Pd Nanoclusters Accelerate Poly(N‑Vinyl‑2‑Pyrrolidone) Combustion via Atomic Oxygen Generation
Abstract
Zero‑valent palladium (Pd(0)) is renowned for converting molecular oxygen into highly reactive atomic oxygen, thereby catalyzing oxygen‑involving reactions. By embedding a trace amount of Pd(0) nanoclusters (≈2.8 nm, 0.3 wt %) into poly(N‑vinyl‑2‑pyrrolidone) (PVP, μW≈10,000 g mol-1), we demonstrate a pronounced enhancement of incineration kinetics. Isothermal thermogravimetric analysis (TGA) reveals that, after a brief induction period, the combustion pathway shifts from second‑order to zero‑order, and the activation energy drops from ~300 to ~260 kJ mol-1 relative to pure PVP. These findings highlight the super‑catalytic potential of Pd nanoclusters for polymer waste treatment and high‑temperature applications.
Background
On the nanoscopic scale, noble metals exhibit a “super‑catalytic effect” [1, 2], arising from increased surface‑to‑volume ratios and altered electronic structures that enhance Lewis acidity of surface sites [1]. The distribution of basal, edge, and corner sites shifts markedly as particle size shrinks, giving rise to higher reactivity and altered selectivity [3, 4, 5].
Polymer incineration, a technologically critical process, involves high‑temperature oxidation of organic polymers. PVP, widely used in cosmetics, biomedicine, and drug formulation, serves as an ideal model polymer to study catalytic combustion relevant to ceramic sintering [8], sensor fabrication [8, 9], battery electrodes [6], waste destruction [10, 11], and solid propellant development [12, 13].
Our work demonstrates that a nano‑sized noble metal catalyst, particularly Pd, can quantitatively convert O2 into reactive O· atoms, thereby accelerating PVP combustion. Pd clusters were synthesized via a facile polyol reduction route, yielding highly dispersed, sub‑3‑nm particles [21, 22].
Thermogravimetric analysis (TGA) provides a reliable kinetic probe for polymer combustion. Isothermal TGA at temperatures just above the PVP ignition point (~370 °C) allows precise determination of reaction order, kinetic constants, and activation energies for both pristine and Pd‑catalyzed systems.
Experimental
Samples were prepared following the polyol method [22]. PVP (30 mM in EG) was dissolved at 90 °C. A 0.35 wt % K2PdCl4 solution (10 mL) was injected into the hot polymer solution (Pd(II):PVP = 1:10). After 120 min, the mixture was cast in acetone to precipitate the Pd/PVP composite, dried, and stored in a desiccator. A second set of samples used commercial <1 µm Pd powder (0.3 wt %) dispersed in PVP for comparison.
Morphology was examined by TEM (FEI Tecnai G2 Spirit twin, 120 kV) and SEM (FEI QUANTA 200 FEG) before and after combustion.
Combustion kinetics were investigated using a Q5000 TA Instruments TGA under flowing air (25 mL min-1) at a heating rate of 10 °C min-1 for dynamic scans, and at 400–440 °C for isothermal runs, recording the full weight loss until completion.
Results and Discussion
Figure 1a displays a TEM micrograph of the Pd/PVP composite, revealing uniformly dispersed, contact‑free clusters with an average diameter of 2.8 ± 0.2 nm (Figure 1b).
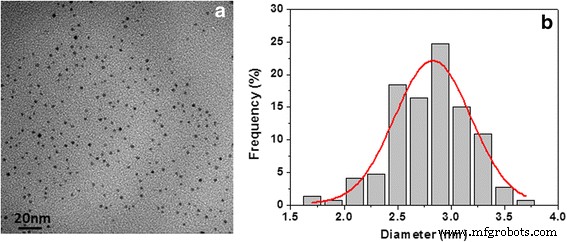
TEM micrograph of nano‑Pd/PVP sample (a) and particle size distribution (b)
SEM imaging (Figure 2) confirms complete combustion of PVP in the presence of Pd, leaving only aggregated Pd clusters. The metallic framework observed after combustion arises from sintering of Pd particles once the organic matrix is removed.

SEM micrograph of the residual TGA product (palladium powder)
Dynamic TGA (Figure 3) shows that the primary weight loss for both pure PVP and Pd/PVP occurs between 400–500 °C. Pd addition accelerates the decomposition: peak rates shift to lower temperatures (≈420 °C for Pd/PVP vs. 540 °C for pure PVP) and the overall curve flattens faster. A residual weight of ~0.3 % corresponds to the Pd content.
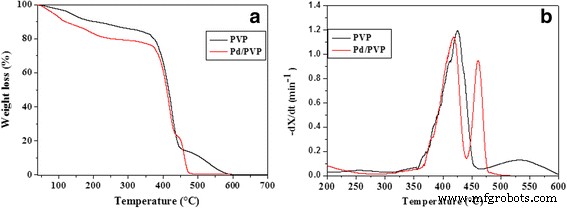
Weight loss (a) and weight loss rate (b) for pure PVP (black) and nano‑Pd/PVP (red) at 10 °C min-1
Isothermal TGA (Figure 4) reveals two distinct stages: an initial period where both systems behave similarly, followed by a rapid, linear decay for Pd/PVP that asymptotically approaches the residual Pd mass. The initial stage reflects oxygen consumption by the polymer; the second stage is dominated by atomic oxygen generation on Pd, which drives the zero‑order kinetics.
