Icariin‑Loaded mPEG Nanoparticles: Enhanced Protection Against LPS‑Induced Cardiotoxicity in H9c2 Cells
Abstract
Icariin (ICA), a flavonol glycoside from Epimedium, exhibits cardioprotective, antioxidant, and anti‑inflammatory properties but suffers from poor water solubility, rapid clearance, and low oral bioavailability. We synthesized a hydrophilic polyethylene glycol monomethyl ether (mPEG) conjugate (mPEG‑ICA) via succinic anhydride‑mediated esterification, and subsequently encapsulated free ICA into the polymer by dialysis to form ICA‑loaded mPEG‑ICA nanoparticles (NPs). Fourier transform infrared spectroscopy (FT‑IR) and proton nuclear magnetic resonance (^1H‑NMR) confirmed successful conjugation. Dynamic light scattering (DLS) revealed an average hydrodynamic diameter of 220 ± 13.7 nm with a ζ‑potential of +2.30 ± 1.33 mV, while transmission electron microscopy (TEM) showed uniform, spherical morphology. In phosphate‑buffered saline (PBS) at pH 7.4, the nanoparticles released 52.8 ± 1.7 % ICA over 72 h; at pH 6.8, release accelerated to 75.7 ± 0.2 % within 48 h. In vitro, the NPs preserved H9c2 cell viability, reduced lactate dehydrogenase (LDH) leakage, and inhibited apoptosis induced by lipopolysaccharide (LPS). Moreover, mRNA levels of the pro‑inflammatory cytokines TNF‑α, IL‑1β, and IL‑6 were markedly suppressed. These results demonstrate that ICA‑loaded mPEG‑ICA NPs offer a promising nanomedicine platform for mitigating LPS‑mediated myocardial injury.
Introduction
Cardiac remodeling following myocardial injury is driven largely by sustained inflammatory signaling, particularly through TNF‑α, IL‑1β, IL‑6, and IL‑18. These cytokines trigger oxidative stress, fibroblast activation, and cardiomyocyte apoptosis, ultimately compromising cardiac function. Icariin (C_33H_40O_15) has been extensively reported to exert immunomodulatory, antioxidant, and anti‑apoptotic effects in preclinical models, yet its therapeutic potential is limited by hydrophobicity, rapid systemic clearance, and poor oral absorption. Nanocarrier systems—such as micelles, liposomes, and polymeric nanoparticles—have emerged as effective strategies to enhance drug solubility, extend circulation time, and achieve targeted delivery to inflamed cardiac tissue, leveraging the enhanced permeability and retention (EPR) effect.
Polyethylene glycol monomethyl ether (mPEG) offers superior hydrophilicity, biocompatibility, and minimal immunogenicity compared with linear PEG. By covalently attaching ICA to mPEG‑COOH through an ester linkage, we create a hydrophilic–hydrophobic amphiphile capable of self‑assembly into nanoparticles. The ester bond is acid‑labile, allowing pH‑responsive release of ICA at the mildly acidic microenvironment (pH ≈ 6.8) characteristic of inflamed myocardium. This study details the synthesis of mPEG‑ICA, encapsulation of ICA into nanoparticles, physicochemical characterization, in‑vitro release profiling, and evaluation of cardioprotective efficacy against LPS‑induced injury in H9c2 cells.
Materials and methods
Experimental instruments
Key analytical instruments included a JA302 electronic balance, RE52CS‑1 rotary evaporator, 78HW‑1 magnetic agitator, 101‑OA blast dryer, NEXUS670 FT‑IR spectrometer, UV–Vis spectrophotometer, TEM Glacios, JP‑010S ultrasonic disperser, real‑time qPCR system, enzyme‑linked fluorescence analyzer, Olympus inverted microscope, Leica fluorescence microscope, and a Thermo CO_2 incubator.
