Impact of Decomposed PbI₂ on Scattering Mechanisms in CH₃NH₃PbI₃ Perovskite Films
Abstract
Hybrid organic‑inorganic perovskites (HOIPs) are celebrated for their long carrier diffusion lengths, high absorption coefficients, and outstanding photovoltaic performance. Charge‑transport properties, particularly microscopic scattering mechanisms, are central to device efficiency. In this study, CH₃NH₃PbI₃ (MAPbI₃) films were fabricated via a vapor‑solution reaction. Temperature‑dependent Hall measurements were employed to probe scattering in these films. Two distinct temperature‑mobility trends emerged after different thermal treatments, revealing varied scattering processes. We found that decomposed PbI₂—formed at grain boundaries (GBs) during annealing—dominates the scattering behavior. The passivation role of PbI₂ in MAPbI₃ films was further examined, underscoring its influence on charge transport.
Background
HOIPs have rapidly advanced as high‑efficiency optoelectronic materials for photovoltaics, photodetectors, LEDs, and lasers. Perovskite solar cells now routinely exceed 20% efficiency, driven by their favorable band gaps, high absorption, and scalable processing. However, a comprehensive understanding of charge‑transport mechanisms—especially scattering at GBs—remains incomplete. Previous studies report carrier mobilities in the 1–10 cm²/V·s range, often limited by GB barriers and acoustic‑phonon scattering. Conflicting evidence exists regarding GB impact: some work indicates potential barriers reduced under illumination, while others point to deep traps at GBs. This study addresses these gaps by examining how decomposed PbI₂ influences scattering in MAPbI₃ films.
Methods
MAPbI₃ films were prepared by vapor‑solution reaction, following established protocols. PbI₂ (99.99%) was dissolved in DMF (99.9%) at 1 mol L⁻¹ and stirred at 70 °C for 3 h. Films were spin‑coated at 4000 rpm for 30 s and annealed at 70 °C for 10 min. MAI powder and PbI₂ films were placed in separate zones of a tubular furnace under vacuum. After 10 min pumping, the zones were heated to 180 °C (MAI) and 140 °C (PbI₂) for >100 min. Resulting MAPbI₃ films, darkened in color, were subsequently annealed at 100 °C, 120 °C, or 145 °C for 1 h after isopropanol rinsing. All steps were conducted in ambient air (≈45 % RH).
Microstructure was characterized by XRD (Bruker MXP‑III) and SEM (Hitachi S‑3400 N‑II). Time‑resolved photoluminescence (TRPL) employed a fluorescence spectrophotometer with time‑correlated single‑photon counting (FLS920, Edinburgh). Temperature‑dependent Hall measurements (LakeShore 8400 series) used Al electrodes in a van der Pauw geometry under 0.6 T. Measurements spanned 300–350 K in 10 K steps with an Ar atmosphere.
Results and Discussion
Figure 1 shows XRD patterns before and after annealing. Samples annealed at 120 °C retained pure MAPbI₃ peaks at 14.04°, 28.42°, and 43.08° (110, 220, 330). Annealing at 145 °C introduced a new peak at 12.56°, corresponding to the (001) plane of PbI₂, indicating perovskite decomposition above 145 °C. This aligns with prior reports that thermal treatment above 145 °C releases CH₃NH₃⁺, leaving PbI₂.
