Nano‑Selenium in Monogastric Nutrition: How Gut Microbiota Drives Its Conversion to Active Selenoproteins
Abstract
Recent work in the field of nanotechnology and animal nutrition has highlighted the promise of nano‑selenium (nano‑Se) as a highly bioavailable supplement for poultry and other monogastric species. A recent comprehensive review in the Nanoscale Research Letters detailed the production routes and potential applications of nano‑Se, yet it left the underlying biochemical pathways largely unexplored. Emerging evidence suggests that gut microbiota may oxidize elemental selenium into selenite, H2Se, or selenophosphate, thereby enabling the synthesis of selenoproteins. Further research is required to evaluate the advantages and potential drawbacks of nano‑Se compared with conventional selenium sources, and to clarify its exact role in animal physiology.
Background
A recent review in Nanoscale Research Letters [1] surveyed the growing body of literature on nano‑Se in livestock and aquaculture. The authors described the unique physicochemical properties of sub‑100 nm selenium particles, including their ability to cross biological membranes and serve as delivery vehicles for essential trace elements. Selenium is of particular interest because global soil variability often results in dietary deficiency in both humans and animals [2]. Soil acidification and the use of sulfur‑rich fertilizers reduce selenium bioavailability, leading to low selenium intake in feed grains and oilseeds that form the bulk of monogastric diets. Consequently, selenium supplementation has been a standard practice in poultry, swine, and fish nutrition since the 1970s, primarily through sodium selenite premixes.
While sodium selenite has been effective in eliminating clinical selenium deficiency, it has notable limitations. The compound is chemically reactive, susceptible to reduction by ascorbic acid and other feed constituents, and can volatilize under moist conditions. It also exhibits dose‑dependent pro‑oxidant activity that may compromise gut health. Moreover, its transfer to eggs, placental tissues, and fetal stores is suboptimal, limiting its capacity to buffer stress‑induced selenium demands. Although sodium selenite toxicity is a concern only with over‑mixing or dosage errors, the narrow therapeutic window (typical poultry supplementation ~0.3 ppm versus toxicity >1–2 ppm) necessitates precise feed formulation [3].
Organic selenium forms—most notably selenomethionine (SeMet), selenium‑yeast, and 2‑hydroxy‑4‑(methylthio)butanoic acid‑selenium (OH‑SeMet)—have emerged as superior alternatives. Analytical advances have revealed that >50 % of the total selenium in common feed ingredients exists as SeMet [4], a form readily incorporated into body proteins and efficiently retained for later use during periods of heightened demand. The digestibility and bioavailability of organic selenium surpass those of inorganic salts, making them attractive candidates for precision nutrition programs that aim to match selenium supply to actual physiological needs [5].
Despite these insights, the metabolic fate of nano‑Se remains poorly understood. Current literature has largely focused on a handful of selenoproteins, such as glutathione peroxidase (GSH‑Px), while overlooking the full complement of ~25 selenoproteins identified in mammals and birds [6]. The prevailing hypothesis that nano‑Se may directly convert to selenophosphate and subsequently form selenocysteine (SeCys) lacks experimental validation. Furthermore, claims that nano‑Se can be enzymatically transformed into SeMet are incorrect, as SeMet biosynthesis is confined to plants and certain bacteria [7].
Evidence is mounting that gut microbiota can mediate the redox transformation of elemental selenium. In vitro studies demonstrate that incubation of nano‑Se with lactic acid bacteria such as Lactobacillus delbrueckii subsp. bulgaricus LB‑12 partially dissolves the particles, yielding inorganic selenium species and organic selenium compounds like SeCys and SeMet [8]. Similarly, anaerobic bacteria such as Bacillus selenitireducens and Veillonella atypica reduce elemental selenium to H2Se, a highly reactive intermediate that can be incorporated into selenoproteins [9,10]. These microbial processes involve both oxidative and reductive pathways, often mediated by specific enzymes such as YedE (selenium transporter) and YedF (redox protein) [11]. The extent to which these transformations occur in vivo within the gastrointestinal tract of monogastric animals remains an open question.
