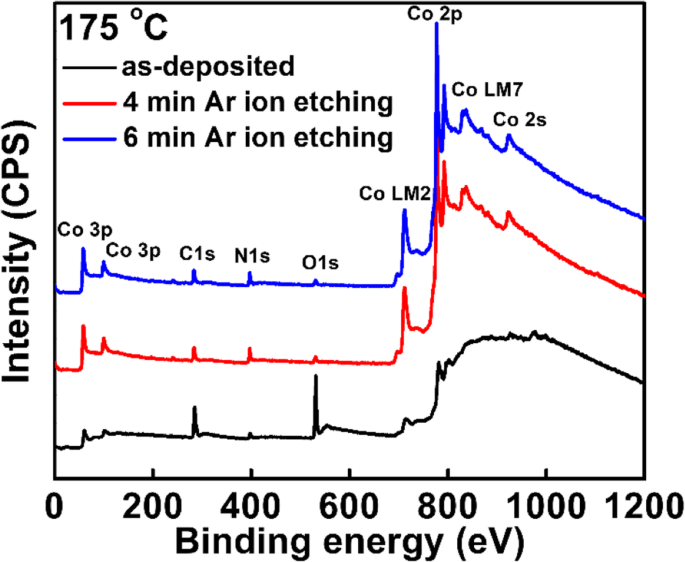
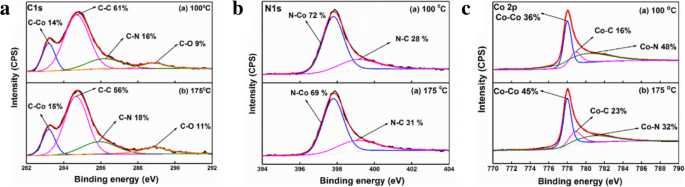
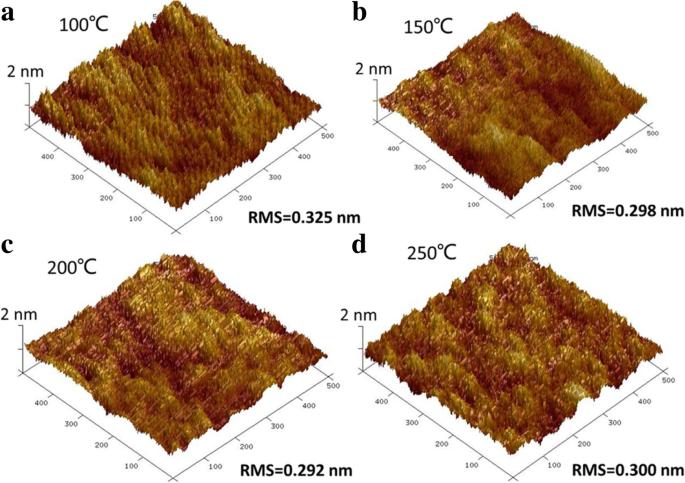
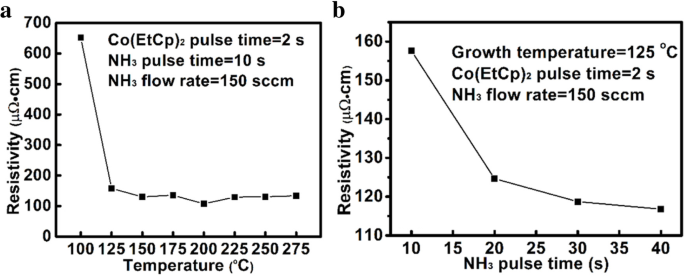
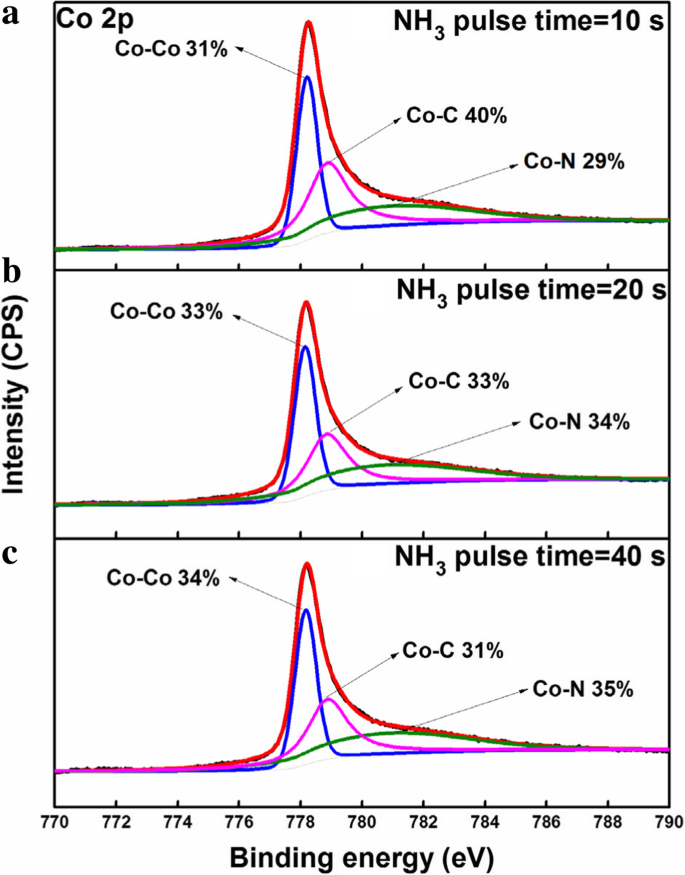
For next‑generation copper interconnects, cobalt films are increasingly explored as a liner and seed‑layer alternative because they wet copper better than tantalum. In this study, cobalt is deposited by plasma‑enhanced atomic layer deposition (PE‑ALD) using bis‑ethylcyclopentadienyl cobalt (Co(EtCp)₂) as the metal precursor. The process window is 125–225 °C, delivering a stable growth rate of ~0.073 Å per cycle. The Et ligand enables growth down to 125 °C, whereas cobalt precursors without the Et group require temperatures above 200 °C. The films contain Co and C, along with measurable amounts of N and O. Extending the NH₃ plasma pulse improves conductivity; a resistivity as low as 117 µΩ·cm is achieved with a 40‑s NH₃ pulse. Root‑mean‑square roughness remains consistently low (~0.3 nm), indicating a smooth film surface. In high‑speed, ultra‑large‑scale integrated circuits, copper interconnects rely on a barrier layer—typically TaN—to block copper diffusion into the surrounding interlayer dielectric (ILD) and a liner—usually Ta—to promote adhesion. Because copper electroplating on Ta alone has poor nucleation, a seed layer is added, forming a TaN/Ta/Cu stack. As device dimensions shrink, the available space for copper diminishes, and the high resistivity of this stack motivates the search for seed‑free barrier/liner systems. Replacing Ta with cobalt improves copper wettability, enabling direct electroplating on Co and eliminating the need for a separate seed layer. Traditional deposition of barrier/liner films uses physical vapor deposition (PVD), which struggles to provide uniform, conformal coverage in high‑aspect‑ratio trenches and vias. Atomic layer deposition (ALD) offers self‑limiting growth, delivering ultrathin, continuous films with excellent step coverage. Many Co ALD precursors based on cyclopentadienyl (Cp) ligands have been examined, including CoCp₂, Co(MeCp)₂, and Co(CpAMD). These precursors typically require temperatures above 200 °C for stable growth. Adding an ethyl group to the Cp ligand lowers the necessary temperature, as the NH₃ plasma more readily reacts with the EtCp ligand. Here we employ Co(EtCp)₂ with NH₃ plasma in a PE‑ALD process, systematically studying how temperature, pulse durations, and post‑annealing affect film growth, composition, and