Impact of Sulfate Anions on Ultrafine Titania Nucleation During TiCl4 Hydrolysis
Abstract
We examined how sulfate anions affect the synthesis of nanodispersed TiO₂ via TiCl₄ hydrolysis. The data indicate that the formation of both chelating and bridging bidentate complexes between sulfate and octahedrally coordinated titanium hydroxide monomers governs anatase phase nucleation.
Background
Ultrafine TiO₂ is prized for its broad applicability—from environmental photocatalysis such as degradation of hazardous organics, wastewater treatment, NOₓ and SOₓ abatement, to emerging uses in sensor technology and solar cells. Its catalytic performance, photosensitivity, and adsorption capacity hinge on phase composition, particle size, and surface state. Reducing particle size sharply boosts catalytic activity, while the photocatalytic behavior is highly sensitive to the TiO₂ polymorph—anatase, brookite, or rutile.
Choosing a synthesis route that precisely controls these physical and chemical attributes is essential. The sol–gel technique offers unparalleled flexibility, allowing manipulation of precursor type, hydrolysis conditions, temperature, and pH to steer nanoparticle nucleation and growth. While titanium alkoxides are traditionally used, substituting the cost‑effective TiCl₄ as a precursor holds promise for scalable production. TiCl₄ hydrolysis can be tailored by adding specific ions, which influence the polycondensation pathways and direct the nucleation of desired TiO₂ phases.
The present study focuses on how SO₄²⁻ ions influence oligomeric polycondensation and oxide network formation during TiCl₄‑based sol–gel synthesis.
Methods
TiCl₄ (Merck, 99.9 %, ρ = 1.73 g cm⁻³ at 20 °C) was cooled to 0 °C and diluted with 36 % HCl; the TiCl₄:HCl molar ratio was 2:1. Sodium bicarbonate solution was added dropwise to the titanium oxychloride sol to reach pH 5.0–5.5 under vigorous stirring, during which gelation occurred. The nanoparticle suspension was aged at 80 °C for 3 h, then washed with distilled water to remove Na⁺ and Cl⁻. The precipitated TiO₂ was dried at 150 °C, yielding material S1. For S2, crystalline Na₂SO₄ was introduced directly into the TiCl₄ during hydrolysis, following the same protocol.
X‑ray diffraction (DRON‑4‑07, Cu Kα, 40 kV, 30 mA, Bragg–Brentano geometry, Ni Kβ filter) provided phase analysis. Scherrer’s equation, with K = 0.9, λ = 0.154 nm, and peak FWHM β, calculated coherent scattering domain sizes. FTIR spectra (Thermo‑Nicolet Nexus 670, 4000–400 cm⁻¹) were obtained from KBr pellets. Transmission electron microscopy (JEOL JEM‑100CX II, 100 kV) examined morphology.
Results and Discussion
The inclusion of Na₂SO₄ markedly altered the phase composition (Fig. 1). Sample S1, synthesized without sulfate, contained 65 ± 4 wt % anatase and 35 ± 5 wt % rutile. Coherent scattering domains measured 14 nm for anatase and 9 nm for rutile, indicating good crystallinity, yet a halo in the 2θ = 16–32° range suggested some amorphous character. S1’s specific surface area was ~152 m² g⁻¹.
In contrast, S2 was predominantly amorphous ultrafine titania with discernible anatase features. Its halo was narrower and shifted to higher 2θ, and the domain size averaged 4–5 nm. The specific surface area increased to 328 m² g⁻¹.
Figure 1 illustrates the XRD patterns of S1 and S2.
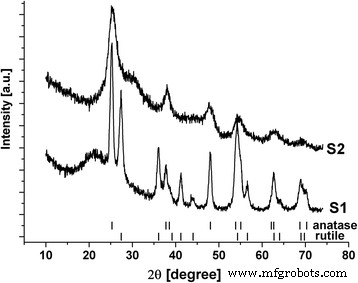
XRD patterns of S1 and S2 materials
TEM images of S1 (Fig. 2a) show lamellar‑like primary particles 10–15 nm in size, with no distinct crystalline boundaries (Fig. 2b). S2 exhibits bubble‑shaped agglomerates (Fig. 2c,d). High‑resolution TEM reveals lattice fringes with interplanar spacings of 0.34–0.37 nm, matching the anatase (101) plane (0.352 nm), indicating preferred growth along the [010] axis.

