Temperature‑ and pH‑Responsive PNIPAM‑Based Nanogels for Dual‑Stimuli Controlled Delivery of β‑Lapachone
Abstract
Poly‑N-isopropyl acrylamide (PNIPAM) nanogels were co‑polymerized with acrylic acid (AAc) to tune the lower critical solution temperature (LCST). The resulting PNIPAM‑co‑AAc nanogels exhibit two distinct volume phase transitions, with the LCST rising as the AAc content increases. When loaded with β‑lapachone (β‑LP), drug release is governed by both temperature and pH, and is strongly influenced by the AAc ratio. Notably, the PNIPAM‑co‑AAc20 formulation retains <10 % drug release at acidic–neutral pH and body temperature, while releasing >70 % at basic pH. These findings demonstrate that temperature‑ and pH‑responsive PNIPAM nanogels are promising carriers for intestine‑specific drug delivery via oral administration.
Introduction
Stimuli‑responsive nanocarriers are increasingly employed in drug delivery, imaging, and diagnostics. Among the triggers used—pH, temperature, redox, magnetic field, light—temperature and pH remain the most widely exploited for controlled release. PNIPAM, a thermo‑responsive polymer, collapses above its LCST (~32 °C) due to hydrophobic interactions, whereas deprotonated carboxyl groups in AAc introduce additional electrostatic repulsion that shifts the LCST upward. Co‑polymerization of PNIPAM with AAc therefore offers a tunable platform that can respond to both temperature and pH. β‑Lapachone, a natural anti‑cancer agent, has limited therapeutic window owing to its rapid metabolism and off‑target toxicity; encapsulation in responsive nanogels can mitigate these drawbacks. In this study, we report the synthesis of PNIPAM‑co‑AAc nanogels with varying AAc content, their dual‑stimuli drug‑release behavior, and their cytotoxic efficacy in fibroblast cells.
Methods
Materials
NIPAM (97 %, Sigma‑Aldrich), N,N′‑methylenebisacrylamide (MBA), AAc, distilled water, EtOH, potassium persulfate (KPS, 98 %, Dae Jung), β‑LP (Natural Products), and PBS were analytical grade and used as received.
Synthesis of PNIPAM‑co‑AAc Nanogels
In a 500 mL flask, 2.26 g NIPAM, 0.154 g MBA, and 0–0.145 g AAc were dissolved in 200 mL water at 75 °C. After nitrogen purging, 37.5 mg KPS was added and the mixture was refluxed for 10 min, producing a turbid solution. The polymer was dialyzed (12–14 kDa) for 7 days, frozen in liquid nitrogen, and lyophilized for 3 days to yield dry nanogels (PNIPAM, PNIPAM‑co‑AAc5, PNIPAM‑co‑AAc10, PNIPAM‑co‑AAc20).
β‑LP Loading
1 mg of dried nanogel was dissolved in 1 mL EtOH, and 0.1 mg β‑LP was added. The mixture was stirred overnight in the dark, then dialyzed (6–8 kDa), frozen, and lyophilized. The resulting β‑LP‑loaded nanogels were stored at 4 °C.
Characterization
Morphology was examined by TEM (200 kV) and FE‑SEM (15 kV). FT‑IR spectra were recorded on a Nicolet 6700 spectrometer. Drug loading and release were quantified by UV–Vis spectroscopy (200–800 nm). LCST was determined by dynamic light scattering (DLS) at 1 °C increments from 30 to 50 °C.
Drug Release Studies
10 mL of β‑LP‑loaded nanogels were placed in 3.5 kDa dialysis tubes and incubated at 24 °C and 37 °C in PBS at pH 2, 4, 7.4, and 8.0. At specified time points (0–12 h), 2 mL aliquots were withdrawn and analyzed by UV–Vis. Cumulative release was calculated relative to the total loaded β‑LP.