Experimental reagents
Reagents of analytical grade were sourced from commercial suppliers: Icariin (95 %, 1 g, DL070208), LPS (55 mg, Sigma, Z06J9Y52452), mPEG‑Me (250 g, Sigma, MKBT7172), dichloromethane (500 mL, Sinopharm), 4‑dimethylaminopyridine (100 g, Aladdin), N‑hydroxysuccinimide (100 g, Ruiyong), FastKing cDNA synthesis kit (Tiangen), Biyuntian apoptosis kit, SYBR Green qPCR kit (QIAGEN), among others.
Synthesis of mPEG‑COOH
mPEG (5 g), succinic anhydride (0.40 g), and DMAP (0.50 g) were reacted in 50 mL dichloromethane at 60 °C for 2 h under reflux. The mixture was evaporated, and the resulting white solid was purified by dialysis (2 kDa MWCO) against water for 48 h. Freeze‑dried product yielded mPEG‑COOH.
Synthesis of mPEG‑ICA polymer
mPEG‑COOH (0.37 g) was activated with NHS (0.024 g) and DMAP (0.026 g) in 10 mL dry DMSO, then reacted with 100 mg ICA (dissolved in 5 mL dry DMSO) under nitrogen at room temperature for 48 h. After extensive dialysis against water and freeze‑drying, mPEG‑ICA polymer was obtained.
Preparation of ICA‑loaded mPEG‑ICA nanoparticles (NPs)
mPEG‑ICA (5 mg) was dissolved in 2 mL DMSO and mixed with 5 mg ICA in DMSO, followed by the addition of 5 mL distilled water. After 15 min stirring at room temperature, the mixture was dialyzed (3500 Da MWCO) for 24 h with frequent water changes, then filtered to yield ICA‑loaded mPEG‑ICA NPs.
Fourier transform infrared spectroscopy
Samples were mixed with KBr, pressed into pellets, and scanned from 4000 to 4400 cm^−1 to confirm functional group transformations.
Nuclear magnetic resonance hydrogen spectrum
Samples were dissolved in DMSO‑d_6 and analyzed on a 500 MHz NMR spectrometer to confirm chemical shifts consistent with esterification.
UV–Vis spectrum
A calibration curve of ICA at 270 nm was generated; concentrations of polymer and nanoparticles were quantified using this curve.
Characterization of ICA‑loaded mPEG‑ICA NPs
Dynamic light scattering detection
Particle size distribution and ζ‑potential were measured with a Zetasizer 3000hs after lyophilization and redispersion in water; each sample was analyzed in triplicate.
Observation on the morphology by TEM
1.0 mg/mL NP suspension was applied to carbon‑coated copper grids, stained with 2 % phosphotungstic acid, and imaged at 80 kV.
Measurement of the stability
NPs were freeze‑dried with 5 % mannitol, re‑dispersed in water, and size/PDI monitored to assess storage stability.
Determination of drug loading and entrapment efficiency
Drug loading (EE%) and encapsulation efficiency (LC%) were calculated from UV‑Vis absorbance using the formulas: EE = drug mass in NPs / total mass of NPs × 100 %; LC = drug mass in NPs / initial drug mass × 100 %.
Drug release in vitro
NPs were placed in 25 mL PBS (pH 7.4 or 6.8) at 37 °C with 100 rpm agitation. Samples (2 mL) were withdrawn at defined intervals, replaced with fresh medium, and analyzed by UV–Vis at 270 nm. Cumulative release (%) was calculated using the standard equation.
Cell testing experiment
H9c2 rat cardiomyoblasts were cultured in DMEM + 10 % FBS at 37 °C/5 % CO_2. Cells were plated in 96‑well plates (10^4 cells/well) for viability, LDH, apoptosis, and qPCR assays.
Cell treatment with LPS or ICA‑nanoparticles
Cells were divided into five groups: (1) control (no treatment), (2) LPS (10 µg/mL) for 24 h, (3) ICA (20 µM) + LPS, (4) mPEG‑ICA polymer (20 µM) + LPS, (5) ICA‑loaded mPEG‑ICA NPs (20 µM) + LPS.
MTT
After 24 h treatment, 0.5 mg/mL MTT was added for 4 h. Formazan was dissolved in DMSO and absorbance measured at 570 nm.
