ZnSe/N‑Doped Carbon Composites Derived from ZIF‑8 Deliver High Capacity and Long‑Term Cycling for Lithium‑Ion Batteries
Abstract
We report the synthesis of three ZnSe/N‑doped carbon (NC) composites with tunable morphologies via a one‑step calcination of ZIF‑8 precursors. By modulating the size of ZIF‑8 particles, the resulting ZnSe/NC nanostructures exhibit distinct particle dimensions and surface areas, which directly influence their electrochemical behavior. The ZnSe/NC‑300 sample delivers a reversible discharge capacity of 724.4 mAh g⁻¹ after 500 cycles at 1 A g⁻¹, while its initial charge/discharge capacities are 906.66/547.48 mAh g⁻¹ at 100 mA g⁻¹. The N‑doped carbon matrix enhances electronic conductivity, provides abundant active sites, and imparts a mesoporous framework that facilitates electrolyte penetration and mitigates volume change. These findings highlight ZnSe/NC composites as promising anode materials for next‑generation lithium‑ion batteries.
Background
Lithium‑ion batteries (LIBs) underpin modern portable electronics and electric vehicles, yet commercial graphite anodes cannot meet the escalating energy demands of high‑power applications. Transition‑metal selenides (TMS) such as SnSe, CoSe, and MoSe₂ have emerged as attractive alternatives due to their high theoretical capacities and robust cycling. Among them, ZnSe offers a compelling combination of low cost, high theoretical capacity, and a conversion‑type reaction that promises superior energy density. However, its practical use is limited by pronounced volume expansion and poor electronic conductivity, which lead to rapid capacity fade.
Engineering nanoscale architectures and integrating conductive carbon matrices can alleviate these drawbacks by buffering mechanical strain and enhancing charge transfer. Nitrogen‑doped carbon, in particular, introduces defects and heteroatom functional groups that increase lithium‑storage sites and improve electronic pathways. Moreover, mesoporous carbon structures provide ample surface area for electrolyte access and reduce diffusion distances.
Metal‑organic frameworks (MOFs) such as ZIF‑8, with their tunable porosity and compositional flexibility, have proven valuable as sacrificial templates for creating nanostructured TMS/carbon composites. Prior work demonstrated ZnSe embedded in N‑doped carbon nanocubes derived from ZIF‑8, achieving initial discharge capacities exceeding 1,100 mAh g⁻¹ and sustained high capacities after 500 cycles. Building on this foundation, we exploit ZIF‑8 to fabricate ZnSe/NC composites with controlled particle sizes, aiming to optimize capacity, rate capability, and long‑term stability.
Methods
Material Preparation
Synthesis of ZIF‑8 Precursors
ZIF‑8 nanoparticles were synthesized by dissolving Zn(NO₃)₂·6H₂O and 2‑methylimidazole in methanol, followed by ultrasonication and 24 h room‑temperature aging. By adjusting the Zn²⁺ precursor concentration, particle sizes of 900, 300, and 40 nm (named ZIF‑8‑900, ZIF‑8‑300, and ZIF‑8‑40) were obtained.
Synthesis of ZnSe/NC Composites
The ZIF‑8 particles were mixed with elemental selenium in a 1:1 mass ratio and calcined at 800 °C for 4 h under argon (2 °C min⁻¹ ramp). The resulting ZnSe/NC‑40, ZnSe/NC‑300, and ZnSe/NC‑900 composites were produced, while commercial ZnSe served as a control.
Material Characterization
X‑ray diffraction (XRD) was performed on a TD‑3500 diffractometer (Cu Kα, 0.15406 nm). Raman spectra (633 nm laser) assessed carbon bonding. BET and BJH analyses measured specific surface area and pore distribution. Morphology was examined by FESEM (FEI Quanta 250) and TEM (FEI Tecnai G2 F20). X‑ray photoelectron spectroscopy (XPS) (Thermo VG ESCALAB 250XI) identified elemental composition and bonding states.
Electrochemical Measurements
Coin cells (CR2032) were assembled in an Ar glove box. Working electrodes comprised 80 wt.% ZnSe/NC (or commercial ZnSe), 10 wt.% acetylene black, and 10 wt.% PVDF, dispersed in NMP. Electrolyte was 1 M LiPF₆ in EC/DMC (1:1). Galvanostatic cycling, CV (0.2 mV s⁻¹), and EIS (0.1 Hz–100 kHz) were conducted on a Neware BTS‑610 and CHI760E, respectively.