electrical performance. The result is a robust 125–225 °C window and a resistivity of ~130 µΩ·cm for the cobalt films. Cobalt thin films were deposited by PE‑ALD on a 200 nm SiO₂ layer grown by thermal oxidation on p‑type Si substrates. Co(EtCp)₂ was stored at 70 °C and delivered into the reactor via N₂ carrier gas. A remote plasma generator produced NH₃ plasma at 2800 W, serving as the reducing agent. N₂ flow was 50 sccm and the working pressure was ~1000 Pa during growth. Substrate temperature was varied from 100 to 270 °C in 25 °C increments. The pulse times of Co(EtCp)₂ and NH₃ plasma were also varied to optimize growth. Samples deposited at different temperatures were annealed in forming gas (N₂/4 % H₂) at 400 °C for 30 min to assess post‑anneal effects. Film thickness and density were obtained by X‑ray reflectivity (XRR). Microstructure was examined by grazing‑incidence X‑ray diffraction (GIXRD) using a Bruker D8 Discover with Cu Kα radiation. Surface topography was imaged by atomic force microscopy (AFM, Bruker Icon) and scanning electron microscopy (SEM, Zeiss SIGAMA HD). Elemental composition and bonding states were determined by X‑ray photoelectron spectroscopy (XPS, Kratos Axis Ultra DLD). Sheet resistance was measured with a four‑point probe, and resistivity was calculated from thickness and sheet resistance. Figure 1a illustrates the growth rate versus substrate temperature. A steady rate of 0.073 ± 0.02 Å per cycle is achieved between 125 and 225 °C, defining the optimal process window. Below 125 °C the growth slows due to insufficient activation energy; above 250 °C the rate rises because of precursor thermal decomposition. The inclusion of the Et group in Co(EtCp)₂ shifts the window to lower temperatures compared with CoCp₂ and Co(MeCp)₂, confirming the higher reactivity of EtCp ligands with NH₃ plasma. Figure 1b shows how the growth rate depends on the pulse durations of the precursor and the plasma. Increasing the Co(EtCp)₂ pulse from 1 to 2 s raises the rate from ~0.06 to ~0.073 Å per cycle, after which it plateaus, indicating saturation of surface adsorption at 2 s. Extending the NH₃ plasma pulse beyond 20 s also raises the growth rate, saturating near 0.12 Å per cycle. Dependence of the growth rate on a substrate temperature, b Co(EtCp)₂ pulse, and NH₃ pulse time. Survey XPS spectra (Figure 2) confirm that the films comprise Co, C, N, and O. Ar ion sputtering for 6 min removes surface contaminants, as shown by the unchanged C and O contents after 