Cell Viability Assays
NIH3T3 fibroblasts (2×10^4 cells/well) were treated with free β‑LP, PNIPAM‑co‑AAc5‑β‑LP, or PNIPAM‑co‑AAc20‑β‑LP at 0–20 µg/mL. After 3 h, MTT assay (595 nm) and LIVE/DEAD® staining were performed to assess cytotoxicity.
Results and Discussion
Preparation and Morphology
TEM and SEM images (Fig. 1) revealed monodisperse nanogels with mean diameters of ~250 nm for PNIPAM‑co‑AAc5. Zeta potentials decreased from −13.6 mV (PNIPAM) to −23.6 mV (PNIPAM‑co‑AAc20), reflecting increased surface carboxyl groups.
Temperature‑Responsive Behavior
DLS measurements (Fig. 2) showed two distinct swelling–collapse transitions. The first transition began at ~30 °C, and the second appeared near 40 °C, shifting to 45 °C for PNIPAM‑co‑AAc20. Thus, LCST rises with AAc content, consistent with previous reports.
Dual‑Stimuli Drug Release
At 37 °C, PNIPAM‑co‑AAc20 released only ~61 % β‑LP within 6 h, whereas PNIPAM and PNIPAM‑co‑AAc5 released ~80 %. At pH 8.0, release from PNIPAM‑co‑AAc20 increased by ~10 % compared to neutral or acidic pH (Fig. 3). These results confirm that drug release is synergistically enhanced by basic pH and elevated temperature.
In‑Vitro Cytotoxicity
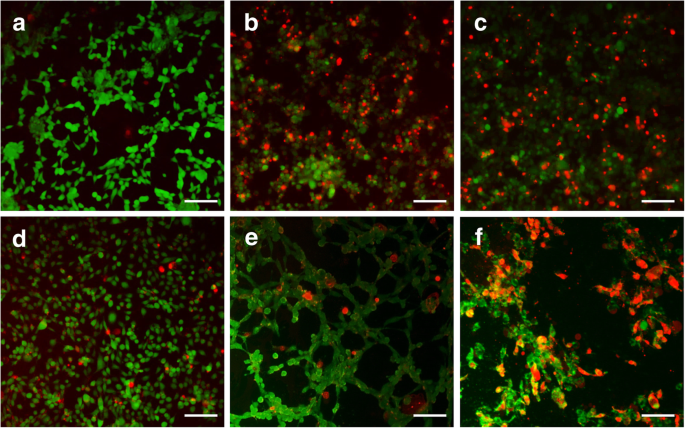
Free β‑LP reduced cell viability more than the nanogel formulations. PNIPAM‑co‑AAc20 displayed higher cell viability at 20 µg/mL, correlating with its lower drug release at 37 °C. Live/dead staining (Fig. 4) showed pronounced cytotoxicity for PNIPAM‑co‑AAc5‑β‑LP at 3 h, while PNIPAM‑co‑AAc20‑β‑LP remained largely non‑toxic until 6 h at pH 8.0, where synergistic cell death was observed.
Conclusions
We successfully engineered PNIPAM‑co‑AAc nanogels whose LCST can be tuned by AAc content. The high‑AAc (20 %) formulation exhibits minimal β‑LP release at physiological temperature but releases the drug efficiently at basic pH, mirroring the environment of the small intestine. These dual‑stimuli responsive nanogels therefore represent a compelling platform for oral, intestine‑targeted delivery of β‑lapachone.
Abbreviations
- AAc
- Acrylic acid
- DLS
- Dynamic light scattering
- FE‑SEM
- Field emission scanning electron microscopy
- FT‑IR
- Fourier‑transform infrared spectrometer
- KPS
- Potassium persulfate
- LCST
- Lower critical solution temperature
- MBA
- N,N′‑methylenebisacrylamide
- PNIPAM
- Poly‑N-isopropyl acrylamide
- TEM
- Transmission electron microscopy
- β‑LP
- β‑lapachone
Figures
Figure 1. TEM (a) and SEM (b) images of PNIPAM‑co‑AAc5 nanogels. (c) Sol–gel transition at 30 °C. (d) Zeta potential vs. AAc content. (e) Hydrodynamic diameter at 30 °C and 50 °C.