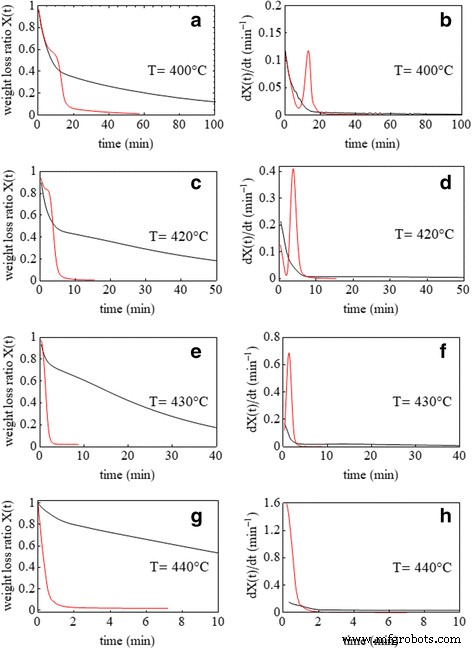
Isothermal weight loss ratio as a function of time during combustion of pure PVP (black curves) and nano‑Pd/PVP (red curves) at 400 °C (a), 420 °C (c), 430 °C (e), and 440 °C (g) and corresponding derivative curves (b, d, f, h)
Regression analysis indicates second‑order kinetics for pure PVP (R²≈0.97) and zero‑order for Pd/PVP (R²=0.98), confirming the catalytic shift. Table 1 summarizes kinetic constants across temperatures.
Arrhenius analysis (Figure 5) yields a pre‑exponential factor A≈1.7×1019 min-1 and activation energy Ea≈260 kJ mol-1 for Pd‑catalyzed combustion, compared to A≈7.7×1021 min-1 and Ea≈300 kJ mol-1 for pure PVP. The reduced Ea aligns with the energy required to dissociate O2 into O· (≈498 kJ mol-1), indicating that atomic oxygen formation on Pd is the rate‑limiting step.
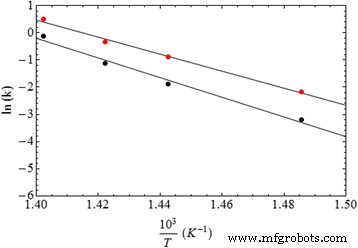
Arrhenius plot for pure PVP (black points) and nano‑Pd/PVP samples (red points)
Comparative isothermal curves (Figure 6) demonstrate that micron‑sized Pd (μ‑Pd) fails to replicate the catalytic acceleration observed with nano‑Pd, underscoring the importance of high surface area and active site density in nanostructured catalysts.
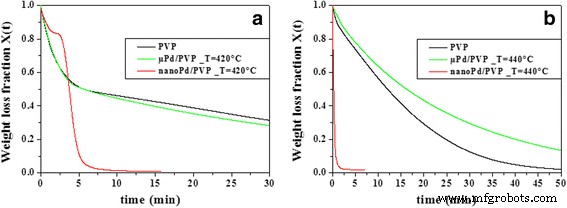
Isothermal weight loss ratio as a function of time during combustion of pure PVP (black curves), μ‑Pd/PVP (green curves), and nano‑Pd/PVP (red curves) at 420 °C (a) and 440 °C (b)
These results confirm that Pd nanoclusters accelerate polymer combustion through atomic oxygen generation, offering a scalable strategy for waste incineration and other high‑temperature polymer processing.
Conclusions
We have shown that 0.3 wt % of ~2.8 nm Pd(0) clusters dramatically enhances PVP incineration. The catalytic pathway is dominated by atomic oxygen formation, which operates at zero‑order kinetics and lowers the activation energy to ~260 kJ mol-1. Compared to micron‑sized Pd, nanoscopic Pd exhibits a super‑catalytic effect, suggesting its potential for efficient polymer waste treatment and other combustion‑based industrial processes.
Nanomaterials
- Understanding the Skin Effect: Impact on AC Conductors and RF Applications
- How Cryogenic Temperatures Affect High‑Performance Plastics: Expert Insights
- Impact of a Trace SiO₂ Addition on the Early‑Stage Sintering of 3Y‑TZP Nanopowders
- Impact of Sulfate Anions on Ultrafine Titania Nucleation During TiCl4 Hydrolysis
- Polyethylene Glycol Enhances NiO Photocathodes for Quantum‑Dot Tandem Solar Cells
- Fe₃O₄–PNIPAAm Nanocomposites: Preparation Method Determines Antibacterial Efficacy and Physicochemical Performance
- Structural, Electronic, and Magnetic Characterization of Ag<sub>n</sub>V (n = 1–12) Clusters via DFT and CALYPSO Search
- Impact of Morphology and Crystal Structure on Titania Nanotube Thermal Conductivity
- How Temperature Influences the Young’s Modulus of Electrospun Polyurethane Nanofibers
- How Inclusions Influence Steel Performance and Strength



