Graphitic Carbon Nitride Nanoparticles: Structure, Photocatalytic Properties, Synthesis Methods, and Emerging Applications
Abstract
Graphitic carbon nitride (g‑C3N4) has emerged as a leading material for photocatalytic applications—including CO2 reduction, water splitting, and removal of organic pollutants—as well as for field‑emission technologies. This mini‑review examines recent advances in the crystal structure, synthesis routes, and nanostructuring of g‑C3N4 composites and films. We detail the key structural motifs, discuss scalable preparation strategies, and highlight the material’s versatile photocatalytic performance and future research directions.
Introduction
Solar energy is the most abundant renewable resource, capable of meeting the world’s energy demands for millennia [1]. Its intermittent nature, however, poses challenges for efficient harvesting, storage, and utilization [4]. Technologies that convert solar photons directly into chemical energy—most notably photocatalysis pioneered by Becquerel in 1839 and later advanced by Fujishima and Honda in 1972—offer a promising route for sustainable energy and environmental remediation [3,5]. In particular, photocatalytic degradation of wastewater, a major source of organic contaminants from industrial processes, remains a critical application [3,5].
Polymeric g‑C3N4 is a visible‑light responsive semiconductor with a band gap of 2.7 eV and conduction/valence band positions of –1.1 eV and +1.6 eV vs. NHE, respectively [6]. Its robust thermal, acidic, and alkaline stability [7] and simple synthesis from inexpensive N‑rich precursors such as melamine or urea distinguish it from metal‑based photocatalysts that require costly salts [6,8]. Various synthetic strategies—including thermal condensation, solvothermal, CVD, microwave‑assisted, and hydrothermal methods—enable tailored nanostructures with enhanced photocatalytic activity [9].
Given its versatility in water splitting, CO2 reduction, organic pollutant degradation, and catalytic synthesis, g‑C3N4 has attracted extensive research attention in recent years. This review focuses on the latest developments in its structure, synthesis, and applications, with an emphasis on nanostructuring and composite formation.
Review
Graphitic Carbon Nitride and Photocatalysis
Photocatalysis refers to the acceleration of oxidation–reduction reactions by a semiconductor under light irradiation. The process generates electron–hole pairs that transfer to adsorbed species, producing reactive radicals such as •OH and O2•– [11]. In g‑C3N4, visible‑light excitation creates these charge carriers, enabling water oxidation, hydrogen evolution, and CO2 reduction when coupled with suitable co‑catalysts or sacrificial agents [12‑16].
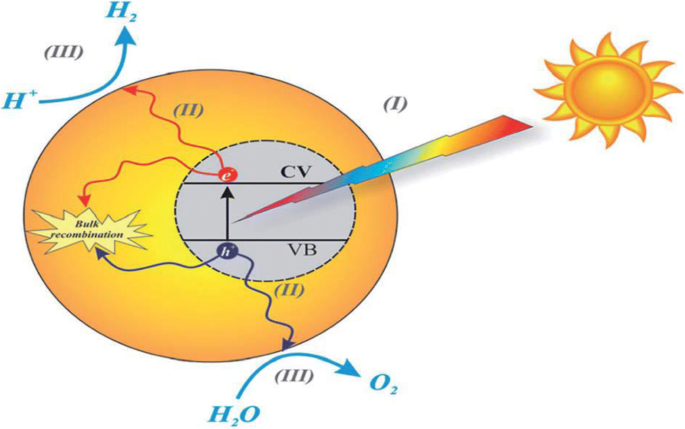
Schematic diagram of the basic mechanisms of the photocatalytic activity of water splitting. Reproduced with permission [113, 114]. Copyright 2015 & 2018. The Royal Society of Chemistry
Unlike conventional oxides such as TiO2 or ZnO, g‑C3N4 features a graphitic‑like framework of sp2 C–N bonds, enabling efficient charge transport and a tunable band structure [26‑30]. The material’s structure can be manipulated via polymorph selection (α, β, or melon‑based networks) and nanostructuring to improve photocatalytic performance [18,32].