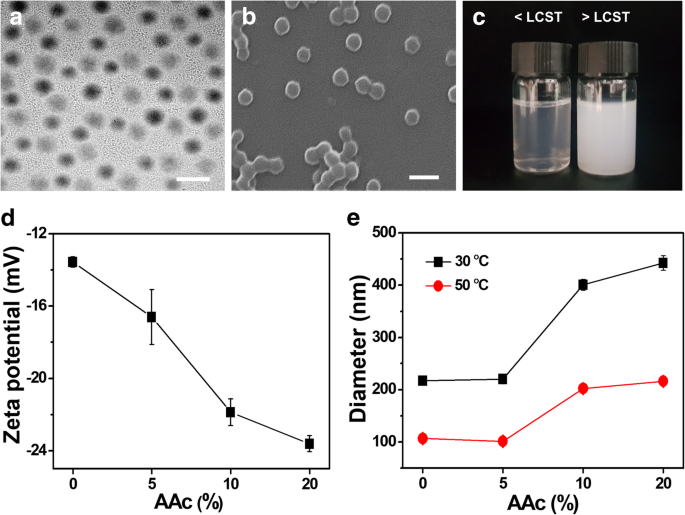
Figure 2. FT‑IR spectra of PNIPAM with 0–20 % AAc.
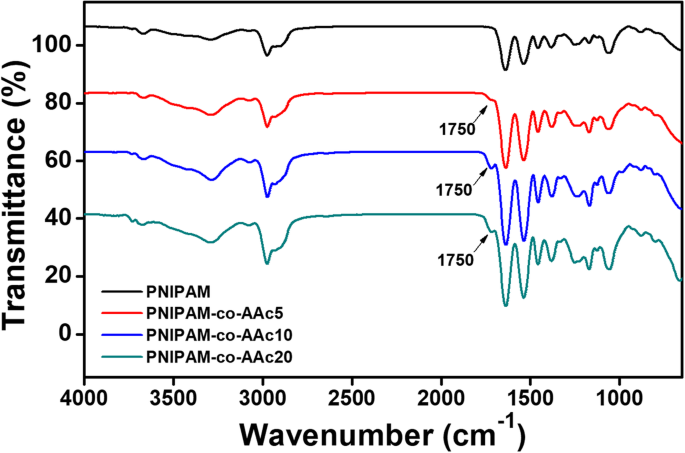
Figure 3. Temperature dependence of hydrodynamic diameter for PNIPAM and PNIPAM‑co‑AAc derivatives at pH 7.4.
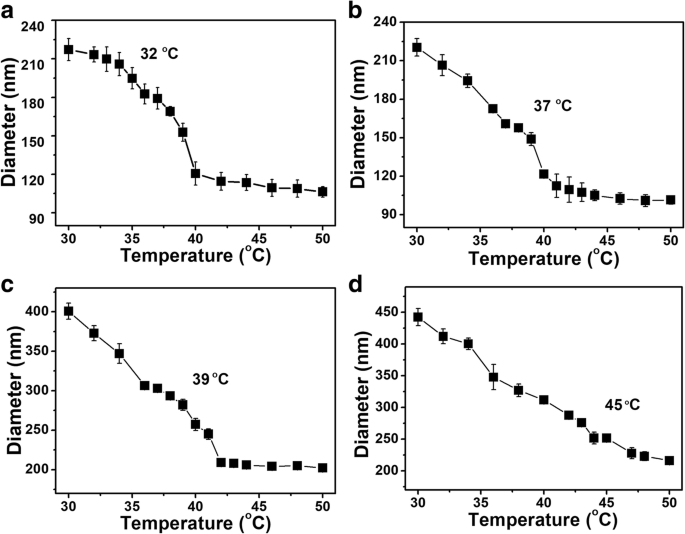
Figure 4. Cumulative β‑LP release from PNIPAM, PNIPAM‑co‑AAc5, and PNIPAM‑co‑AAc20 at 24 °C and 37 °C (pH 7.4).
