Post‑Treatment Strategies to Modulate Photoluminescence of Nitrogen‑Doped Carbon Dots: Solvent, Reduction, and Metal‑Induced Enhancements
Abstract
Recent advances in nitrogen‑doped carbon dots (NCDs) have highlighted their tunable photoluminescence (PL) as a promising feature for optical sensing and lighting. However, the precise mechanism behind PL modulation remains debated. Here, we report a facile hydrothermal synthesis of yellow‑emitting NCDs and systematically evaluate three post‑treatment strategies—solvent exchange, chemical reduction with NaBH₄, and metal‑ion addition—to probe the influence of surface states on PL. Solvent‑dependent studies reveal hydrogen‑bond‑mediated PL shifts and intensity changes. NaBH₄ treatment selectively reduces C=O to C–O groups, enhancing intrinsic (n–π*) emission. Ag⁺ addition induces aggregation‑induced emission enhancement (AIEE), further boosting fluorescence. These findings confirm that surface functional groups govern PL behavior, opening avenues for multi‑modal imaging and advanced lighting applications.
Background
Carbon dots (CDs) are emerging as eco‑friendly, biocompatible nanoscale emitters, offering tunable photoluminescence and versatile synthesis routes from inexpensive precursors such as citric acid, fruit extracts, and food waste [1–4]. Compared with conventional quantum dots (e.g., InP, ZnSe, ZnInS, CuInS), CDs lack heavy metals, simplify fabrication, and broaden application prospects in bio‑imaging, LED displays, fluorescent sensing, and photodetection [5–10]. Despite these advantages, CD emission has historically been confined to the blue region. Reports of longer‑wavelength emission often rely on excitation‑dependent spectral shifts rather than true tunability, and the intensity of shifted bands is typically weak, limiting practical use [11,12]. The origin of CD PL remains contested, attributed to intrinsic core emission or surface‑defect states [13,14]. Addressing this knowledge gap requires controlled post‑treatments that modulate surface chemistry while preserving core integrity. Surface states profoundly influence a nanomaterial’s optical, electronic, and catalytic properties [15–17]. For example, surface passivation enhances perovskite optoelectronics, and acid–base characteristics improve CeO₂ catalysis [18,19]. Solvent‑dependent PL, driven by hydrogen bonding between surface functional groups and solvent molecules, has been observed in nitrogen‑rich CDs, producing blue‑to‑green shifts [20,21]. Metal‑ion addition can also induce aggregation‑induced emission enhancement (AIEE), as demonstrated for Fe³⁺‑triggered CD aggregation [22]. In this study, we synthesize yellow‑emitting NCDs via a single‑step hydrothermal method and apply three post‑treatments—solvent exchange, NaBH₄ reduction, and Ag⁺ addition—to systematically investigate the role of surface states in PL modulation.
Methods
Chemicals
O‑Phenylenediamine (OPD, 99.9 %), analytical‑grade solvents (water, ethylene glycol, ethanol, DMSO, acetone, toluene), metal chlorides/nitrates, and HEPES were sourced from commercial suppliers and used without further purification. Milli‑Q water was employed for all aqueous solutions.
Synthesis of NCDs
0.05 g OPD was dissolved in 10 mL Milli‑Q water under vigorous stirring until clear. The solution was transferred to a 25 mL Teflon‑lined autoclave and heated at 180 °C for 6 h. After cooling, the product was centrifuged at 9,000 rpm to remove solids, and the supernatant was freeze‑dried for 24 h, yielding a light‑yellow powder.
Post‑Treatment Protocols
For solvent‑dependent studies, 0.01 g of dried NCDs was dispersed in 10 mL of each solvent (water, EG, ethanol, DMSO, acetone, toluene). NaBH₄ solutions (10 mL, 0–0.04 g mL⁻¹) were added to 0.01 g NCDs to investigate reduction effects. Metal‑ion titrations involved adding 50 µM of Ag⁺, Cd²⁺, Cs⁺, Cu²⁺, Fe³⁺, In³⁺, Mg²⁺, Mn²⁺, Pb²⁺, or Zn²⁺ to 10 mL of 0.01 g NCDs in 10 mM HEPES buffer (pH 7.2). Ag⁺ concentrations were varied from 0 to 300 µM to study AIEE.
Characterization
NCD morphology and size were examined by HRTEM (JEOL JEM‑2s100F). Structural analysis used XRD (Bruker D8 Advance) and XPS (Thermo ESCALAB 250XI). FT‑IR spectra were recorded on a Nicolet 6700. UV‑vis absorption and steady‑state PL were measured with a Shanghai Lengguang 759S spectrophotometer and F97XP fluorescence spectrometer, respectively. Time‑resolved PL lifetimes were obtained using an Edinburgh Instruments FLS 920. Zeta potentials were determined with a Malvern Zetasizer.