Graphitic Carbon Nitride Nano‑Based Particle
One‑dimensional nanostructures of g‑C3N4 exhibit anisotropic electron transport and enhanced surface area, leading to superior photocatalytic activity [17]. 2D nanosheets derived from bulk g‑C3N4 via exfoliation (thermal, ultrasonic, or chemical) provide high dispersion and additional reactive sites [46‑48].
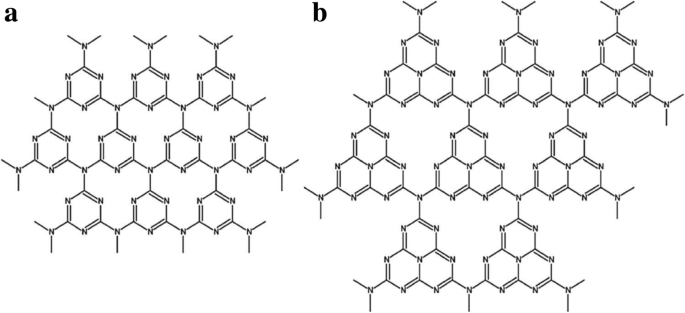
a Tri‑s‑triazine and b tri‑s‑triazine as unit structures of g‑C3N4. Reproduced with permission [25, 31]. Copyright 2008 Royal Society of Chemistry
Structure of Graphitic Carbon Nitride Nano‑Based Particle
g‑C3N4 is a two‑dimensional polymeric network of sp2‑hybridized C and N atoms. Its crystalline structure can be probed by XRD, XPS, and Raman spectroscopy, revealing characteristic peaks corresponding to C–N–C and C=N bonds [19‑23]. The α‑C3N4 phase, identified by Yu et al. [24], is a graphite‑like lattice with interlayer spacing slightly larger than that of graphite, reflecting nitrogen substitution in the graphitic framework [25].
Electronic Structure and Properties of g‑C3N4
First‑principles studies have identified the most stable heptazine‑based polymorphs (phase 1 > phase 2 > phase 3) and highlighted the role of nitrogen lone pairs in shaping the valence band [26,32]. These insights explain the material’s visible‑light responsiveness and its superior photocatalytic performance compared to traditional oxides.
Preparation of Graphitic Carbon Nitride Nano‑Based Particle
Synthesis
Thermal polycondensation of N‑rich precursors (melamine, urea, cyanamide, dicyandiamide) yields bulk g‑C3N4 with low surface area (<10 m2 g−1). Mesoporous and nanoscale variants can be achieved via hard or soft templating, sol–gel, or exfoliation, dramatically increasing surface area (up to 830 m2 g−1) and porosity [35,49,50].

TEM images of TCN (a & b) and MCN (c & d) using a hard templating approach. Reproduced with permission from [120]. Copyright 2015. Elsevier
Techniques Used in Preparing Graphitic Carbon Nitride Nano‑Based Particle
Key synthesis routes include:
- Thermal condensation (high‑temperature calcination of precursors)
- Solvothermal and hydrothermal methods (providing crystalline products without post‑annealing)
- Chemical vapor deposition (CVD) and plasma‑enhanced CVD for thin films
- Microwave‑assisted synthesis (rapid heating, high surface area)
- Sol–gel and physical vapor deposition (PVD) for controlled morphologies
Applications of Graphitic Carbon Nitride
g‑C3N4 demonstrates broad applicability across several domains:
Solar Energy Utilization
By tuning its electronic structure through doping, mesoporosity, or heterojunction formation, g‑C3N4 can drive visible‑light photocatalytic water splitting, CO2 reduction, and solar fuel generation with high efficiency [77,78]. Recent strategies include sacrificial templating to produce mesoporous spheres and rods for enhanced charge separation [79].
