S‑Doped Sb₂O₃ Nanocrystals: Tailored pH‑Controlled Visible‑Light Photocatalysts for Efficient Organic Degradation
Abstract
We report the facile, one‑step hydrothermal synthesis of sulfur‑doped antimony trioxide (S‑Sb₂O₃) nanocrystals using SbCl₃ and thioacetamide (TAA). By adjusting the precursor solution pH, the sulfur content can be precisely tuned, with S²⁻ occupying interstitial sites in the Sb₂O₃ lattice. This doping narrows the bandgap and extends light absorption into the visible region, enabling superior photocatalytic degradation of methyl orange (MO) and 4‑phenylazophenol under visible‑light irradiation. A plausible mechanism involving photogenerated holes and superoxide radicals is proposed.
Background
Semiconductor photocatalytic oxidation offers a sustainable route for environmental remediation by harnessing solar energy to decompose organic pollutants without generating secondary waste [1–6]. Titanium dioxide (TiO₂) remains the benchmark material, yet its wide bandgap (3.2 eV) limits its efficiency under the solar spectrum [7, 8]. Consequently, the design of visible‑light‑active photocatalysts has become a priority.
Visible‑light‑responsive materials can be engineered by (i) synthesizing new narrow‑bandgap semiconductors (e.g., CdS, Sn₂Nb₂O₇, CaBi₂O₄, BiWO₄, SnIn₄S₈) or (ii) modifying conventional UV‑active photocatalysts. Sulfur doping is a versatile strategy that has been successfully applied to TiO₂, In(OH)₃, Zn₂SnO₄, and others, resulting in significant bandgap narrowing and improved photocatalytic performance [18–22]. However, the oxidation state and incorporation mode of S strongly influence the activity, underscoring the importance of controlled doping conditions.
Antimony oxides, including Sb₂O₃ and its complex derivatives, possess crystal structures that promote charge separation, yet most are UV‑responsive. Here, we introduce S²⁻‑doped Sb₂O₃ nanocrystals prepared by a simple hydrothermal route, investigate the effect of precursor pH on sulfur incorporation and photocatalytic activity, and demonstrate their efficacy in degrading MO and 4‑phenylazophenol under visible light.
Methods
Synthesis of S‑Doped Sb₂O₃ Nanocrystals
All reagents were purchased from Aladdin reagent and used without further purification. In a 100‑mL Teflon‑lined stainless‑steel autoclave, 3 mmol SbCl₃ was dissolved in 70 mL deionized water and stirred for 15 min. Subsequently, 4 mmol TAA was added under continuous stirring. The precursor pH was adjusted to 2, 5, 10, 12, or 14 using HCl or NaOH. The mixture was heated at 120 °C for 12 h. The precipitate was washed with distilled water and absolute ethanol, then dried at 70 °C. Samples obtained at different pH values were designated Sb₂O₃‑S‑pH (pH = 2, 5, 10, 12, 14). For the undoped reference, the same procedure was followed without TAA.
Material Characterization
X‑ray diffraction (XRD) patterns were recorded on a Bruker D8 Advance using CuKα radiation (40 kV, 40 mA). UV‑vis diffuse reflectance spectra (DRS) were measured on a Cary 500 Scan Spectrophotometer with BaSO₄ as reference. TEM and HRTEM images were captured on a JEOL JEM‑2010 EX (200 kV). X‑ray photoelectron spectra (XPS) were obtained with a PHI Quantum 2000 system; binding energies were referenced to the C 1s peak at 284.8 eV. Raman spectra were collected on a Renishaw inVia microscope at room temperature.
Photocatalytic Activity Measurements
Photocatalytic tests were performed in a 500‑W halogen lamp setup equipped with 420–800 nm cut‑off filters to ensure visible‑light irradiation. In 80 mL of 3 × 10⁻⁵ mol L⁻¹ MO or 1.2 × 10⁻⁴ mol L⁻¹ 4‑phenylazophenol solutions, 80 mg of catalyst was dispersed. After 1 h of dark adsorption equilibrium, the suspension was irradiated. Aliquots (4 mL) were withdrawn at regular intervals, centrifuged, and the supernatant was analyzed by a Shimadzu UV‑1750 spectrophotometer.
Results and Discussion
Figure 1 shows the XRD patterns of the undoped and S‑doped samples. All peaks match the orthorhombic Sb₂O₃ phase (JCPDS 11‑0689). Samples synthesized at pH ≥ 10 retain the pure phase; lower pH values yield impurities and reduced crystallinity. The relative peak intensities are slightly diminished in S‑doped samples, indicating that TAA moderates crystallite growth.