Lactate dehydrogenase (LDH) release
LDH activity in culture supernatants was quantified using a commercial kit; absorbance read at 490 nm.
Hoechst 33342 staining
Nuclear morphology was examined after 15 min staining with Hoechst 33342 (1 µg/mL) and imaged under a fluorescence microscope. Apoptosis index = apoptotic nuclei/total nuclei × 100 %.
TUNEL assay
DNA fragmentation was detected using a TUNEL kit; apoptotic nuclei were counted as green fluorescence relative to total DAPI‑stained nuclei.
Flow cytometry to detect apoptosis
Annexin V‑FITC/PI double staining was performed; early and late apoptotic populations were quantified by flow cytometry.
Reverse transcription quantitative polymerase chain reaction (RT–qPCR)
RNA was extracted with TRIzol, reverse‑transcribed, and amplified on a Bio‑Rad CFX96 system using SYBR Green. Gene expression of TNF‑α, IL‑1β, IL‑6, and actin (housekeeping) was normalized to actin. Primer sequences are listed in the original manuscript.
Results and discussion
FTIR and ^1H NMR spectra
FT‑IR spectra displayed characteristic ester carbonyl absorptions at 1700 and 1740 cm^−1, confirming successful conjugation. ^1H‑NMR revealed phenolic hydroxyl signals at 12.6 ppm, aromatic protons at 7.5 ppm, and methylene protons of the PEG chain at 3.5 ppm, confirming the presence of both ICA and mPEG moieties.
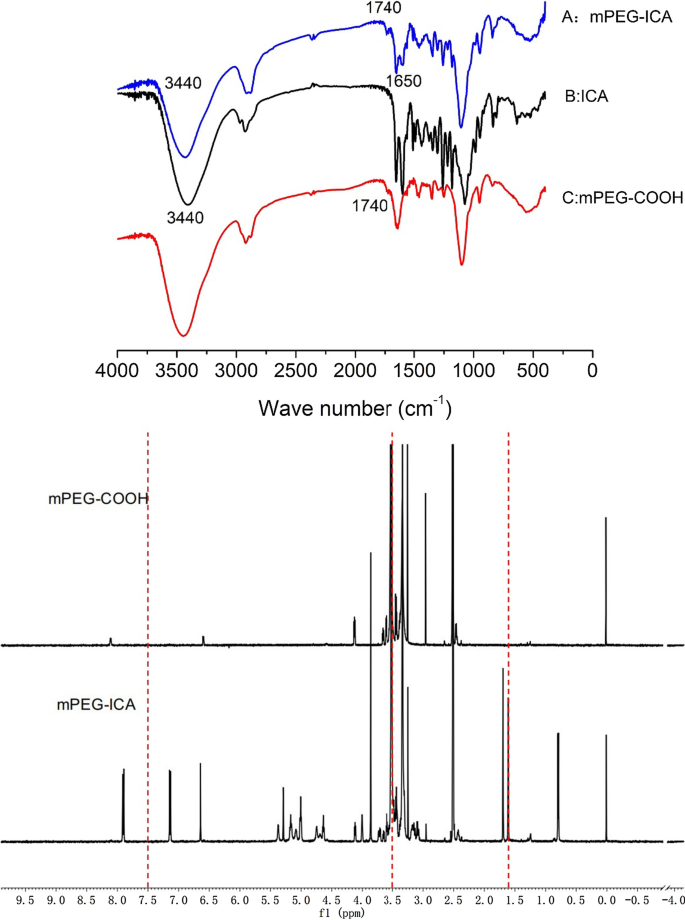
FTIR spectra of mPEG‑ICA (A), ICA (B), and mPEG‑COOH (C). ^1H‑NMR spectra for mPEG‑COOH and mPEG‑ICA.
