Impact of a Trace SiO₂ Addition on the Early‑Stage Sintering of 3Y‑TZP Nanopowders
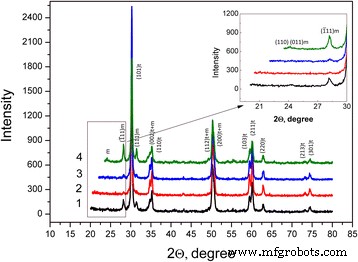
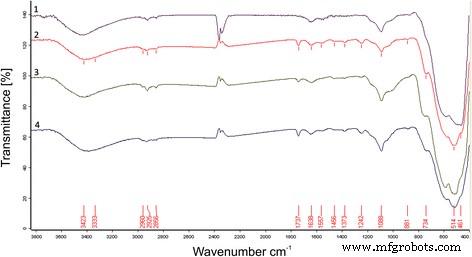
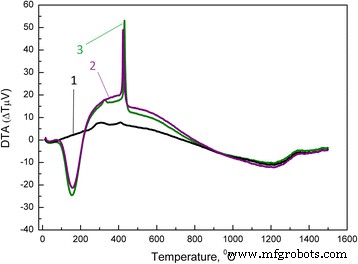
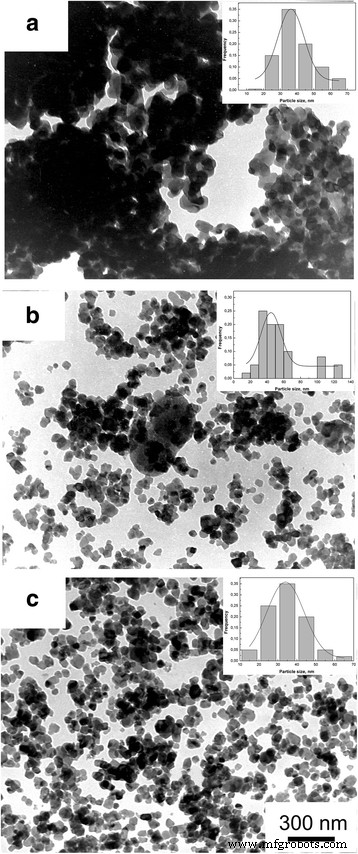
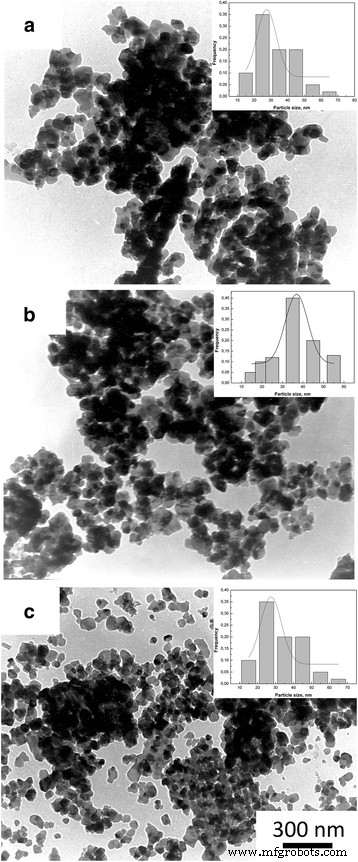
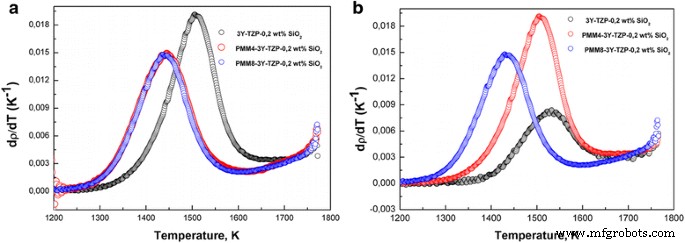
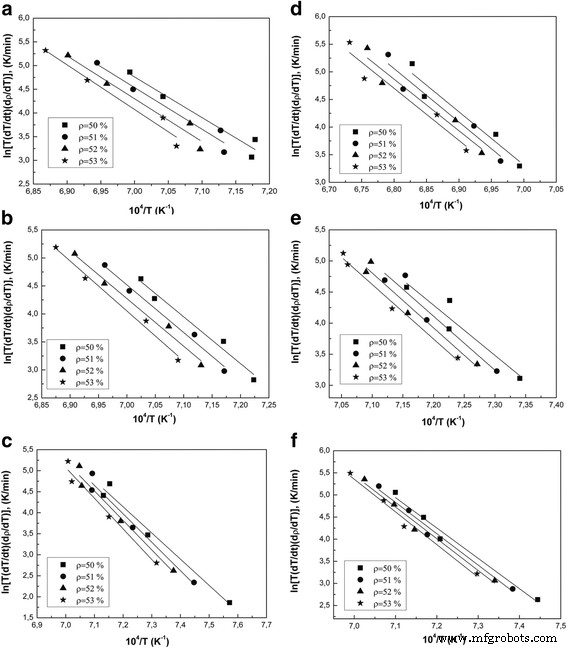
In this study we examined how a trace addition of 0.2 wt % SiO₂ influences the early‑stage sintering of 3 mol % yttria‑stabilized zirconia (3Y‑TZP) nanopowders. SiO₂ was incorporated either via co‑precipitation or by mechanical mixing, and its effect on densification kinetics was compared. For co‑precipitated powders, the presence of SiO₂ shifted the dominant mechanism from volume diffusion (VD) to grain‑boundary diffusion (GBD). In contrast, mechanically mixed powders retained VD as the prevailing mechanism. Overall, both the addition route and mechanical activation accelerated sintering, underscoring the pivotal role of trace SiO₂. Zirconia is celebrated for its exceptional fracture toughness, high strength, hardness, biocompatibility, ionic conductivity, and resistance to radiation and chemicals [1]. These remarkable properties arise from the material’s ability to undergo reversible phase transformations [2]. Zirconia can exist in monoclinic, tetragonal, or cubic phases, and each phase can be stabilized with additives such as Y₂O₃, MgO, or CaO [3]. Yttria‑stabilized tetragonal zirconia (Y‑TZP) has become a cornerstone structural ceramic, finding use in grinding media, optical fiber connectors, and precision parts. When working with zirconia nanopowders, the sintering regime—pressure, temperature, and time—must be finely tuned to achieve dense or porous nanostructures. Nanopowders enable low‑temperature sintering, promoting a homogeneous ceramic structure. The key parameters governing sintering are the particle size and shape, agglomerate size, phase composition, and the “hardness” of agglomerates [2]. Soft agglomerates, weakened by van der Waals forces, can be disintegrated by ultrasonic agitation or dispersants, whereas hard agglomerates—often the result of excessive calcination or inappropriate chemistry—impede the exploitation of nanoscale advantages. High sintering temperatures typically produce bimodal grain sizes and phase segregation. Thus, preventing hard agglomeration and achieving uniform particle morphology remains a primary goal in nanopowder synthesis. Additives such as Al₂O₃, NiO, Cr₂O₃, SiO₂, and GeO₂ can tailor the properties of zirconia ceramics [3–7]. Several studies have examined how these additives influence sintering kinetics. Notably, Matsui reported that SiO₂ accelerates sintering by altering the diffusion mechanism from grain‑boundary to volume diffusion [3,4]. In the