Results and Discussion
Figure 1 depicts the fabrication route: ZIF‑8 precursors of controlled size are calcined with selenium to yield ZnSe/NC composites whose morphology follows the original ZIF‑8 template. Rapid nucleation and growth of ZIF‑8 determine the final particle dimensions, which are inherited by the ZnSe/NC products.
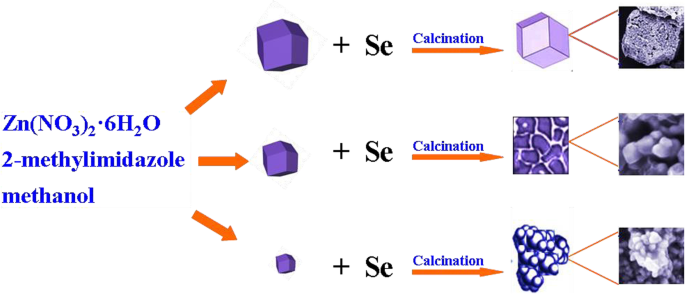
Schematic illustration of the fabrication process of ZnSe/NC composites
Powder XRD (Fig. 2a) confirms the formation of crystalline ZnSe (JCPDS 88‑2345) in all composites. Peak intensities increase from ZnSe/NC‑40 to ZnSe/NC‑900, reflecting the larger grain size inherited from the larger ZIF‑8 templates. Raman spectra (Fig. 2b) show characteristic D (≈1350 cm⁻¹) and G (≈1597 cm⁻¹) bands, with ID/IG values around 1.03–1.04, indicative of a disordered, defect‑rich N‑doped carbon matrix that provides additional lithium‑storage sites.
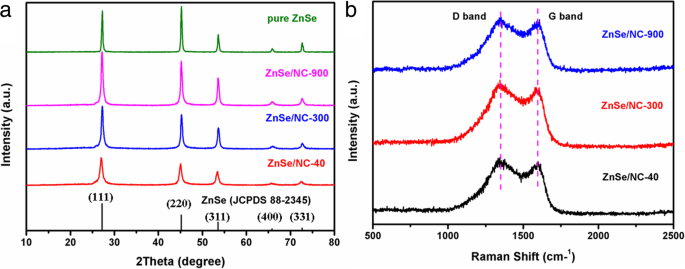
a XRD patterns of pure ZnSe, ZnSe/NC‑900, ZnSe/NC‑300, and ZnSe/NC‑40. b Raman spectra of ZnSe/NC‑900, ZnSe/NC‑300, and ZnSe/NC‑40.
FESEM images (Fig. 3a–c) reveal that ZnSe/NC‑900 retains a rhombic dodecahedron shape with ~100 nm ZnSe cores coated by a thin carbon layer. ZnSe/NC‑300 consists of 20–50 nm nanoparticles encapsulated by carbon, while ZnSe/NC‑40 displays ~10–20 nm particles that tend to agglomerate. BET analysis of ZnSe/NC‑300 (Fig. 3d) shows a type IV isotherm with an H3 hysteresis loop, a specific surface area of 93.9 m² g⁻¹, and an average pore size of 4.4 nm, confirming a mesoporous structure that facilitates electrolyte infiltration and accommodates volume changes.
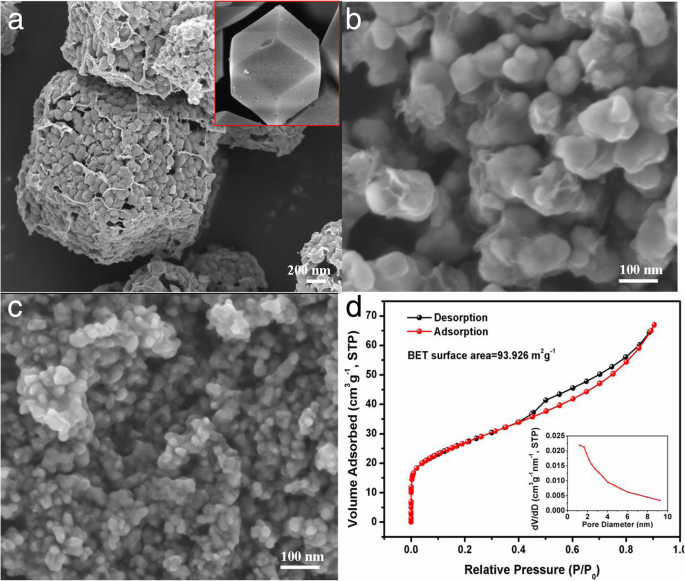
a–c SEM images of ZnSe/NC‑900, ZnSe/NC‑300, and ZnSe/NC‑40; inset: ZIF‑8. d Nitrogen adsorption–desorption isotherms of ZnSe/NC‑300 and pore size distribution.