Particle size, zeta potential and TEM
The core polymer NPs averaged 145 ± 15.2 nm (PDI = 0.277), whereas ICA‑loaded NPs expanded to 220 ± 13.7 nm (PDI = 0.119). Zeta potentials shifted from +0.44 ± 0.26 mV to +2.30 ± 1.33 mV upon loading, indicating improved colloidal stability. TEM images confirmed a spherical, uniform morphology (Fig. 2).
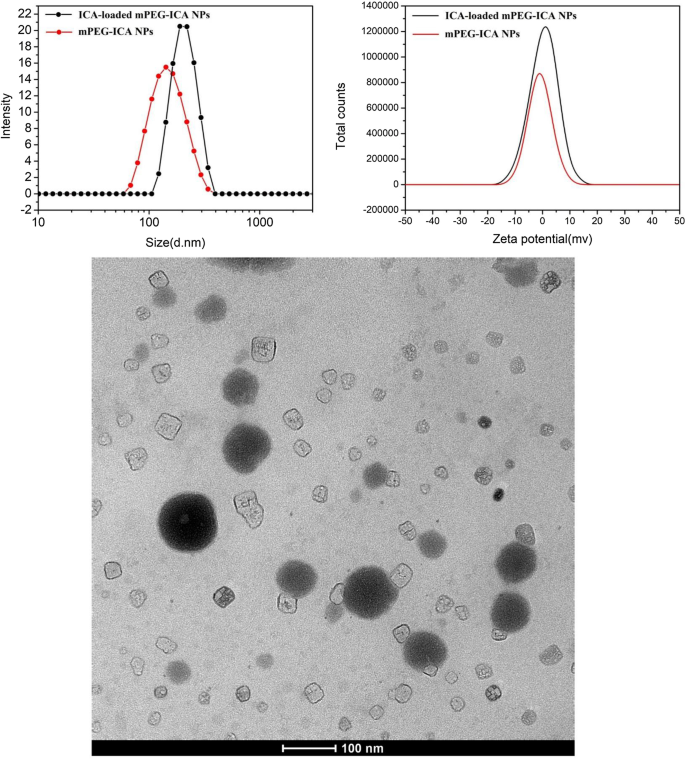
Particle size, zeta potential and TEM of ICA‑loaded mPEG‑ICA NPs.
Drug loading and encapsulation efficiencies
UV‑Vis analysis yielded drug loading of 21.9 ± 0.013 % and encapsulation efficiency of 54.9 ± 0.032 % for the ICA‑loaded NPs, outperforming polymer‑only NPs (loading = 16.5 %; EE = 41.3 %). These values are consistent with the desired therapeutic payload.
Stability determination
Post freeze‑drying with 5 % mannitol, re‑dispersed NPs exhibited a modest size increase to 255 nm (PDI = 0.326) yet maintained acceptable uniformity, confirming storage stability (Fig. 3).
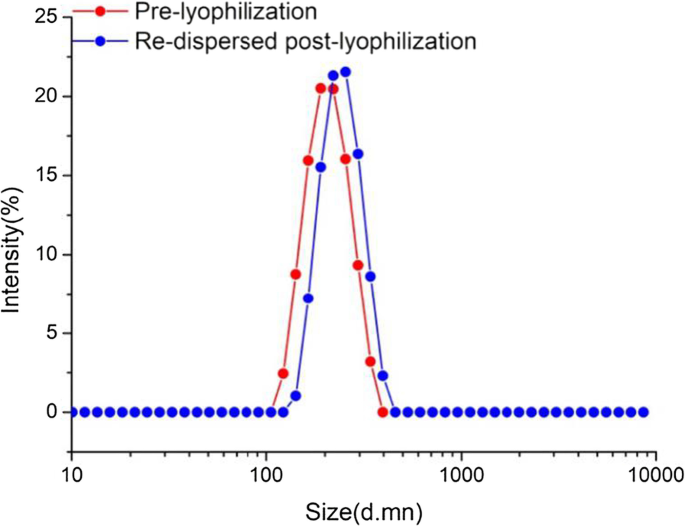
Stability of ICA‑loaded mPEG‑ICA NPs.
mPEG‑ICA‑ICA drug release in vitro
In pH 7.4 PBS, free ICA released 77.2 % within 12 h, whereas mPEG‑ICA NPs released 44.1 % over 72 h. ICA‑loaded NPs released 52.8 % over 72 h at neutral pH and 75.7 % within 48 h at pH 6.8, reflecting pH‑responsive accelerated release (Fig. 4).