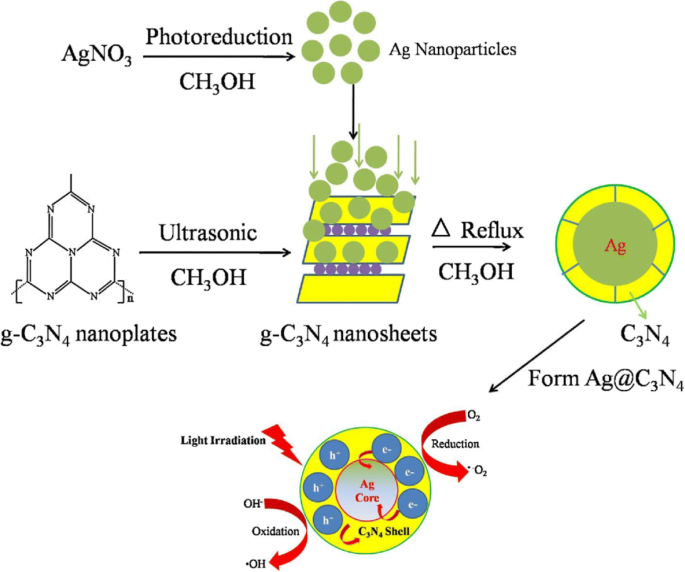
Schematic drawing illustrating synthetic route (templating method) and the mechanism of charge separation and photocatalytic process over C3N4 and Ag@C3N4 photocatalysts under light irradiation. Reproduced with permission [124]. Copyright 2014 Elsevier.
Wastewater and Environmental Treatment
Semiconductor photocatalysts such as g‑C3N4 effectively degrade a wide range of organic pollutants—including dyes, pesticides, and pharmaceuticals—without generating secondary waste [86‑90]. Enhancements via metal (Cu, Fe) or non‑metal (B, C, O, S) doping, heterojunctions, and noble‑metal loading further boost degradation rates [93‑112]. Ultrafine g‑C3N4 nanosheets exhibit superior activity for methylene blue and methyl orange degradation due to their high surface area and reactive edge sites [92,125].
Biomedical and Sensing Applications
g‑C3N4 nanoparticles are attractive for bioimaging, drug delivery, and sensing because of their photoluminescence, biocompatibility, and pH‑responsive behavior. Ultrathin nanosheets can label cell membranes and serve as photosensitizers for cancer therapy [53,53].
Future Perspectives
Advancing g‑C3N4 research requires:
- Developing scalable synthesis routes that yield high‑surface‑area, defect‑controlled nanostructures.
- Systematic evaluation of photocatalytic efficiency, cost, energy input, and recyclability for real‑world applications.
- Exploring multifunctional composites for integrated solar‑fuel production, environmental remediation, and biomedical devices.
Conclusions
g‑C3N4 remains a promising, metal‑free photocatalyst with tunable structure and high stability. Continued innovation in synthesis and nanostructuring will unlock its full potential across energy, environmental, and biomedical fields.
Abbreviations
- g‑C3N4:
Graphite carbon nitride
- TiO2:
Titanium oxide
- ZnO:
Zinc oxide
Nanomaterials
- Top 10 Properties of Aluminium and Their Industrial Applications
- Hafnium Oxide (HfO₂): Structure, Properties, and Key Applications
- Applications and Properties of Manganese Steel: A Comprehensive Overview
- Comprehensive Atomic & Electronic Analysis of Stacked-Cup Carbon Nanofibers
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- How Water Alters the Structure and Dielectric Behavior of Microcrystalline Cellulose
- Au@TiO₂ Yolk–Shell Nanostructures: Tailored Synthesis and Their Superior Visible‑Light Photocatalytic Degradation and SERS Detection of Methylene Blue
- Comparative Analysis of Electrochemical, Biomedical, and Thermal Properties of Natural vs. Synthetic Nanomaterials
- Aluminum‑Doped Zinc Oxide Thin Films: Infrared Optical Properties and Near‑Perfect Absorption Design
- Hydrothermally Synthesized Nano-Copper Sulfide: Superior Adsorption of 17α-Ethynyl Estradiol



