Graphene‑Assisted Thermal Interface Materials with Optimized Interface Contact Between Matrix and Fillers
We engineered thermal interface materials (TIMs) by combining reduced graphene oxide (RGO) and three‑dimensional graphene networks (3DGNs). The 3DGNs create a high‑conductivity phonon network, while RGO bridges the graphene and epoxy resin (ER) matrix, especially when its surface is enriched with carboxyl groups. Optimising filler loading and morphology yielded a thermal conductivity of 6.7 W m⁻¹ K⁻¹—an increase of 3 250 % over pristine ER—while the RGO(OOH) composites also exhibited superior mechanical performance. With the relentless rise of power density in modern electronics, efficient heat removal via TIMs has become critical [1–4]. Traditional fillers such as SiC, Al₂O₃, and BN require mass fractions above 50 % to meet thermal demands, which compromises mechanical integrity. Graphene, with a monolayer thermal conductivity of ~5 000 W m⁻¹ K⁻¹ [5], offers a compelling alternative: a 20 wt % RGO loading can raise TIM conductivity to ~4 W m⁻¹ K⁻¹ and achieve ~2 000 % enhancement while maintaining acceptable mechanics [6,7]. Hybridisation with carbon nanotubes further boosts performance [8,9].
However, the high defect density of RGO—introduced during oxidation–reduction—restricts phonon transport [10]. Studies of vacancy‑induced phonon scattering in bulk and 2D materials reveal that defects diminish thermal pathways [11]. High‑quality 3DGNs fabricated by CVD exhibit minimal D‑band Raman intensity, yet their interface contact with ER remains sub‑optimal because the basal planes lack an efficient chemical link [12]. A modest defect density in 3DGNs can improve interfacial bonding, but controlling this parameter is challenging [13]. Prior work using RGO and 3DGNs together has yet to realise the expected synergy [14].
In this study, we synthesise RGO with tailored surface functional groups (carboxyl, hydroxyl, epoxy) and combine it with 3DGNs in an ER matrix. We systematically investigate how the functional group chemistry and RGO morphology influence phonon transport across the filler–matrix interface and, consequently, the overall thermal and mechanical performance of the TIMs. Figure 1 presents SEM micrographs of pristine RGO, 3DGNs, and the resulting composites. The composites exhibit smooth surfaces, with digital photographs of ER, RGO, and RGO‑3DGNs‑ER provided in the insets. Unlike the small wrinkles in RGO, 3DGNs display larger surface undulations due to thermal expansion mismatches with the nickel substrate. The raw ER shows pores and cracks indicative of low conductivity, which are largely eliminated after incorporating graphene fillers. RGO‑ER surfaces display partial RGO patches, while 3DGNs‑ER surfaces reveal concave–convex features from internal 3DGNs. These characteristics are also present in the co‑modified RGO‑3DGNs‑ER composite, as confirmed by cross‑sectional SEM (Figure 1h inset).