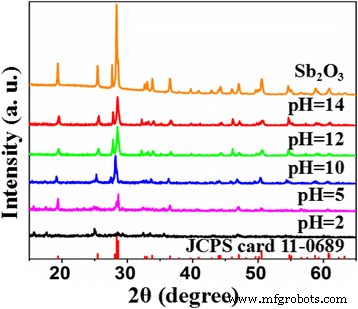
XRD patterns of Sb₂O₃ and S‑doped Sb₂O₃ synthesized at various pHs (pH = 2, 5, 10, 12, 14)
UV‑vis DRS (Figure 2) reveals that undoped Sb₂O₃ absorbs only up to ~380 nm, confirming its UV‑only activity. S‑doped samples exhibit pronounced red‑shifts, with the most significant shift at pH = 14. This bandgap narrowing is attributed to S²⁻ 3p states hybridizing with the valence band, raising its energy and extending absorption into the visible range. Higher pH promotes TAA hydrolysis, generating more S²⁻ and thus higher doping levels.
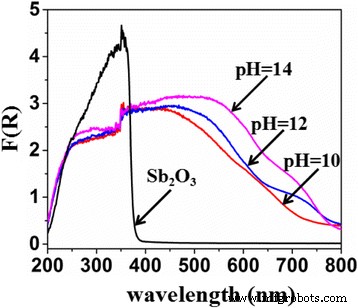
Diffuse reflectance spectra of Sb₂O₃ and S‑doped Sb₂O₃ (pH = 10, 12, 14)
TEM images (Figure 3) reveal that Sb₂O₃‑S‑12 consists of rod‑shaped nanostructures (~40 nm diameter, 100–200 nm length) composed of ~5 nm nanoparticles. HRTEM confirms lattice spacings of 0.25 nm and 0.27 nm, corresponding to (200) and (131) planes. EDS confirms the presence of Sb, O, and S, confirming successful doping.

a TEM. b HRTEM and c EDS of Sb₂O₃‑S‑12
XPS analysis (Figure 4) shows Sb 3d peaks at 539.8 eV (3/2) and 530.5 eV (5/2), indicative of Sb³⁺. Sulfur 2p peaks at 161.5 and 162.7 eV confirm S²⁻ incorporation. The binding energy shift toward lower values with higher S content (pH = 12 vs. 10) reflects increased electron density around Sb. These results corroborate interstitial S²⁻ incorporation.
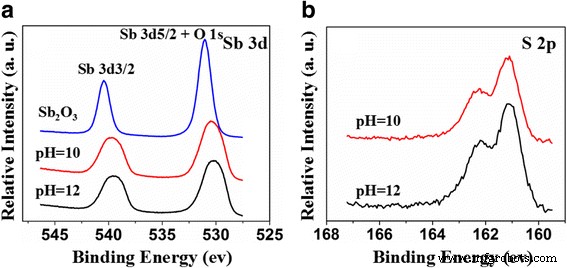
XPS spectra of Sb₂O₃ and S‑doped Sb₂O₃ (pH = 10, 12). a Sb 3d. b S 2p
Raman spectra (Figure 5) show the characteristic Sb₂O₃ peaks (216, 257, 293, 442, 498, 593, 680 cm⁻¹). A new band at 1440 cm⁻¹ appears in the S‑doped sample, and peak broadening indicates increased defect density due to S incorporation.
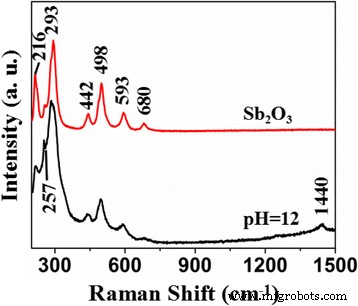
Raman spectra of Sb₂O₃ and S‑doped Sb₂O₃ (pH = 12)
Photocatalytic performance (Figure 6) demonstrates that undoped Sb₂O₃ is inactive under visible light. S‑doped samples exhibit markedly enhanced activity, with Sb₂O₃‑S‑12 achieving 99.2 % MO degradation in 40 min. The activity follows a bell‑shaped trend: low S content (pH = 10) yields insufficient bandgap narrowing; excessive S (pH = 14) introduces recombination centers, reducing performance.
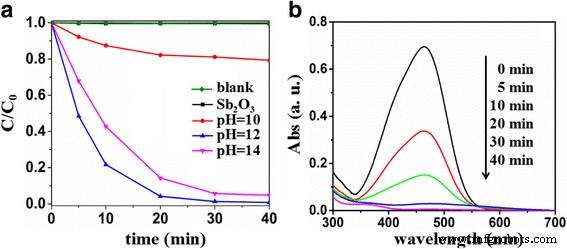
a MO concentration decay under visible light for Sb₂O₃ and S‑doped samples (pH = 10, 12, 14). b Spectral evolution of MO during degradation over Sb₂O₃‑S‑12
Similarly, Sb₂O₃‑S‑12 degrades 94.3 % of 4‑phenylazophenol in 150 min under visible light, confirming its broad applicability (Figure 7).