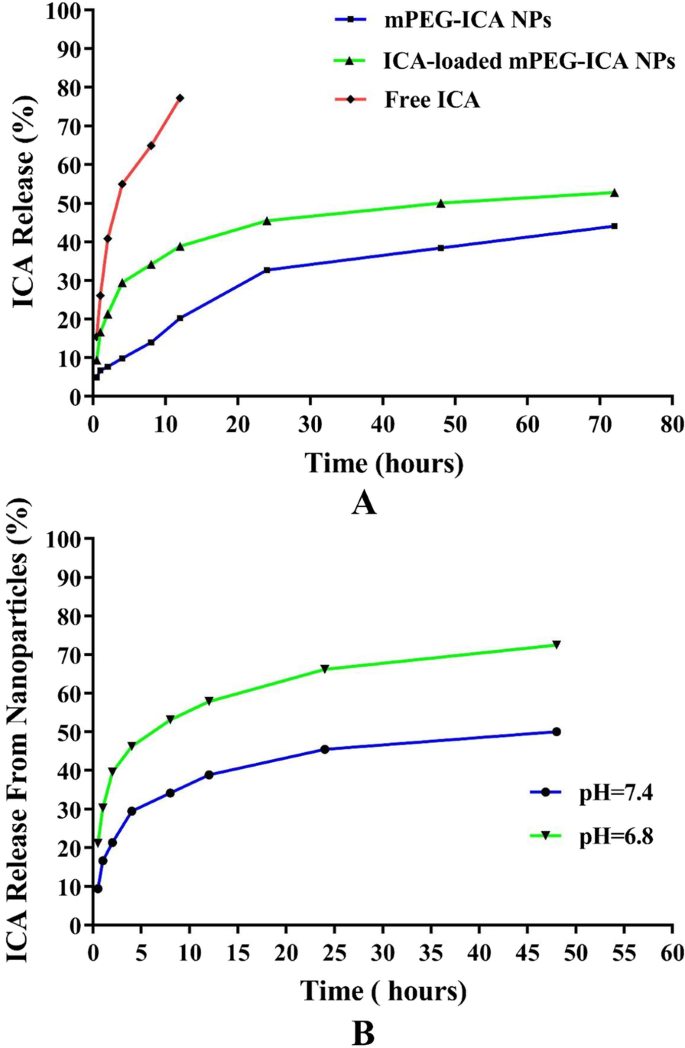
A ICA release from ICA‑loaded mPEG‑ICA NPs in PBS at pH 7.4. B Release at pH 7.4 vs pH 6.8.
H9C2 cell viability and lactate dehydrogenase release
MTT assays revealed no cytotoxicity of ICA, mPEG‑ICA, or ICA‑loaded NPs at 20 µM. Following LPS exposure, viability dropped to 50.0 % (control = 100 %). Treatment with ICA, mPEG‑ICA, and ICA‑loaded NPs restored viability to 55.9 %, 61.0 %, and 65.4 % respectively (p < 0.05) (Fig. 5, 6).
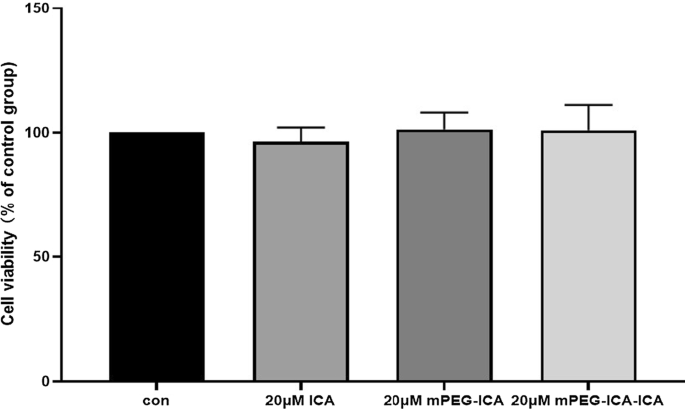
Evaluation of the cytotoxicity of ICA nanoparticles on H9c2 cells.