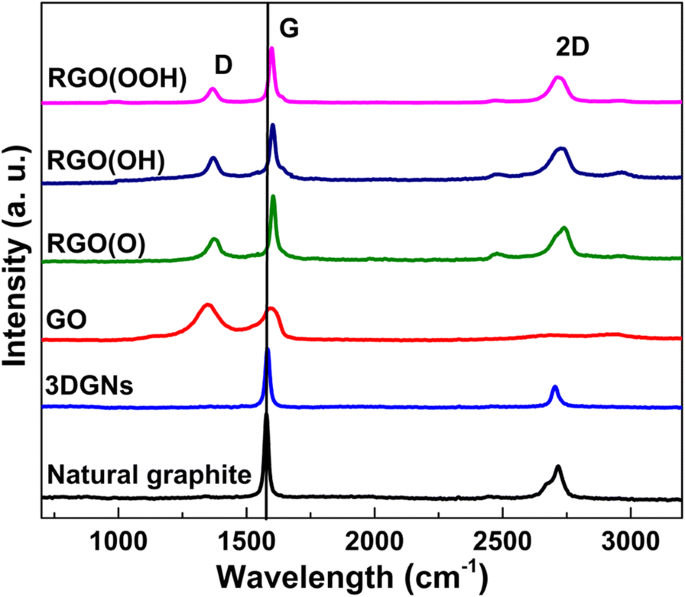
Figure 2 shows Raman spectra of natural graphite, various RGO derivatives, and 3DGNs. The 3DGNs exhibit negligible D‑band intensity, confirming high crystallinity, whereas GO shows a pronounced D‑band due to oxidation‑induced defects. Reduction restores the 2D‑band and reduces the D‑band, enabling calculation of defect density via the I_D/I_G ratio (Table S1). The G‑band shift from 1580 cm⁻¹ (graphite/3DGNs) to 1600 cm⁻¹ (RGO) confirms higher defect levels in RGO. XRD and XPS analyses (Figures S1–S2, Table S2) reveal the presence and proportion of carboxyl, hydroxyl, and epoxy groups; selective retention of these groups is achieved by tuning oxidation–reduction protocols [10,12,19].
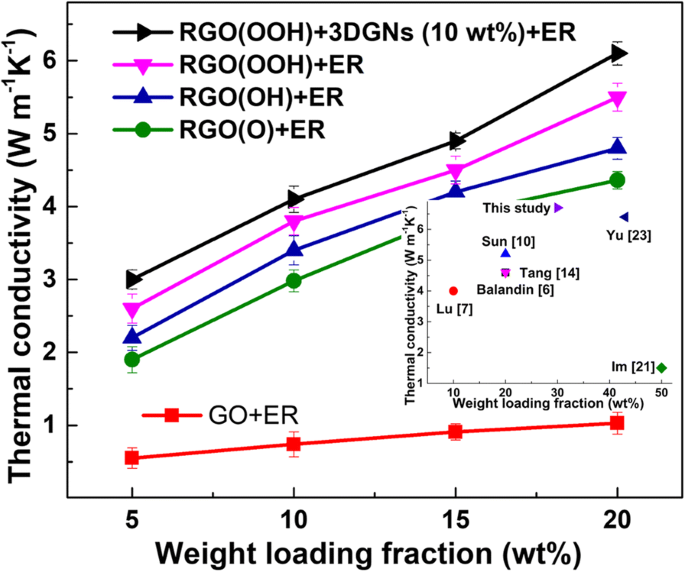
Thermal conductivity measurements (Figure 3) demonstrate that RGO(OOH)‑assisted composites outperform RGO(OH) and RGO(O) variants, with a 12 % improvement at 20 wt % loading. Adding 3DGNs further raises conductivity to 6.1 W m⁻¹ K⁻¹. The superior performance of RGO(OOH) aligns with the lowest interface thermal boundary resistance (δ) predicted by Balandin’s model (Eq. 1), where δ is strongly dependent on the surface functional groups (Table 1). Carboxyl groups react with epoxy groups during solidification (110 °C), forming covalent bonds that reduce phonon mismatch and enhance heat transfer [14,25].
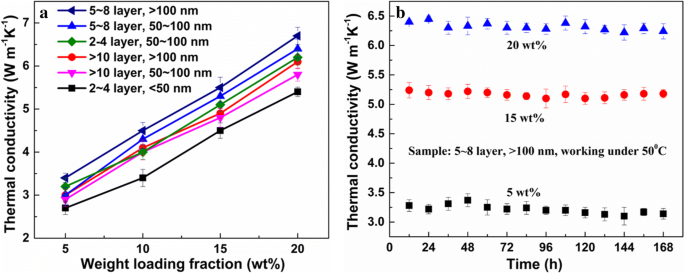
Figure 4a correlates thermal performance with RGO morphology: average size > 100 nm and thickness ~2 nm yield the highest conductivity (6.7 W m⁻¹ K⁻¹), 25 % higher than prior reports [7,10]. The impact of size dominates over thickness, highlighting the importance of basal‑plane contact area. Mass ratios of 10 wt % 3DGNs to 20 wt % RGO optimise the synergy without compromising adhesion. Stability tests (Figure 4b) show negligible conductivity loss over 7 days at 50 °C, underscoring suitability for high‑temperature electronics.
