Enhanced Cycling Capacity of Graphene Aerogel/ZnO Nanomembrane Anodes Fabricated via Atomic Layer Deposition
Abstract
We report a ZnO‑nanomembrane/graphene aerogel (GAZ) composite anode fabricated by atomic layer deposition (ALD) that delivers a reversible capacity of 1,200 mAh g⁻¹ at 1,000 mA g⁻¹ after 500 cycles—a >2× capacity gain during the first 500 cycles. By varying ALD cycles, the ZnO content can be precisely tuned, enabling optimization of rate performance and cycling stability. Electrochemical analysis indicates that the pronounced capacity growth originates from an alloying process coupled with the formation of a polymeric layer driven by low‑voltage electrolyte degradation, which stores additional lithium ions. The GAZ composite thus offers a promising route toward high‑capacity, high‑stability anodes for next‑generation Li‑ion batteries.
Background
Lithium‑ion batteries (LIBs) dominate portable electronics due to their safety, high energy density, and low self‑discharge.1–4 However, the graphite anode’s theoretical capacity of 372 mAh g⁻¹ falls short of the energy demands of electric vehicles.5–7 Consequently, researchers have turned to non‑graphitic carbons and transition‑metal oxides such as ZnO, Fe₂O₃, Co₃O₄, and MnO₂, and their composites, as promising alternatives.8–16
ZnO offers nearly twice the theoretical capacity of graphite (978 mAh g⁻¹) and boasts excellent lithium‑ion diffusion and low cost.17–18 Yet, its large volume change (~163%) and poor electronic conductivity hinder cycling stability.18–20 Various strategies—including ZnO nanorods, nanosheets, and carbon composites—have been pursued to mitigate these issues.21–23 Graphene, with its superior conductivity, flexibility, and chemical stability, has been integrated into aerogel architectures (GA) to form 3D networks that combine mechanical strength with high porosity and adsorption.24–26 Such structures can accommodate ZnO volume changes and facilitate ion transport.
In this work, we combine GA with ZnO nanomembranes deposited by ALD to create a GAZ composite anode. The ALD process allows precise control of ZnO thickness via cycle number, enabling systematic tuning of electrochemical properties. We demonstrate that a 100‑cycle GAZ electrode exhibits optimal capacity and rate performance, while also displaying a remarkable capacity increase during cycling attributed to polymer layer formation at low potentials.
Methods
Synthesis of GA
Graphene oxide (GO) was synthesized from natural graphite via a modified Hummers’ method.15 5 mg of dopamine was added to the GO dispersion, followed by 10 min vigorous stirring. Subsequently, 15 mg of L‑ascorbic acid was introduced under continuous magnetic stirring until fully dissolved. The mixture was sealed in a glass vessel and heated at 95 °C for 10 h, converting the brown solution into a black graphene hydrogel. The hydrogel was then placed on a metal plate, immersed in liquid nitrogen to remove soluble species, and subjected to directional freezing from the metal interface to the surface. Freeze‑drying yielded the aerogel, which was further treated in a perfluorooctyltriethoxysilane (PFOES)/ethanol (2 wt.%) bath at 70 °C for 8 h, followed by thorough air drying to produce GA.
Preparation of GAZ Composite
The GA was coated with ZnO nanomembranes in an ALD chamber using dimethylzinc and deionized water as precursors at 150 °C. A typical ALD cycle comprised a 30 ms diethylzinc pulse, 3 s waiting, 15 s N₂ purge, a 30 ms water pulse, 3 s waiting, and 15 s N₂ purge. The ZnO thickness was controlled by the number of cycles: 20, 100, and 300 cycles (GAZ20, GAZ100, GAZ300). Samples were then annealed at 700 °C for 120 min under N₂. Pure GA was annealed similarly for comparison.
Microstructural Characterizations
SEM (Zeiss Sigma) and TEM (Nova NanoSem 450) examined morphology. XRD patterns were recorded on a Bruker D8A Advance with Cu Kα radiation (λ = 1.5405 Å). Energy‑dispersive spectroscopy (EDS) measured composition.
Electrochemical Measurements
CR2016 coin cells were assembled in an argon glove box (H₂O, O₂ < 1 ppm) with Li metal as counter/reference electrodes. The working electrode comprised 80 wt.% GAZ composite, 10 wt.% Super P, and 10 wt.% PVDF binder in NMP. The electrolyte was 1 M LiPF₆ in EC/DEC (1:1 v/v). Galvanostatic cycling was performed on a LAND CT2001A system from 0.01 to 3.0 V. Cyclic voltammetry (CV) used a Zein/IM6 workstation at 0.1 mV s⁻¹ from 0.001 to 3.0 V.