present work, we probe how a minuscule, slightly soluble SiO₂ addition affects the initial sintering stage of 3Y‑TZP ceramics. Prior investigations with DIPE‑produced nanopowders revealed contradictory results when compared to Tosoh’s commercial TZ‑3Y powders, a discrepancy we attribute to mechanical activation effects on powder structure and sintering behavior [8]. We employed 3Y‑TZP nanopowders (3 mol % Y₂O₃‑stabilized tetragonal zirconia) synthesized at the Donetsk Institute for Physics and Engineering (DIPE) using a chloride‑based co‑precipitation method. The process, detailed in our previous publication [9], incorporated 0.2 wt % SiO₂ in two distinct manners: All powders were calcined at 1000 °C for 2 h. Subsequently, the mechanically milled samples were further milled in a planetary mill. Compacts were pressed at 300 MPa and sintered to 1500 °C using a NETZSCH DIL 402 PC dilatometer at heating rates of 2.5, 5, 10, and 20 °C min⁻¹. Shrinkage data were calibrated against Al₂O₃, with thermal expansion corrected by the cooling curve method [7,9]. Final densities were measured via the Archimedes method. Powder characterization included X‑ray diffraction (XRD) on a Dron‑3 diffractometer (Cu‑Kα radiation), with peak fitting via Powder Cell 2.4. Crystallite sizes (d_XRD) were extracted using the Debye–Scherrer equation [10]. Specific surface area (S_BET) and crystallite sizes (d_BET) were measured by BET on a “SORBI‑4” device. TEM (Jem 200A, JEOL) provided particle size histograms from 200–250 images. FTIR (Tensor 27, Bruker) and TG‑DTA (Linses 1600, Germany) examined functional groups and thermal behavior. Agglomeration factors were calculated per [2,11,12]. Energy‑dispersive X‑ray spectroscopy (EDS) on a JSM 6490 LV JEOL verified composition and mapping. SEM (JSM 6490LV) examined microstructure on polished and fractured surfaces. To analyze the initial sintering stage, we employed the constant‑rate‑of‑heating (CRH) technique [13–15], applicable up to 4 % relative shrinkage. Activation energies (Q) were derived using the Wang‑Raj equation [13]: \[\mathrm{T}\cdot\mathrm{c}\frac{\mathrm{d}\uprho}{\mathrm{d}\mathrm{T}}=\frac{1}{\mathrm{F}'(\uprho)}\cdot \frac{\mathrm{K}\gamma \Omega \mathrm{D}}{\mathrm{kTa}^{\mathrm{p}}}\cdot \exp\left(-\frac{\mathrm{Q}}{\mathrm{RT}}\right)\] From the Arrhenius plot of ln[T(dT/dt)(dρ/dT)] vs. 1/T, the slope S₁ yields Q = –RS₁. The sintering mechanism parameter n was extracted via Yang and Cutler’s formulation [14]: \[\frac{\mathrm{d}\left(\Delta L/L_0\right)}{\mathrm{d}\mathrm{T}}=\left(\frac{\mathrm{K}\gamma \Omega \mathrm{D}_0\mathrm{R}}{\mathrm{ka}^{\mathrm{p}}\mathrm{cQ}}\right)\cdot \left(\frac{\mathrm{nQ}}{\mathrm{RT}^{2-\mathrm{n}}}\right)\cdot \exp\left(-\frac{\mathrm{nQ}}{\mathrm{RT}}\right)\] Plotting ln[T^{2–n} d(ΔL/L₀)/dT] vs. 