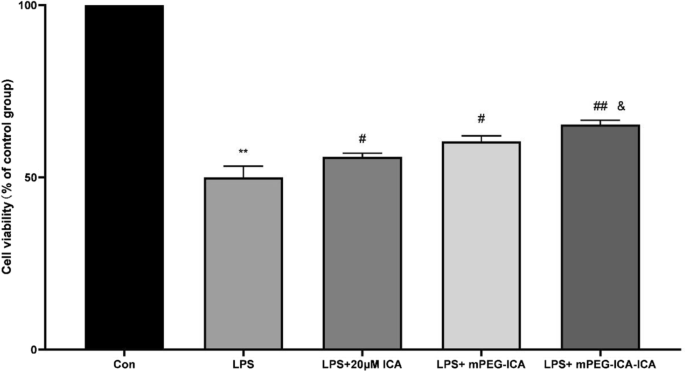
Effect of ICA nanoparticles on LPS‑induced H9c2 cell injury.
LDH assays showed a significant rise in enzymatic leakage after LPS treatment; ICA, mPEG‑ICA, and ICA‑loaded NPs markedly reduced LDH release, with the most pronounced effect observed for ICA‑loaded NPs (Fig. 7).
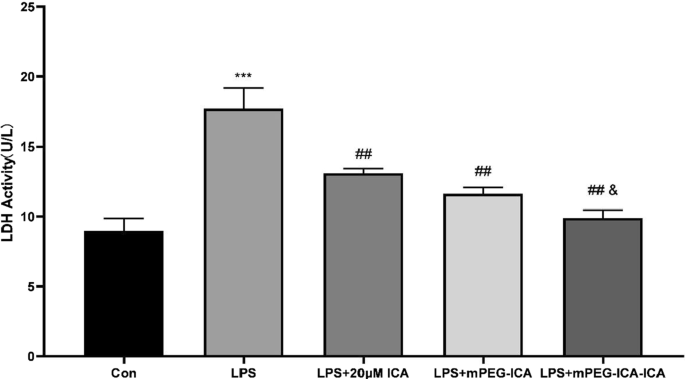
Effect of ICA nanoparticles on the LDH level induced by LPS in H9c2 cells.
Cell apoptosis induced by LPS
Hoechst 33342 staining revealed chromatin condensation and fragmentation in LPS‑treated cells; ICA‑loaded NPs significantly attenuated apoptotic morphology (Fig. 8). TUNEL assay quantified apoptosis rates: LPS induced 47.6 % apoptosis, which was reduced to 35.5 % (ICA), 26.5 % (mPEG‑ICA), and 17.9 % (ICA‑loaded NPs). Flow cytometry corroborated these findings, showing total apoptosis of 35.9 % in LPS versus 16.7 % (ICA‑loaded NPs) and 19.2 % (mPEG‑ICA) (Fig. 9‑10).