Results and Discussion
The fabrication scheme of GAZ composites is shown in Fig. 1a. GA was first produced via a template‑free freeze‑drying method, then coated with ZnO nanomembranes by ALD. SEM images (Fig. 1b–e) reveal that GA consists of stacked graphene nanosheets, while ZnO coverage increases with ALD cycles: GAZ20 shows sparse ZnO islands, GAZ100 displays uniform nanomembranes, and GAZ300 presents a thick, dense layer. This confirms precise compositional tuning by cycle number.
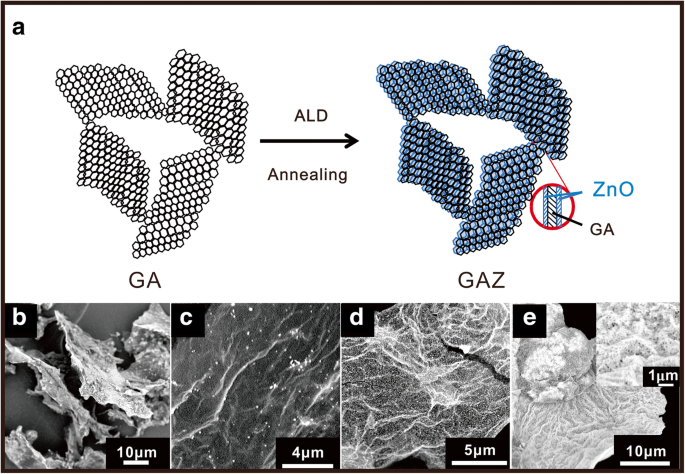
a Fabrication schematic of GAZ composites. SEM images of b GA, c GAZ20, d GAZ100, and e GAZ300. The inset in e is an enlarged SEM image of GAZ300.
EDS analysis (Fig. 2a) confirms the presence of Zn and O, with Zn atomic percentage increasing linearly with ALD cycles. XRD patterns (Fig. 2b) show characteristic wurtzite ZnO peaks (100, 002, 101, etc.) for GAZ100 and GAZ300, while GAZ20 exhibits weak ZnO peaks due to low loading. These results verify successful deposition and tunable composition.
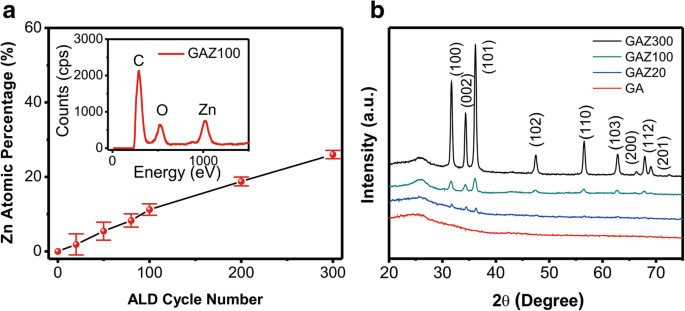
a Atomic percentage of Zn in GAZ composites. The inset shows the EDS spectrum of GAZ100. b XRD patterns of GA and GAZ composites with different ALD cycles.
Rate performance (Fig. 3a) indicates that GAZ100 delivers the highest capacities at 1,500–2,500 mA g⁻¹ (520, 450, and 400 mAh g⁻¹, respectively) and fully recovers to 600 mAh g⁻¹ at 1,000 mA g⁻¹ after cycling. GA20 shows stable capacity at high rates, while GAZ300 exhibits reduced performance due to excessive ZnO thickness hindering Li⁺ transport and exacerbating volume changes. Thus, an optimal ZnO loading exists for balancing conductivity, porosity, and mechanical stability.
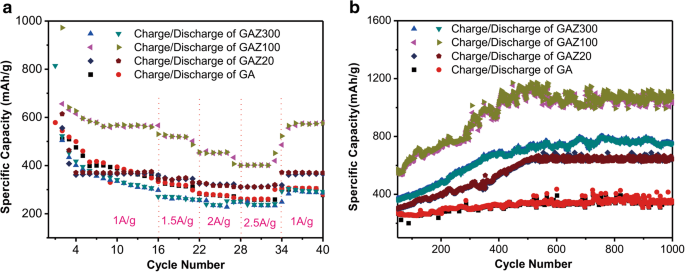
a Rate performance of GA and GAZ composites. b Cycle performance at 1,000 mA g⁻¹.
Long‑term cycling (1,000 cycles at 1,000 mA g⁻¹) reveals a pronounced capacity increase (Fig. 3b): GAZ100 rises from 580 to 1,200 mAh g⁻¹, GAZ300 from 450 to 700 mAh g⁻¹, and GAZ20 from 300 to 600 mAh g⁻¹. The highest areal capacity of GAZ100 reaches 0.61 mAh cm⁻², surpassing the others. Pure GA shows only a modest increase, underscoring that the synergistic effect of ZnO and GA drives the capacity growth. Similar phenomena have been reported for other metal‑oxide anodes and are often linked to reversible polymeric layer formation during low‑voltage electrolyte degradation.7, 32, 38–39
CV analysis of GAZ100 (Fig. 4a) across cycles shows four cathodic peaks at 1.6, 0.9, 0.2, and 0.06 V in the first cycle, corresponding to SEI formation, ZnO reduction, Zn alloying, and GA lithiation, respectively. After 300 and 800 cycles, the 1.6 V peak diminishes, while peaks at 0.9 and 0.2 V shift to 0.62 and 0.30 V, indicating ZnO reduction coupled with polymer layer formation. Anodic peaks shift to higher potentials in subsequent cycles, reflecting increased interfacial resistance and hindered Li⁺ deintercalation. The integrated peak areas grow with cycles, mirroring the capacity enhancement.