TEM and HRTEM (Fig. 4) confirm the nano‑granular architecture of ZnSe/NC‑300 and reveal clear lattice fringes at 0.33 nm corresponding to the (111) plane of ZnSe. SAED patterns show polycrystalline rings, consistent with XRD.

a, b TEM images of ZnSe/NC‑300. c HRTEM image. d SAED pattern.
XPS analysis (Fig. 5) confirms the presence of Zn, Se, C, N, and O. Zn 2p peaks at 1021.6 and 1044.7 eV confirm Zn²⁺ in ZnSe, while Se 3d peaks at 53.8 and 54.7 eV denote Se²⁻. Carbon spectra show sp² C (284.7 eV) and N‑doped C (N‑sp² at 285.5 eV, N‑sp³ at 287.5 eV). N 1s peaks at 398.5, 399.3, and 400.7 eV correspond to pyridinic, pyrrolic, and graphitic nitrogen, which act as active sites for lithium insertion and improve electronic conductivity.
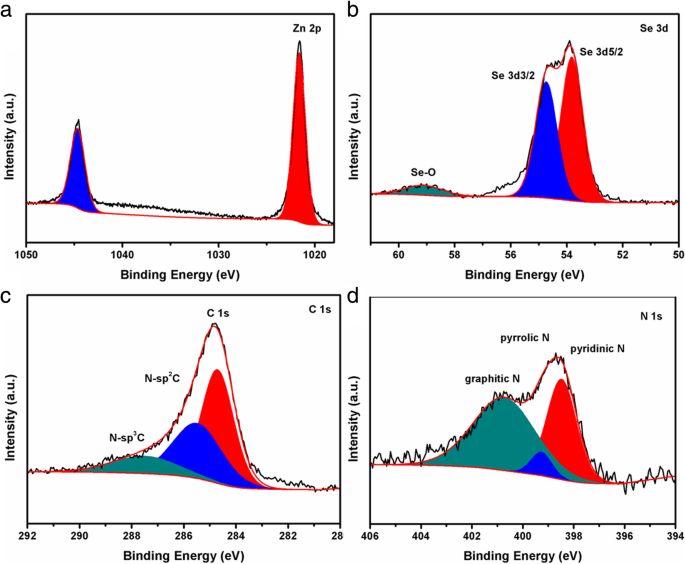
High‑resolution XPS spectra of ZnSe/NC‑300: a Zn 2p, b Se 3d, c N 1s, d C 1s.
CV curves (Fig. 6a) display a SEI‑forming peak at 1.5 V and a conversion peak at 0.35 V in the first discharge, followed by alloy de‑intercalation peaks below 1.0 V and a ZnSe re‑formation peak near 1.4 V. Subsequent cycles show peak shifts due to structural rearrangement. Galvanostatic charge–discharge (Fig. 6b) at 100 mA g⁻¹ shows initial discharge/charge capacities of 906.66/547.48 mAh g⁻¹, with a 60.3 % first‑cycle coulombic efficiency. The capacity rises in later cycles, indicating pseudo‑capacitive behavior and electrode activation.