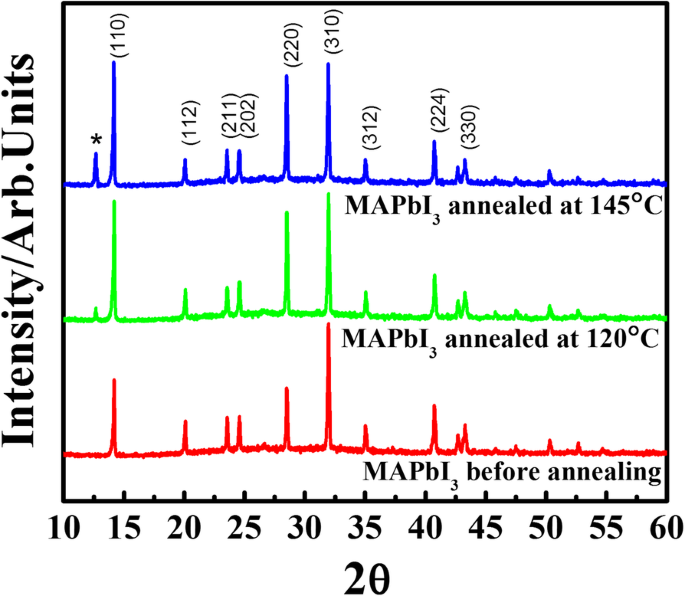
The XRD pattern for MAPbI₃ films before and after annealing at 120 °C and 145 °C
SEM images (Figure 2) reveal compact, conformal films across all treatments. At 145 °C, bright contrast at GBs suggests PbI₂ formation, corroborating the XRD data. Thus, annealing above 145 °C drives PbI₂ segregation to GBs.
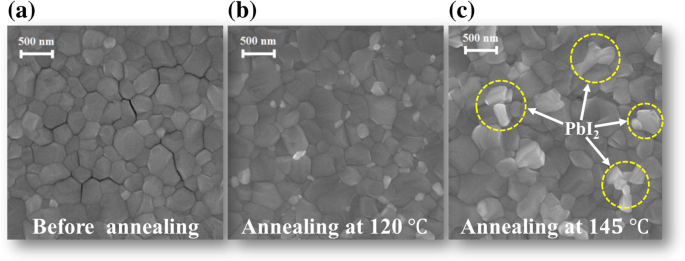
The SEM images for MAPbI₃ films before (a) and after annealing at 120 °C (b) and 145 °C (c)
Hall mobilities (Figure 3) were ~0.6–1 cm²/V·s for unannealed, 100 °C, and 120 °C films, matching literature values. Remarkably, the 145 °C film achieved ~5 cm²/V·s—an order of magnitude increase. This suggests reduced scattering, likely due to PbI₂ passivation of GBs, lowering potential barriers.
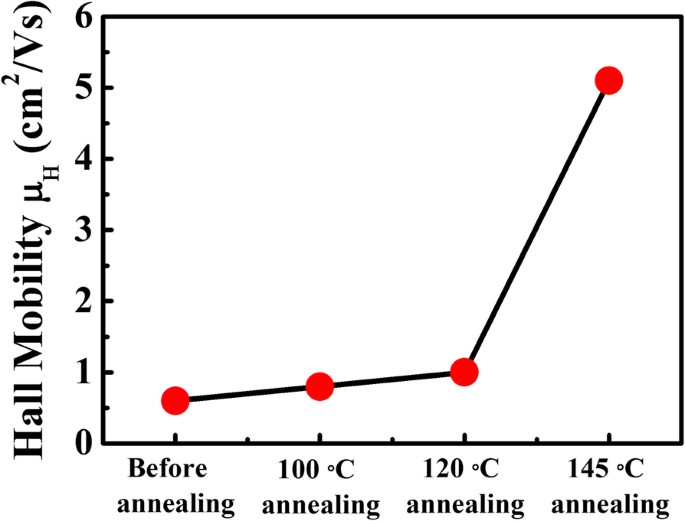
Hall mobilities of all MAPbI₃ films at room temperature
Temperature‑dependent Hall data (Figure 4a) show that mobility rises with temperature for unannealed and 120 °C films—characteristic of GB‑limited transport, where carriers overcome potential barriers. Applying Seto’s model, ln μ versus 1000/T yields barrier heights of 208 meV (unannealed) and 147 meV (120 °C). In contrast, the 145 °C film displays a T⁻².⁰ dependence, indicative of acoustic‑phonon scattering and negligible GB influence. Thus, decomposed PbI₂ at GBs effectively suppresses barrier heights, shifting the dominant scattering mechanism.