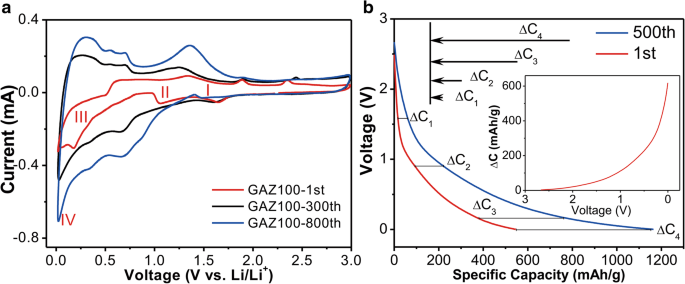
a CV for GAZ100 after different charge/discharge cycles. b Selected discharge voltage profiles. Red and blue lines illustrate the discharge profiles of the 1st cycle and 500th cycle, respectively. The inset shows the capacity difference between the 1st and 500th cycles as a function of discharge voltage.
Discharge profile analysis (Fig. 4b) partitions the capacity increment into four voltage ranges: SEI formation, ZnO reduction, alloying, and GA lithiation. The largest gain (≈ 258 mAh g⁻¹) occurs during alloying, attributed to the polymeric layer’s Li⁺ storage capacity. Additional gains from GA lithiation (≈ 206 mAh g⁻¹) suggest gradual exposure of active sites over cycles.
After 500 cycles, TEM imaging (Additional file 1: Figure S1) shows intact ZnO nanocrystals with clear lattice fringes, confirming structural integrity and supporting the observed cycling stability.
Conclusion
We have demonstrated that GAZ composites, synthesized by ALD, exhibit tunable composition and exceptional electrochemical performance. The optimal 100‑cycle ZnO loading yields a reversible capacity of 1,200 mAh g⁻¹ at 1,000 mA g⁻¹ after 500 cycles—a >2× capacity increase—while maintaining excellent rate capability. Electrochemical analysis attributes this enhancement to a polymeric layer formed at low potentials, which stores additional lithium. The straightforward fabrication route and high capacity position GAZ composites as strong candidates for next‑generation LIB anodes.
Abbreviations
- ALD
Atomic layer deposition
- CV
Cyclic voltammetry
- DEC
Diethyl carbonate
- EC
Ethylene carbonate
- EDS
Energy dispersive spectroscopy
- GA
Graphene aerogel
- GAZ
Zinc oxide/graphene aerogel
- GO
Graphene oxide
- LIBs
Lithium‑ion batteries
- NMP
N‑Methyl‑2‑pyrrolidone
- PFOES
Perfluorooctyltriethoxysilane
- SEI
Solid electrolyte interphase
- SEM
Scanning electron microscopy
- TEM
Transmission electron microscope
- XRD
X‑ray diffractometer
- ZnO
Zinc oxide
Nanomaterials
- Optimized HfO2/TiO2/HfO2 Trilayer RRAM: Electrode-Dependent Bipolar Switching and Enhanced Uniformity via Atomic Layer Deposition
- Hierarchical ZnO/Polyamide‑6 Nanofibers with Enhanced Antibacterial Activity Fabricated by Atomic Layer Deposition and Hydrothermal Growth
- Plasma‑Enhanced ALD of Co₃O₄ on TiO₂ Boosts UV Photocatalytic Degradation of Methylene Blue
- Supercycled ALD with In‑Situ O₂ Plasma Tunes ZnO Film Resistivity and Carrier Concentration Across Five Orders
- High‑Capacity Si/Graphene Composite Anode Fabricated via Magnesium‑Thermal Reduction for Li‑Ion Batteries
- High-Performance Silicon Nanowire Heterojunction Solar Cells Using Al₂O₃ Passivation via Atomic Layer Deposition
- Ultra‑Thin TiO₂ Nanomembranes via Atomic Layer Deposition Deliver Record‑High Capacitance for Pseudocapacitor Electrodes
- Ru-Based RRAM Devices Show Negative Differential Resistance: Insights from Atomic Layer Deposition Fabrication
- Co‑Sputtering and ALD Fabricated Ag Nanoparticle SERS Substrate: High Sensitivity and 30‑Day Stability for Glycerol Detection
- Optimizing ZnO Thin Film Growth by Co-Depositing Ozone and Deionized Water in Atomic Layer Deposition