Mechanical testing (Table 2) reveals that the RGO(OOH)‑3DGNs‑ER composite achieves the highest ultimate strength (~10 % above other TIMs) and a stretching limit of 280 %, thanks to robust chemical bonding at the interface. Adhesive properties (Table 3) confirm that the RGO(OOH) filler restores joint strength to levels comparable with neat ER, whereas RGO(O) and RGO(OH) composites suffer from agglomeration and weak adhesion. These results demonstrate that carboxyl‑rich RGO not only enhances thermal transport but also preserves mechanical integrity. SEM images of a RGO(OOH), b 3DGNs, c pristine ER, d RGO(OOH)-ER, e RGO(OH)-ER, f RGO(O)-ER, g 3DGNs-ER, and h 3DGNs-RGO(O)-ER. Digital photos of ER, RGO filler, and RGO‑3DGNs‑ER are shown in the insets. Scale bars represent 2 µm. Cross‑sectional view is inset in h. Raman spectra of natural graphite and the various graphene fillers. The 3DGNs lack a D‑band, confirming high crystallinity, whereas GO shows a strong D‑band due to defects. After reduction, the D‑band diminishes and the 2D‑band reappears, allowing defect density assessment via I_D/I_G (Table S1). Thermal conductivities of composite TIMs with varying RGO loadings. RGO(OOH) outperforms RGO(OH) and RGO(O). The inset compares RGO(OOH)-3DGNs-ER with previously reported graphene‑assisted ER, confirming the superior role of carboxyl groups. Adding 3DGNs increases conductivity to 6.1 W m⁻¹ K⁻¹. Interface thermal boundary resistance (δ) is a key metric for interfacial contact. Using Balandin’s expression $$K={K}_g\left[\frac{2p\left({K}_g-{K}_e\right)+3{K}_e}{\left(3-p\right){K}_g+{K}_ep+\frac{\delta {K}_g{K}_ep}{H}}\right]$$ where p is the volume fraction of graphene, K, K_g, and K_e are the composite, graphene, and ER conductivities, H is graphene thickness, and δ is the thermal boundary resistance, we found that δ is minimized for RGO(OOH) (Table 1). This aligns with the observed conductivity gains and confirms that carboxyl groups form strong chemical bonds with the epoxy network during curing at 110 °C. a Relationship between thermal performance and RGO morphology at various loadings. b Thermal conductivity stability of TIMs with different RGO loadings at 50 °C over 7 days. Natural graphite and acetone were sourced from Aladdin Co., Ltd. ER and its curing agent were purchased from Sanmu Co. Ltd. (Suzhou, China). Reagents such as silver nitrate, potassium carbonate, ethanol, sodium hydroxide, phosphorus pentoxide, chloroacetic acid, hydrochloric acid, potassium permanganate, hydrazine peroxide, and sulfuric acid were obtained from Beijing Chemical Reagent Plant. Deionised water (18 MΩ cm) was used for all aqueous solutions. GO was prepared by a modified Hummer’s method and Zhang’s protocol, yielding carboxyl and hydroxyl groups respectively [34,35]. For RGO(OOH), natural graphite underwent the modified Hummer’s oxidation followed by alcohol reduction; for RGO(OH), Zhang’s oxidation and alcohol reduction; for RGO(O), hydroxyl groups were first converted to carboxyl via NaOH and chloroacetic acid, then carboxyls were removed with silver nitrate and potassium carbonate [19,24]. RGO dispersions were sonicated (60 min) in ethylene glycol, heated to 160 °C for 5 h, and washed thrice with water before drying at 60 °C in a vacuum oven.