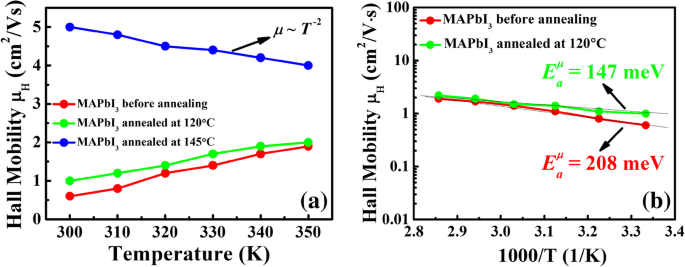
a, b Temperature-dependent Hall mobilities of the MAPbI₃ films before and after annealing at 120 °C and 145 °C
TRPL analysis (Figure 5, Table 1) reveals a stable long‑lived component (~20 ns) across all samples, while the fast component increases from 1.39 ns (unannealed) to 6.05 ns (145 °C). The extended fast decay reflects reduced surface/interface recombination, further confirming PbI₂ passivation at GBs.
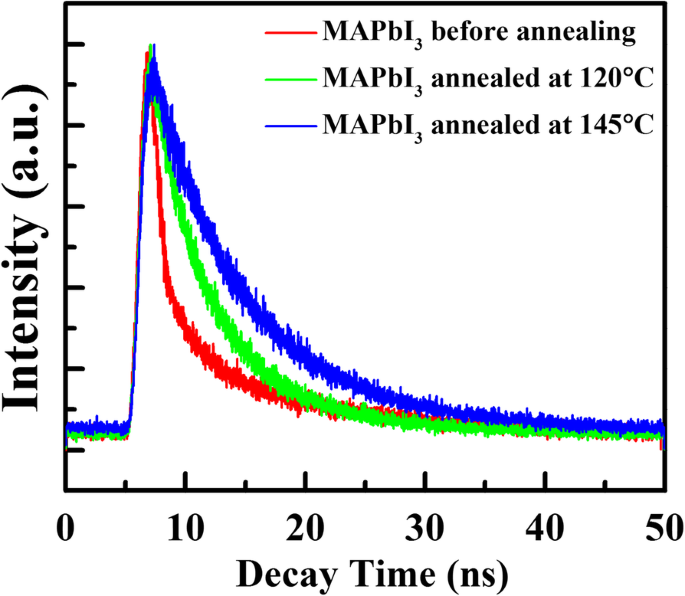
The TRPL decay spectra of the MAPbI₃ films before and after annealing at 120 °C and 145 °C
Conclusions
MAPbI₃ films produced via vapor‑solution reaction exhibited pure perovskite phases and typical optoelectronic properties. Thermal annealing at 145 °C induced PbI₂ formation at GBs, which effectively passivated GB barriers. This passivation suppressed GB scattering, shifted the dominant mechanism to acoustic‑phonon scattering, and elevated Hall mobility to 5 cm²/V·s—substantially higher than the 0.6 cm²/V·s of unannealed films.
Availability of Data and Materials
The datasets supporting this study are available from the corresponding author upon request.
Abbreviations
- GBs:
Grain boundaries
- HOIPs:
Hybrid organic‑inorganic perovskites
- MAPbI₃:
CH₃NH₃PbI₃
- SEM:
Scanning electron microscopy
- SKPM:
Scanning Kelvin probe microscopy
- TRPL:
Time‑resolved photoluminescence
- XRD:
X‑ray diffraction
Nanomaterials
- Understanding the Skin Effect: Impact on AC Conductors and RF Applications
- Impact of a Trace SiO₂ Addition on the Early‑Stage Sintering of 3Y‑TZP Nanopowders
- Impact of Sulfate Anions on Ultrafine Titania Nucleation During TiCl4 Hydrolysis
- Polyethylene Glycol Enhances NiO Photocathodes for Quantum‑Dot Tandem Solar Cells
- Pd Nanoclusters Accelerate Poly(N‑Vinyl‑2‑Pyrrolidone) Combustion via Atomic Oxygen Generation
- Optimizing CH₃NH₃PbI₃ Morphology for Enhanced Perovskite Solar Cell Performance
- Impact of Morphology and Crystal Structure on Titania Nanotube Thermal Conductivity
- Thermal Annealing Enhances Emission in InP/ZnS Quantum Dot Films: Photoluminescence Insights
- How Temperature Influences the Young’s Modulus of Electrospun Polyurethane Nanofibers
- How Inclusions Influence Steel Performance and Strength