Figure 5. β‑LP release from PNIPAM‑co‑AAc20 at pH 2, 4, 7.4, and 8.0.
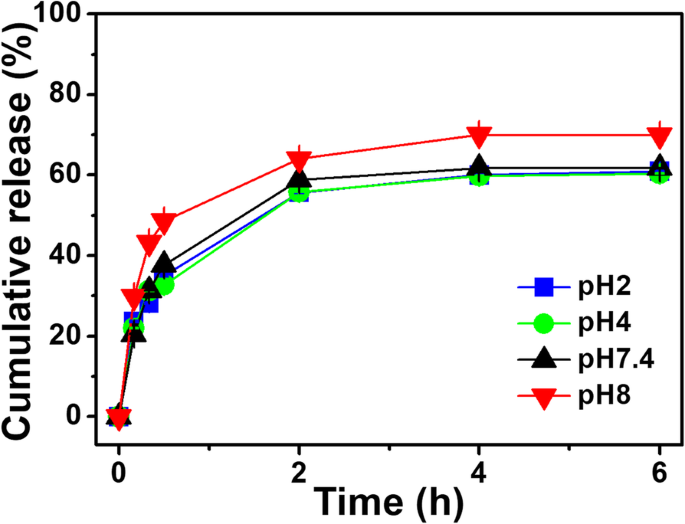
Figure 6. Anti‑proliferative activity of β‑LP and β‑LP‑loaded nanogels on NIH3T3 cells after 3 h at 37 °C.
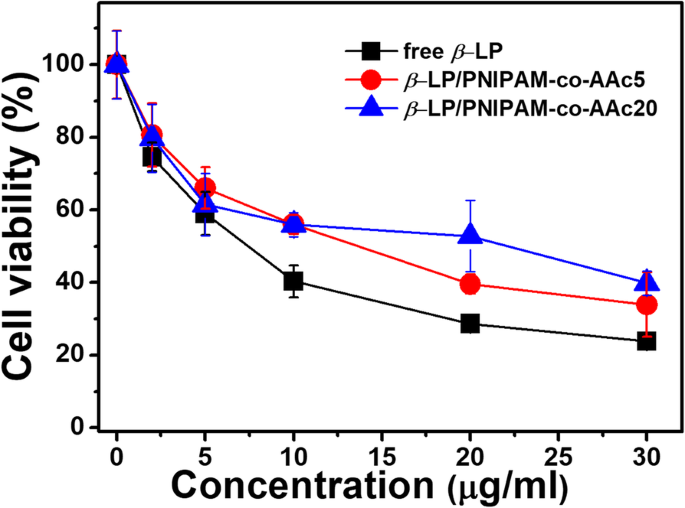
Figure 7. Live/Dead staining of NIH3T3 cells treated with β‑LP, PNIPAM‑co‑AAc5‑β‑LP, and PNIPAM‑co‑AAc20‑β‑LP (pH 7.4 and pH 8.0).
Nanomaterials
- DIY Homebrew Temperature Control: Monitor & Regulate with Arduino
- Precision Thermocouple Measurement and Logging for Lab and Science Experiments
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- High‑Loading 5‑Aminolevulinic Acid–Squalene Nanoparticles: Enhanced Tumor Photodetection and Therapy in Cell Models
- Redox‑Responsive Dual‑Targeting Nanoparticles Co‑Deliver Curcumin to CD44‑Positive Tumor Mitochondria
- Optimized Preparation and Physicochemical Characterization of Dual‑Drug Nanoliposomes Encapsulating Erlotinib and Doxorubicin
- Surface Charge Governs Pullulan Nanoparticle–Human Serum Albumin Complexation and Controlled Drug Release
- High‑Conductivity Nano‑Silver Ink Achieves Low‑Temperature Sintering for Paper‑Based Electronics
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Polydopamine Core–Shell Nanoparticles with Redox‑Responsive Polymer Shells for Targeted Drug Delivery and Synergistic Chemo‑Photothermal Therapy