6 min etching. Table 1 lists the elemental percentages for a 175 °C deposit, while Table 2 compares films grown at 100 and 175 °C. Raising the temperature reduces C content (from 40 % to 32 %) and increases N (14 % to 18 %) and O (5 % to 7 %), indicating more complete ligand removal. The survey spectra of the Co films deposited at 100 °C with different etching times. High‑resolution C 1s spectra (Figure 3a) resolve four components at 283.2, 284.7, 286.1, and 288.9 eV, corresponding to C–Co, C–C, C–N, and C–O bonds. With higher deposition temperatures, the C–C fraction decreases, reflecting greater EtCp ligand decomposition. N 1s spectra (Figure 3b) separate into N–Co (397.8 eV) and N–C (399.2 eV) peaks; the N–Co fraction slightly decreases as temperature rises, indicating nitrogen desorption. Co 2p₃/₂ spectra (Figure 3c) show Co–Co, Co–C, and Co–N components at 778, 778.9, and 780.86 eV, respectively. The Co–N fraction drops from 48 % to 32 % between 100 and 175 °C, consistent with reduced nitrogen bonding. High‑resolution a Co 2p₃/₂, b C 1s, and c N 1s XPS spectra of the films deposited at 100 °C and 175 °C. TEM cross‑sections (Figure 4) confirm that films grown at 100 and 250 °C are fully crystallized. AFM images (Figure 5) reveal an RMS roughness that remains flat (~0.3 nm) across the entire temperature range, underscoring the conformality of the ALD process. The cross‑sectional TEM images of the Co films (1200 cycles) deposited at a 100 °C and b 250 °C. AFM images of the Co films (1200 cycles) deposited with Co(EtCp)₂ pulse time of 2 s and NH₃ plasma pulse time of 10 s at different temperatures: a 100 °C; b 150 °C; c 200 °C; d 250 °C. Resistivity measurements (Figure 6) show a dramatic drop from 652 µΩ·cm at 100 °C to 130 µΩ·cm at 275 °C, then plateauing. This trend aligns with an increasing metallic Co fraction. Extending the NH₃ pulse from 10 to 40 s further reduces resistivity from 158 to 117 µΩ·cm (Figure 6b). XPS of films grown with varying NH₃ times (Figure 7) indicates an increase in Co–Co bonds (31 % to 34 %) and a decrease in Co–C bonds (40 % to 31 %) as the plasma time lengthens, reflecting more complete ligand removal and lower resistivity. The resistivity of the Co film as a function of growth temperature (a) and NH₃ pulse time (b). The XPS spectra of the Co films with different NH₃ plasma times: a 10 s; b 20 s; c 40 s. Table 3 compares the performance of Co films grown from different precursors. Co(EtCp)₂ enables growth at the lowest temperature, but