ER was mixed with 5–20 wt % RGO (or 10 wt % 3DGNs) under mild stirring, then cured at 110 °C for 2 h. 3DGNs were fabricated by CVD on nickel foam: heating to 1100 °C under Ar/H₂, introducing CH₄ for 2 min, then cooling under Ar/H₂. Composite molds were prepared by layering ER (or RGO‑ER) over 3DGNs and curing at 110 °C for 5 h. RGO size was tuned by varying sonication duration (0–12 h). SEM (FEI Sirion 200, 5 kV) provided morphology. Raman spectroscopy (532 nm) assessed defect density. XPS (RBD PHI‑5000C) quantified surface functional groups. Thermal diffusivity and specific heat were measured by laser flash analysis (LFA 2000) and DSC (Diamond DSC), enabling conductivity calculation: k = α × ρ × Cₚ. Mechanical properties were evaluated using DMTA (Triton) in dual‑cantilever mode for Young’s modulus and ASTM D1002‑01 for lap‑shear strength (Al‑Al joints, 0.2 mm TIM thickness, 25 × 12.5 mm² overlap). Surface preparation involved abrasive blasting, degreasing with methyl ethyl ketone, and NaOH etching. By tailoring the surface chemistry of RGO and combining it with 3DGNs, we achieved a TIM that simultaneously excels in thermal conductivity (6.7 W m⁻¹ K⁻¹, 3 250 % over pristine ER) and mechanical robustness. Carboxyl‑rich RGO(OOH) provides a chemical bridge that lowers interface thermal boundary resistance and enhances covalent bonding with the epoxy matrix. Optimised RGO size (~> 100 nm) and thickness (~2 nm) further maximise basal‑plane contact. The resulting composite demonstrates stable performance at 50 °C and superior adhesion, underscoring the viability of functional‑group optimisation for next‑generation thermal management solutions. Three‑dimensional graphene networks Specific heat Dynamic mechanical thermal analysis Differential scanning calorimetry Epoxy resin Graphene oxide Thermal conductivity Reduced graphene oxide RGO with epoxy as primary functional group RGO(O)-modified ER RGO with hydroxyl as primary functional group RGO(OH)-modified ER RGO with carboxyl as primary functional group RGO(OOH) and 3DGNs co‑modified ER RGO(OOH)-modified ER RGO and 3DGNs co‑modified ER Standard‑state cubic centimetre per minute Scanning electron microscope Thermal interface materials X‑ray photoelectron spectroscopy Thermal diffusion coefficient DensityAbstract
Background
Results and Discussion

Methods
Materials
Preparation
Characterization
Conclusions
Abbreviations
Nanomaterials
- Comprehensive Atomic & Electronic Analysis of Stacked-Cup Carbon Nanofibers
- High‑Performance Thermal Interface Materials via Co‑Modification of Epoxy Resin with Reduced Graphene Oxide Nanosheets and 3‑D Graphene Networks
- From Nanoscale to Macroscale: How Rough Surfaces' Contact Area Evolves Under Normal Load
- Impact of Morphology and Crystal Structure on Titania Nanotube Thermal Conductivity
- Lowering Metal–Ge Contact Resistance with ZnO Interlayers and Argon Plasma: Achieving Ohmic Al Contacts on n‑Ge
- Optimizing ZnO‑Based Nanohybrids: How Materials, Heterojunctions, and Crystal Orientation Enhance Methyl Orange Degradation
- High‑Temperature Solutions: Integrating CFRP with Ceramic Matrix Composites
- Understanding DC vs AC Motors: Key Differences and Buying Guide
- Industrial Friction Materials Explained: Brake Pads vs. Brake Shoes
- Master the ADS1115: Features, Pinouts, and Arduino Integration Guide