TEM images of the samples S1 (a, b) and S2 (c, d)

HR TEM images of S2 material showing {101} fringes
FTIR spectra show a broad band around 3400 cm⁻¹, attributed to chemisorbed OH groups, with a shift from the typical 3700–3600 cm⁻¹ range indicating hydrogen bonding. A band near 1600 cm⁻¹ signals adsorbed water. S1’s higher crystallinity yields more distinct TiO₂ bands (400–700 cm⁻¹). S2 displays additional peaks at 1139 and 1060 cm⁻¹, characteristic of chemisorbed SO₄²⁻. The 1384 cm⁻¹ band corresponds to S=O stretching of sulfate‑modified oxides. Two binding modes—chelating (one Ti⁴⁺ via two oxygens) and bridging (two Ti⁴⁺)—produce distinct vibrational signatures between 900–1300 cm⁻¹.
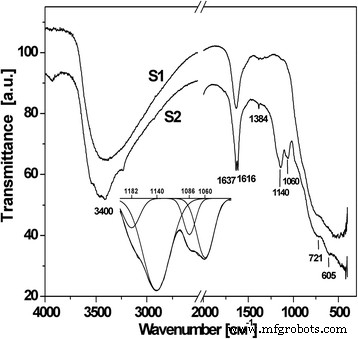
FTIR spectra of S1 and S2 materials
Deconvolution of the 1200–1000 cm⁻¹ region in S2 reveals peaks at 1182, 1140, 1086, and 1060 cm⁻¹, confirming coexistence of chelating and bridging sulfate complexes. These complexes influence the olation pathways that lead to anatase nucleation.
Figure 5 proposes that at pH ≈ 5.5, sulfate ions form Ti(SO₄)(OH)₂(H₂O)₂ chelating complexes and Ti₂(SO₄)(OH)₆(OH₂)₂ bridging complexes. Olation between these species, either via apical edges (chelating) or equatorial planes (bridging), yields skewed zig‑zag tetranuclear Ti clusters and initiates anatase growth.
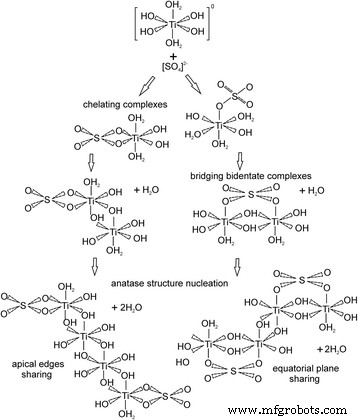
Proposed mechanisms of anatase phase nucleation with chelating and bridging SO₄²⁻ complexes
Conclusions
The study demonstrates that both the reaction pH and the presence of sulfate anions dictate TiO₂ nucleation during TiCl₄ hydrolysis. Sulfate ions form chelating Ti(SO₄)(OH)₂(H₂O)₂ and bridging Ti₂(SO₄)(OH)₆(OH₂)₂ complexes, which, through distinct olation routes, promote the formation of screw polymer chains and anatase nucleation.
Nanomaterials
- Pentodes: The Fifth Element for Enhanced Tube Performance
- Understanding the Skin Effect: Impact on AC Conductors and RF Applications
- Diagnosing a Persistent Gearbox Noise: Lessons from a Refurbished Unit
- Polyethylene Glycol Enhances NiO Photocathodes for Quantum‑Dot Tandem Solar Cells
- Binder Selection for Copper Oxide Anodes: Why SBR+CMC and LA133 Outperform PVDF in Lithium‑Ion Batteries
- Impact of Morphology and Crystal Structure on Titania Nanotube Thermal Conductivity
- Thermal Annealing Enhances Emission in InP/ZnS Quantum Dot Films: Photoluminescence Insights
- How Temperature Influences the Young’s Modulus of Electrospun Polyurethane Nanofibers
- How Inclusions Influence Steel Performance and Strength
- Streamlining Production: Embrace Automation From Day One