also yields a smaller growth rate due to steric hindrance from the larger Et ligands, resulting in sub‑monolayer deposition per cycle. Finally, 1200 cycles of Co deposited on an ALD TaN film at 125 °C produced a continuous, uniform 10 nm film (Figure 8), demonstrating the process’s suitability for integration with existing TaN barriers. The cross‑sectional TEM image of the Co film (1200 cycles) deposited at 125 °C on the ALD TaN surface; inset magnified view. Plasma‑enhanced ALD of cobalt using Co(EtCp)₂ demonstrates a robust 125–225 °C window, delivering high‑quality films with low resistivity (~117 µΩ·cm at 125 °C with a 40‑s NH₃ pulse). The Et ligand promotes ligand removal, reducing carbon content and improving conductivity. The films maintain an ultra‑flat surface (RMS ~0.3 nm) across the entire temperature range and form continuous layers on ALD TaN, indicating strong potential as a seed‑free liner for copper interconnects. Atomic force microscopy Atomic layer deposition Cyclopentadienyl isopropyl acetamidinato cobalt Bis(ethylcyclopentadienyl) cobalt Bis(η-methylcyclopentadienyl) cobalt Bis-cyclopentadienyl cobalt Cyclopentadienyl Interlayer dielectric Plasma‑enhanced Root mean square Scanning electron microscope X‑ray photoelectron spectroscopy X‑ray diffractionAbstract
Background
Methods
Results and Discussion
Optimization of the ALD Process Parameters
Characterization of the Deposited Co Films


Conclusions
Abbreviations
Nanomaterials
- Harnessing Atomic Layer Deposition for Next‑Generation Micro‑LEDs and VCSELs
- Optimizing HfO₂/Ge Gate Dielectrics: In Situ SiO₂ Interlayers via PEALD Enhance Interface, Electrical, and Band Alignment
- Plasma‑Enhanced ALD of Co₃O₄ on TiO₂ Boosts UV Photocatalytic Degradation of Methylene Blue
- Supercycled ALD with In‑Situ O₂ Plasma Tunes ZnO Film Resistivity and Carrier Concentration Across Five Orders
- Low‑Temperature CO₂‑Based PEALD of SiO₂ for Moisture‑Sensitive Applications
- Ru-Based RRAM Devices Show Negative Differential Resistance: Insights from Atomic Layer Deposition Fabrication
- Enhanced Cycling Capacity of Graphene Aerogel/ZnO Nanomembrane Anodes Fabricated via Atomic Layer Deposition
- Al₂O₃‑Coated Microchannel Plates via Atomic Layer Deposition Deliver Five‑Fold Gain and Extended Lifetime
- Co‑Sputtering and ALD Fabricated Ag Nanoparticle SERS Substrate: High Sensitivity and 30‑Day Stability for Glycerol Detection
- Precursors Determine Ferroelectric Performance in ALD Hf0.5Zr0.5O2 Thin Films: TEMA vs. TDMA