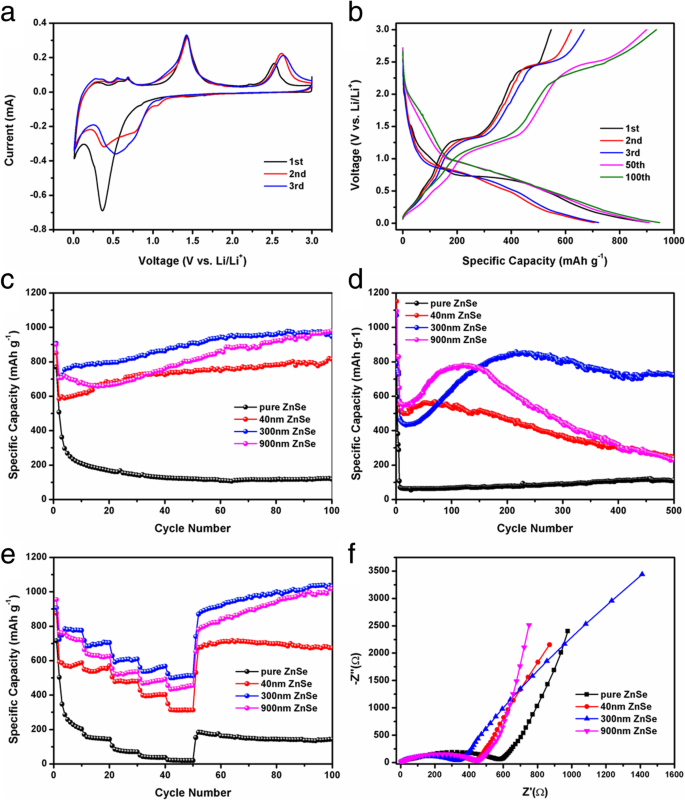
a First three CV cycles of ZnSe/NC‑300. b Charge–discharge curves at 100 mA g⁻¹. c Cycling performance at 100 mA g⁻¹. d Cycling at 1 A g⁻¹. e Rate capability (0.1–2 A g⁻¹). f EIS before cycling.
At 1 A g⁻¹, ZnSe/NC‑300 maintains 724.4 mAh g⁻¹ after 500 cycles, far surpassing ZnSe/NC‑900 (229.5 mAh g⁻¹) and ZnSe/NC‑40 (243 mAh g⁻¹). Rate tests (Fig. 6e) show discharge capacities of 775.6, 704.1, 609.3, 567.7, and 511.6 mAh g⁻¹ at 0.1–2 A g⁻¹, recovering to >1,000 mAh g⁻¹ upon returning to 0.1 A g⁻¹ after high‑rate cycling, confirming structural robustness. EIS (Fig. 6f) reveals lower charge‑transfer resistance for ZnSe/NC composites compared to pure ZnSe, and the resistance decreases further after cycling, indicating improved interfacial kinetics.
Conclusions
We have synthesized ZnSe/N‑doped carbon composites with controllable morphologies using ZIF‑8 as a sacrificial template. The ZnSe/NC‑300 variant exhibits outstanding electrochemical performance: an initial charge/discharge capacity of 906.66/547.48 mAh g⁻¹ and a reversible capacity of 724.4 mAh g⁻¹ after 500 cycles at 1 A g⁻¹. The synergy of N‑doped carbon, high surface area, and mesoporosity enhances conductivity, offers abundant active sites, and buffers volume change, leading to superior cycling stability and rate capability. These results position ZnSe/NC composites as promising anode candidates for next‑generation high‑performance LIBs.
Availability of Data and Materials
All data are fully available without restriction.
Abbreviations
- LIBs:
Lithium‑ion batteries
- NC:
N‑doped carbon
- TMS:
Transition metal selenides
- MOFs:
Metal‑organic frameworks
- XRD:
X‑ray power diffraction
- BET:
Brunauer‑Emmett‑Teller
- BJH:
Barrett‑Joyner‑Halenda
- TEM:
Transmission electron microscopy
- PVDF:
Polyvinylidene fluoride
- EC:
Ethylene carbonate
- DMC:
Dimethyl carbonate
- CV:
Cyclic voltammetry
- EIS:
Electrochemical impedance spectroscopy
Nanomaterials
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Enhancing Lithium‑Ion Battery Cathodes: LiNi0.8Co0.15Al0.05O2/Carbon Nanotube Composite with Superior Electrochemical Performance
- LiNi0.5Mn1.5O4 Cathodes with Cr3+ and F− Composite Doping: Synthesis, Stability, and Electrochemical Performance
- Enhanced Lithium‑Ion Battery Anodes: Polypyrrole‑Coated MnO₂ Core–Shell Micromaterials Deliver Superior Cyclic Stability
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- Scalable Ionothermal Synthesis of Porous Silicon for High‑Performance Lithium‑Ion Battery Anodes
- Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries
- High‑Performance CoSe/NC Anodes from ZIF‑67 Precursors for Lithium‑Ion Batteries
- Advancing Lithium‑Ion Batteries: Binder‑Free Electrode Technology for Higher Energy Density
- NiCo2S4 Nanocrystals on N‑Doped CNTs: A High‑Performance Anode for Next‑Generation Lithium‑Ion Batteries