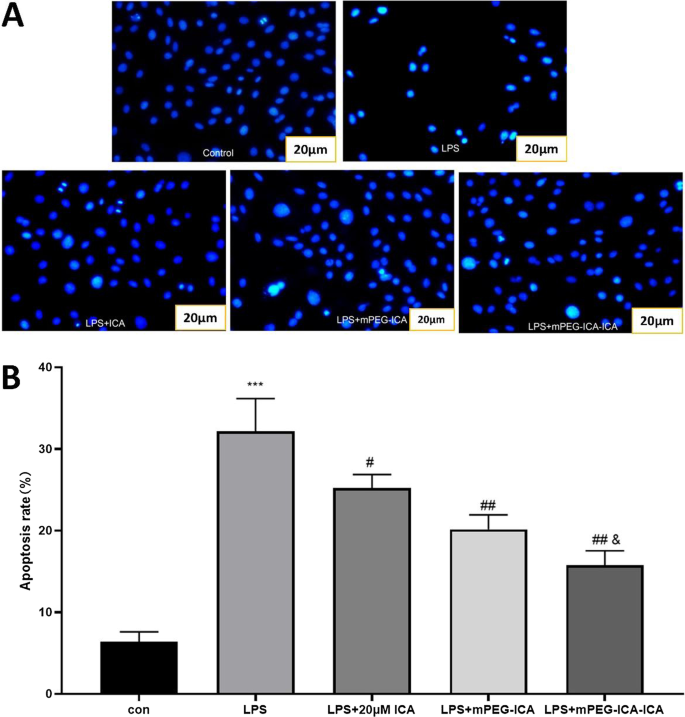
Detection of apoptosis by Hoechst 33342 staining.
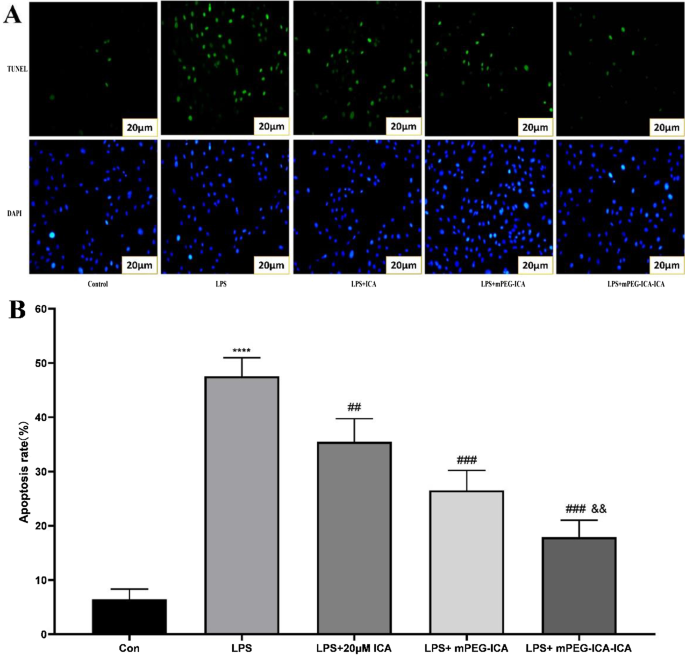
Detection of apoptosis by TUNEL.
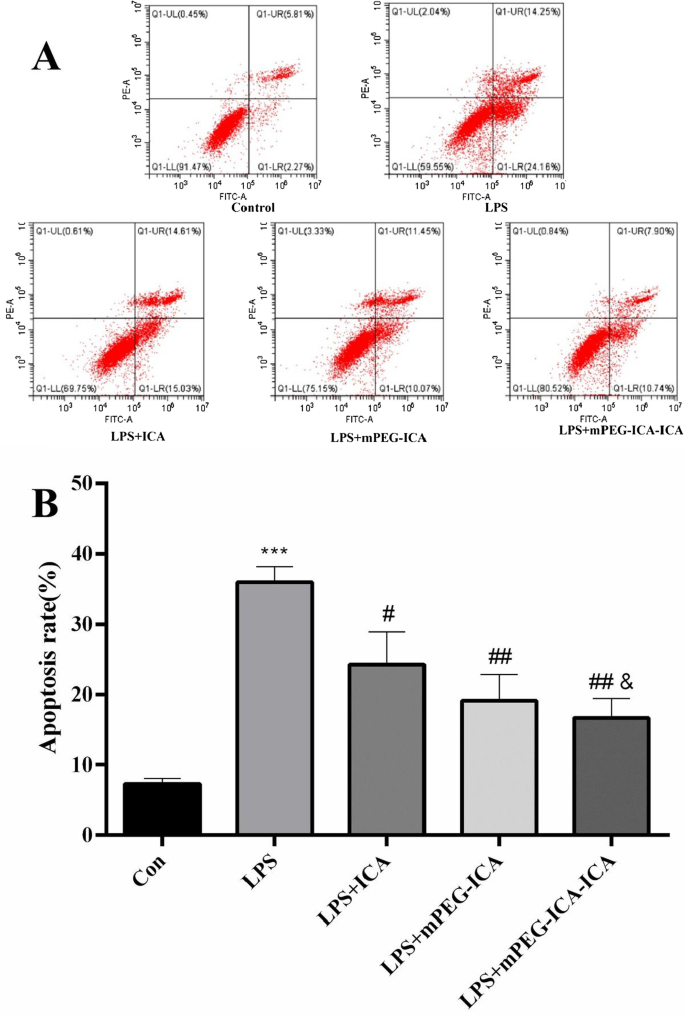
Apoptotic rate detected by flow cytometry.