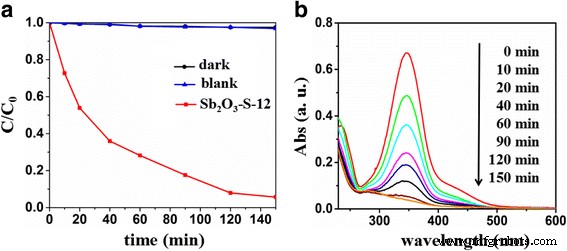
a 4‑phenylazophenol degradation over Sb₂O₃‑S‑12. b Spectral evolution during photocatalysis
Active‑species trapping (Figure 8) using p‑benzoquinone (O₂⁻· scavenger), Na₂‑EDTA (hole scavenger), and tert‑butanol (OH· scavenger) shows that superoxide and photogenerated holes are the primary oxidants; OH· radicals play a negligible role.
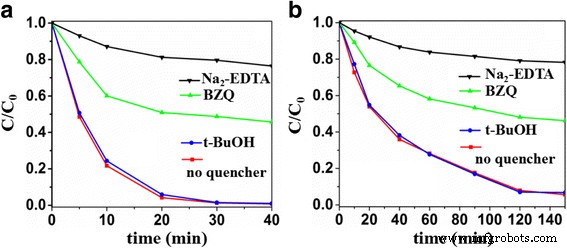
Trapping experiments for MO (a) and 4‑phenylazophenol (b)
Based on these findings, a plausible mechanism is illustrated in Scheme 1: Visible light excites S‑doped Sb₂O₃, generating holes in the valence band and electrons in the conduction band. Electrons reduce adsorbed O₂ to O₂⁻·, while holes directly oxidize the pollutants. The combined action of h⁺ and O₂⁻· yields efficient degradation.
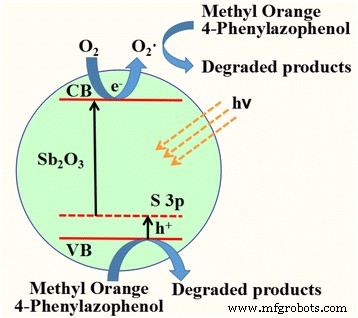
Proposed photocatalytic mechanism of S‑doped Sb₂O₃ (pH = 12) under visible light
Conclusions
We have demonstrated a scalable, one‑step hydrothermal route to produce S²⁻‑doped Sb₂O₃ nanocrystals. Interstitial S²⁻ incorporation extends visible‑light absorption and modulates defect density. By tuning the precursor pH, the sulfur content—and consequently the photocatalytic performance—can be optimized. The sample synthesized at pH 12 delivers the highest activity, achieving 99.2 % MO and 94.3 % 4‑phenylazophenol removal in 40 min and 150 min, respectively, under visible‑light irradiation. The degradation proceeds via photogenerated holes and superoxide radicals, offering a promising route for solar‑driven pollutant remediation.
Nanomaterials
- Why Checklists Drive Plant Efficiency: Proven Benefits and Practical Implementation
- Green One‑Pot Synthesis of Ag‑Decorated SnO₂ Microspheres: A Highly Reusable Catalyst for 4‑Nitrophenol Reduction
- Pd‑Loaded Zr‑MOF for Visible‑Light Photocatalytic Hydrogen Generation
- Triphenylphosphine‑Derived Porous Polymer as a Robust Heterogeneous Catalyst for CO₂‑Driven Cycloaddition to Form Cyclic Carbonates
- Efficient Visible‑Light Hydrogen Production with Eosin Y‑Sensitized g‑C3N4/GO Hybrid Loaded with PtNi Alloy Cocatalyst
- Designing Plasmonic Nanoparticle Strategies for Enhanced Organic Solar Cell Performance
- Ag3PO4/BiFeO3 Heterojunctions: Superior Visible‑Light Photocatalytic Degradation of Acid Orange 7
- One‑Pot Synthesis of Zn(II)‑Porphyrin‑Sensitized TiO₂ Hollow Nanoboxes for Synergistic Visible‑Light Degradation of Organic Dyes
- p-Type Silicon Nanowires: Powerful Catalyst for Efficient Ozone Decomposition
- Enhanced Dye Degradation & Antibacterial Performance of Graphene Oxide‑Doped MgO Nanostructures