The gastrointestinal microbiome of poultry is a complex ecosystem dominated by Firmicutes, Bacteroidetes, Actinobacteria, and Proteobacteria, with Lactobacillus and Enterococcus prevailing in the small intestine and Bacteroides in the ceca [12]. The presence of selenium‑responsive taxa, particularly the anaerobic Veillonella genus, suggests a potential role in selenium metabolism. Moreover, the gut microbiota competes with the host for selenium, and germ‑free animals exhibit lower selenium requirements, underscoring the importance of microbial interactions [13]. Recent studies have also indicated that dietary selenium can alter microbiome composition and diversity, which in turn may influence selenium absorption and retention [14].
In vitro, nano‑Se has been reported to upregulate selenoprotein activity (e.g., GSH‑Px, thioredoxin reductase) and activate the Nrf2 antioxidant pathway [15]. However, these effects may reflect a mild oxidative stress response rather than enhanced selenium supply. The precise relationship between nano‑Se dosage, oxidative stress, and selenoprotein synthesis in vivo remains to be clarified.
From an applied perspective, the benefits of nano‑Se supplementation appear to stem largely from its capacity to prevent performance loss under suboptimal conditions, rather than from direct performance enhancement in adequately balanced diets. Consequently, the commercial viability of nano‑Se as a feed additive hinges on a deeper mechanistic understanding and on robust safety data.
Key challenges for the industrial deployment of nano‑Se include: (1) elucidating the molecular pathways of absorption, distribution, and metabolism; (2) determining whether nano‑Se contributes to long‑term selenium stores; (3) standardizing particle size, coating, and shelf life; (4) harnessing probiotic strains that enhance selenium bioavailability; (5) assessing potential nanotoxicological risks to animals and workers; (6) preventing unintended interactions such as the Trojan horse effect; and (7) distinguishing therapeutic benefits from potential adverse effects, especially in the context of antimicrobial and anticancer claims [16–18].
In summary, while nano‑Se shows promise as a highly bioavailable selenium source, its practical application in poultry and monogastric nutrition will require rigorous investigation into its microbial conversion pathways, safety profile, and cost‑effectiveness.
Conclusion
Over the past decade, research into nano‑selenium has accelerated, yet the critical question of how nano‑Se is metabolized into functional selenoproteins remains unanswered. Current evidence points to the gut microbiota as a plausible mediator, oxidizing or reducing elemental selenium to bioactive forms such as selenite, H2Se, or selenophosphate, which then feed into selenocysteine synthesis (see Fig. 1). Bacterial genera like Veillonella warrant special attention given their prevalence in the avian gut and their documented redox capabilities. Future studies must delineate the specific microbial mechanisms, quantify the efficiency of nano‑Se conversion, and evaluate the net nutritional benefit compared with conventional selenium sources. Only through such comprehensive analysis can the potential of nano‑Se be safely and effectively integrated into monogastric animal production systems.
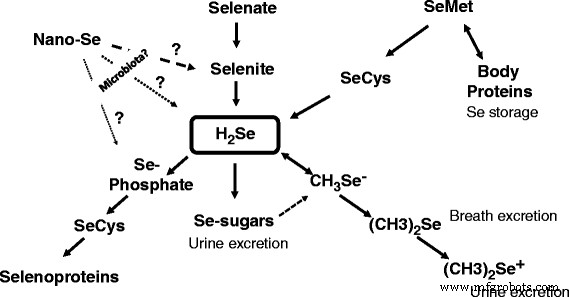
Schematic model depicting the proposed metabolic conversions of selenium species in animals. The diagram illustrates how gut microbiota could transform nano‑Se into selenite, H2Se, or selenophosphate, facilitating selenocysteine synthesis and incorporation into selenoproteins.
Nanomaterials
- Prenatal Exposure to Titanium Dioxide Nanoparticles Alters Maternal Gut Microbiota and Elevates Fasting Blood Glucose in Rats
- Enhanced Dye Degradation & Antibacterial Performance of Graphene Oxide‑Doped MgO Nanostructures
- Why Robot Simulation Drives Success: 3 Key Benefits for Manufacturers and Users
- Transparent Wood & Carbon-Neutral Innovations: The Future of Sustainable Materials
- Key Copper Alloys for Precision Machining: Brass, Bronze, and Beryllium Copper
- Driving Innovation: How a 'Yes' Culture Accelerates Manufacturing and Design Success
- Anodizing Metals: A Professional Guide for Manufacturers
- Nickel 718 Alloy: Composition, Strengths, and Key Aerospace & Defense Applications
- Essential Tube Cutting & Fluid System Tools for Professionals
- Advanced Servo Grippers: Precision Grasping & Force Control