Results and Discussion
Structural Characterization
HRTEM images (Fig. 1) reveal monodisperse NCDs with diameters ranging from 3 to 6 nm (average 3.96 nm). Lattice fringes at 0.21 nm and 0.32 nm correspond to the (100) and (002) planes of graphitic carbon. XRD shows a broad peak near 20° 2θ, indicating small crystalline cores with disordered surfaces.
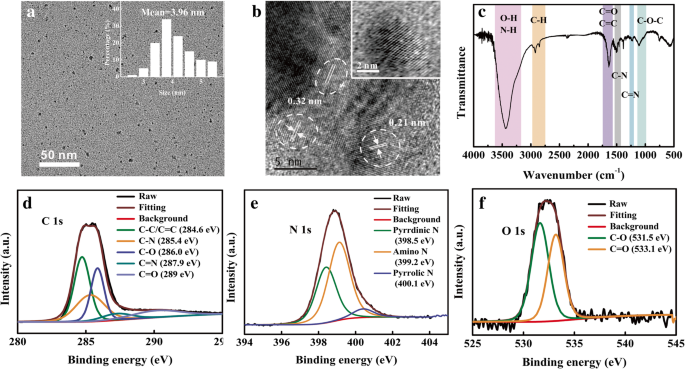
FT‑IR analysis identifies abundant surface groups: O–H/N–H (3100–3600 cm⁻¹), C=O/C=C (1570–1750 cm⁻¹), C–N/C=N (1411/1239 cm⁻¹), and C–O–C (990–1170 cm⁻¹). XPS confirms the presence of C, N, and O (73.8 %, 22.6 %, 3.6 %). High‑resolution C 1s peaks correspond to sp² C–C/C=C, C–N, C–O, C=N, and C=O; N 1s peaks reveal pyridinic, amino, and pyrrolic nitrogen; O 1s peaks correspond to C–O and C=O bonds.
Solvent‑Dependent Photoluminescence
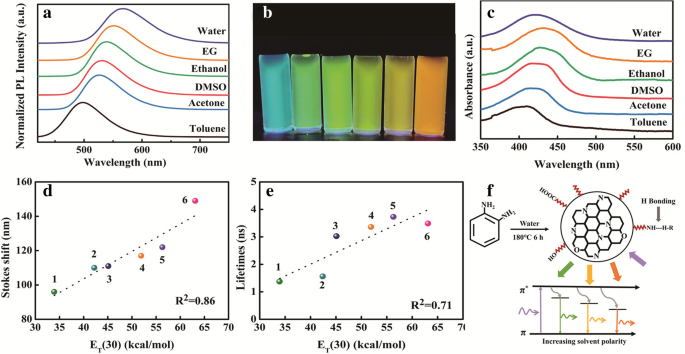
Emission spectra across six solvents (water, EG, ethanol, DMSO, acetone, toluene) show a red‑shift from 500 to 569 nm under 365 nm excitation, with intensity enhancements varying by solvent (Fig. 2a–b). UV‑vis absorption mirrors this trend, shifting to longer wavelengths with increasing solvent polarity (except water, due to limited solubility). PL lifetimes and Stokes shifts increase linearly with the E_T(30) polarity index, indicating stronger hydrogen bonding between solvent and surface functional groups enhances PL (Fig. 2d–e). The observed tunability is attributed to solvent‑mediated modulation of surface‑state contributions to the emissive transition.
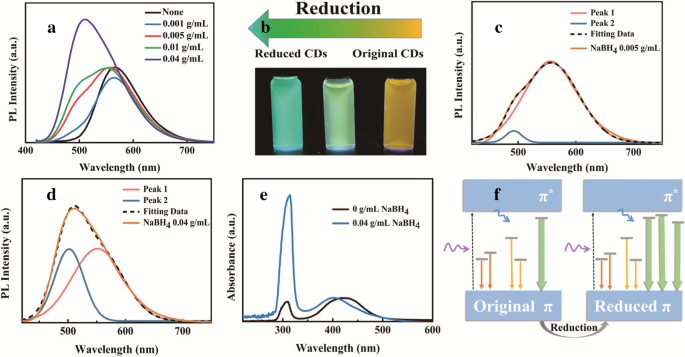
Reduction with NaBH₄
NaBH₄ treatment (0–0.04 g mL⁻¹) significantly brightens PL and blue‑shifts the emission from 567 to 510 nm (Fig. 3a). Deconvolution reveals two Gaussian components (peak 1 ~ 565 nm, peak 2 ~ 496 nm); increasing NaBH₄ raises peak 2 intensity while peak 1 remains stable (Fig. 3c–d). XPS shows increased oxygen and decreased nitrogen content, consistent with C=O reduction to C–O bonds. UV‑vis absorption displays enhanced 320 nm band (n–π*) intensity, corroborating a shift toward intrinsic C–O–C emission. PL lifetime shortens from 3.48 to 2.2 ns, confirming reduced non‑radiative pathways.