1/T gives nQ = –RS₂. An n value of 1 indicates viscous flow, ½ volume diffusion, and ⅓ grain‑boundary diffusion. Table 1 summarizes the powder characteristics. XRD spectra (Fig. 1) reveal that both silica addition and mechanical milling have minimal impact on crystallite size and phase composition. Mechanical activation for 8 h slightly increases the monoclinic fraction. Importantly, silica is incorporated as a solid solution when co‑precipitated; it does not appear as a separate phase. Lattice parameters decrease marginally for co‑precipitated 3Y‑TZP‑0.2 wt % SiO₂, consistent with the smaller ionic radius of Si⁴⁺ (0.040 nm) compared to Zr⁴⁺ (0.072 nm). For mechanically milled powders, lattice contraction is attributed to the milling process (Table 2). The XRD patterns of nanopowders with and without silica additive: 1–PMM8‑3Y‑TZP‑0.2 wt % SiO₂; 2–3Y‑TZP‑0.2 wt % SiO₂; 3–3Y‑TZP; 4–PM8‑3Y‑TZP. FTIR spectra (Fig. 2) display bands between 3700–3200 cm⁻¹ and 1700–1300 cm⁻¹, corresponding to H₂O and hydroxyl vibrations on nanoparticle surfaces. Bands from 1200–1000 cm⁻¹ are attributed to Zr=O (OH) and Si–OH groups, while features below 1000 cm⁻¹ arise from Zr–O–Zr and O–Zr–O bonds. In the Zr–O stretching region, peaks at 734, 590, 514, and 461 cm⁻¹ indicate coexistence of tetragonal and monoclinic phases. The appearance of the 734 cm⁻¹ band in modified systems reflects an increased monoclinic content and surface defect density, raising the surface energy hierarchy (E₀, surf^t < E₀, surf^t,def < E₀, surf^m) [17–18]. The FTIR spectra of nanopowders: 1–3Y‑TZP; 2–PM8‑3Y‑TZP; 3–PMM8‑3Y‑TZP‑0.2 wt % SiO₂; 4–3Y‑TZP‑0.2 wt % SiO₂. Thermal analysis (Fig. 3) shows an endothermic peak near 157 °C (evaporation of physical water) and exothermic peaks around 423–430 °C (crystallization) for both silica‑containing and silica‑free powders. Silica has negligible influence on crystallization dynamics. DTA curves of samples: 1–PMM8‑3Y‑TZP‑0.2 wt % SiO₂ (nanopowders calcined at 1000 °C for 2 h); 2–3Y‑TZP‑0.2 wt % SiO₂ (hydroxide); 3–3Y‑TZP (hydroxide). TEM images (Fig. 4) reveal that 3Y‑TZP (a) exhibits high, yet “soft” agglomeration that can be disrupted by 4‑h or 8‑h milling. Figures 5 and 6 illustrate particle size distributions and agglomeration factors. Mechanical milling increases S_BET with time, achieving a maximum for PMM8‑3Y‑TZP + 0.2 wt % SiO₂. Milling for 4 h reduces agglomeration, but silica distribution remains uneven. Eight hours of milling provides uniform silica dispersion, as shown in Fig. 6. TEM images and histograms of particle size distribution of (a) 3Y‑TZP, (b) PM4‑3Y‑TZP, (c) PM8‑3Y‑TZP nanopowders. TEM images and histograms of particle size distribution of (a) 3Y‑TZP‑0.2 wt % SiO₂, (b) PMM4‑3Y‑TZP‑0.2 wt % SiO₂, (c) PMM8‑3Y‑TZP‑0.2 wt % SiO₂ nanopowders. The SEM images and EDX mapping analysis of sintered to 1500 °C samples (a) 3Y‑TZP‑0.2 wt % SiO₂, and (b) PMM8‑3Y‑TZP‑0.2 wt % SiO₂. Densification rates (dρ/dT) versus temperature (Fig. 7) show that unmilled 3Y‑TZP reaches peak densification at a lower temperature than milled powders, which exhibit shifted curves toward higher temperatures. Arrhenius plots (Fig. 8) and Table 3 confirm that unmilled 3Y‑TZP sinters predominantly via volume diffusion, accounting for its faster densification. Mechanical activation (8 h milling) transitions the mechanism from volume to grain‑boundary diffusion, while silica addition shifts the mechanism from GBD (co‑precipitated) to VD (mixed) in the initial sintering stage. The data demonstrate that a mere 0.2 wt % SiO₂, when introduced by co‑precipitation, can profoundly alter sintering kinetics and mechanism. Temperature dependence of densification rates of (a) 3Y‑TZP, PM4‑3Y‑TZP, PM8‑3Y‑TZP and (b) 3Y‑TZP‑0.2 wt % SiO₂, PMM4‑3Y‑TZP‑0.2 wt % SiO₂, PMM8‑3Y‑TZP‑0.2 wt % SiO₂ nanopowders. Arrhenius-type plots of (a) 3Y‑TZP, (b) PM4‑3Y‑TZP, (c) PM8‑3Y‑TZP, (d) 3Y‑TZP‑0.2 wt % SiO₂, (e) PMM4‑3Y‑TZP‑0.2 wt % SiO₂, and (f) PMM8‑3Y‑TZP‑0.2 wt % SiO₂ nanopowders. Our investigation demonstrates that a trace amount of SiO₂, its mode of incorporation, and mechanical activation decisively influence the early‑stage sintering of 3Y‑TZP nanopowders: 3 mol % yttria‑stabilized tetragonal zirconia polycrystal Constant rate of heating Donetsk Institute for Physics and Engineering Grain‑boundary diffusion National Academy of Sciences Powder milling for 4 and 8 h Powder mixing and milling for 4 and 8 h Specific surface area measured by the Brunauer‑Emmett‑Teller method Transmission electron microscopy Volume diffusion Weight percent X‑ray diffractionAbstract
Background
Methods
Results and Discussion

Conclusions
Abbreviations
Nanomaterials
- Understanding the Skin Effect: Impact on AC Conductors and RF Applications
- Impact of Sulfate Anions on Ultrafine Titania Nucleation During TiCl4 Hydrolysis
- Polyethylene Glycol Enhances NiO Photocathodes for Quantum‑Dot Tandem Solar Cells
- Pd Nanoclusters Accelerate Poly(N‑Vinyl‑2‑Pyrrolidone) Combustion via Atomic Oxygen Generation
- Impact of Morphology and Crystal Structure on Titania Nanotube Thermal Conductivity
- How Temperature Influences the Young’s Modulus of Electrospun Polyurethane Nanofibers
- Enhancing Thermal Diffusivity of Al₂O₃ Nanofluids Through Modulated Laser‑Induced Photothermal Fragmentation
- Impact of Sintering Temperature on the Electrochemical Performance of Ce0.8Sm0.05Ca0.15O2-δ/La0.6Sr0.4Co0.2Fe0.8O3-δ Heterostructure Electrolytes
- Impact of Decomposed PbI₂ on Scattering Mechanisms in CH₃NH₃PbI₃ Perovskite Films
- How Inclusions Influence Steel Performance and Strength