Inflammatory cytokine mRNA in LPS‑induced H9c2 cells
RT‑qPCR demonstrated that LPS markedly elevated TNF‑α, IL‑1β, and IL‑6 transcripts. Treatment with ICA, mPEG‑ICA, and ICA‑loaded NPs suppressed these cytokines, with ICA‑loaded NPs achieving the most substantial down‑regulation (Fig. 11).
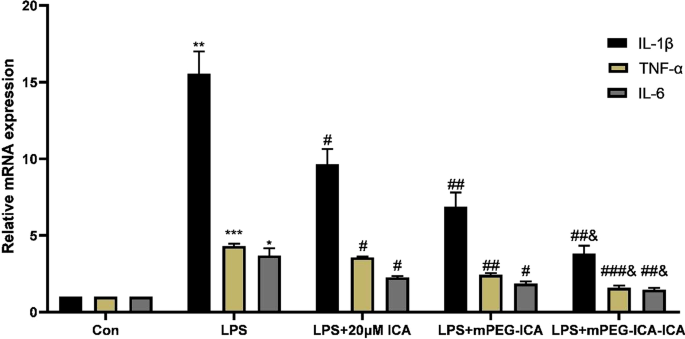
Effects of ICA‑loaded mPEG‑ICA NPs on IL‑1β, TNF‑α, and IL‑6 mRNA expression.
These findings confirm that ICA‑loaded mPEG‑ICA NPs provide enhanced cardioprotection by delivering ICA directly to inflamed myocardium, reducing oxidative stress, apoptosis, and inflammatory cytokine production.
Conclusion
We have successfully fabricated ICA‑loaded mPEG‑ICA nanoparticles via a hybrid chemical‑physical approach that markedly improves ICA solubility, loading capacity, and sustained, pH‑responsive release. In LPS‑challenged H9c2 cells, these NPs restored cell viability, attenuated LDH leakage, inhibited apoptosis, and suppressed pro‑inflammatory cytokine expression more effectively than free ICA or polymer alone. The data support the potential of ICA‑loaded mPEG‑ICA NPs as a targeted nanomedicine for treating inflammatory cardiac injury.
Nanomaterials
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Au@TiO₂ Yolk–Shell Nanostructures: Tailored Synthesis and Their Superior Visible‑Light Photocatalytic Degradation and SERS Detection of Methylene Blue
- Green Synthesis of Metal‑Oxide Nanoparticles with Gum Karaya and Their Ecotoxicological Impact on *Chlamydomonas reinhardtii*
- Sulfuric‑Acid‑Assisted Synthesis of Bright Red Carbonized Polymer Dots for Two‑Photon Bio‑Imaging
- Controlling Silver Nanoparticle Morphology via CTAB‑Capped Seeds and Aging Time
- Comprehensive Review of Luminescent Silica Nanoparticles: Classification, Synthesis, and Practical Applications
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
- Developing a Precise Lysosomal pH Sensor Using Self-Decomposing SiO2 Nanoparticles
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy
- Eco‑Friendly, Cost‑Effective Synthesis of Tin Oxide Nanoparticles: A Comprehensive Review of Methods, Formation Mechanisms, and Applications