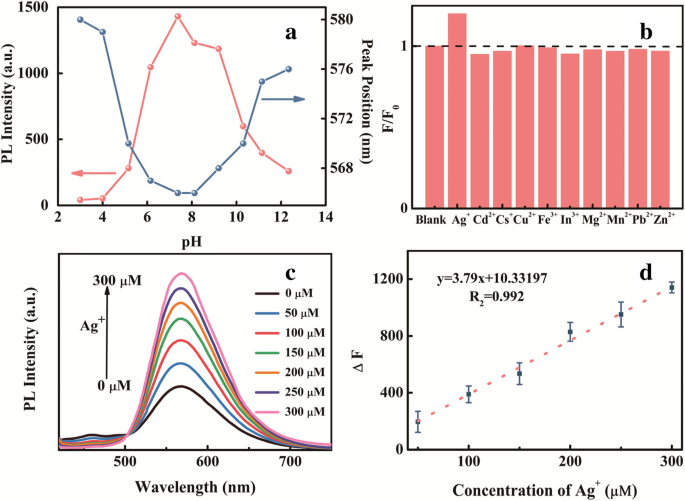
Metal‑Enhanced Photoluminescence
Among tested metal ions, Ag⁺ uniquely amplifies PL intensity without shifting the emission peak (Fig. 4c). A linear relationship (R² = 0.992) exists between ΔF and Ag⁺ concentration (0–300 µM) (Fig. 4d). Time‑resolved PL shows a subtle lifetime change, suggesting complex formation. Zeta potential shifts from –34.0 mV (neutral) to –27.8 mV upon Ag⁺ addition, reducing electrostatic repulsion and promoting aggregation‑induced emission enhancement (AIEE). Aggregation observations (Fig. S9) confirm that Ag⁺‑induced clustering passivates surface defects, yielding brighter fluorescence.
Conclusion
We successfully synthesized yellow‑emitting NCDs via a simple hydrothermal route and demonstrated that PL can be precisely tuned by three post‑treatments: solvent exchange, NaBH₄ reduction, and Ag⁺ addition. Solvent‑mediated hydrogen bonding shifts and amplifies PL; chemical reduction transforms C=O to C–O, enhancing intrinsic (n–π*) emission; and Ag⁺ triggers AIEE, boosting fluorescence. These results validate the central role of surface functional groups in governing NCD photophysics and suggest practical strategies for designing advanced luminescent probes and lighting devices.
Abbreviations
- AIEE:
Aggregation‑induced emission enhancement
- CDs:
Carbon dots
- DMSO:
Dimethyl sulfoxide
- EG:
Ethylene glycol
- FT‑IR:
Fourier transform‑infrared spectroscopy
- HB:
Hydrogen bonding
- HEPES:
4-(2‑Hydroxyethyl)piperazine‑1‑ethanesulfonic acid
- HRTEM:
High‑resolution transmission electron microscopy
- NCDs:
Nitrogen‑doped carbon dots
- OPD:
O‑Phenylenediamine
- PL:
Photoluminescence
- QDs:
Quantum dots
- rpm:
Revolution per minute
- UV‑vis:
Ultraviolet‑visible
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffraction
Nanomaterials
- High‑Efficiency, Excitation‑Independent Blue‑Emitting Carbon Dots with Tunable Photoluminescence
- Boosting MgZnO MSM Photodetector Performance with Pt Nanoparticle-Driven Surface Plasmons
- High‑Performance Dual‑Emissive Mn‑Doped InP/ZnS Quantum Dots with 78 % Photoluminescence Quantum Yield: A Growth‑Doping Approach
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- Temperature and Excitation Intensity Modulate Photoluminescence in InGaAs/GaAs Surface Quantum Dots
- How Edge Functionalization Alters Fluorescence in Carbon Quantum Dots
- Carbon Dots: Advanced Fluorescent Platforms for Sensitive Metal‑Ion Detection
- Affordable Green Fluorescent Carbon Dots: Enhanced Bioimaging & Targeted Drug Delivery
- Bright, Multicolor N‑Doped Carbon Dots from Ascorbic Acid and Phenylenediamine: Tunable Emission via Solvent Polarity and pH
- Stable Carbon‑Dot Frameworks with Superior Antibacterial Efficacy



