Fast Microwave‑Synthesis of Pyridinic‑Rich N,S‑Co‑Doped Carbon Quantum Dots with Superior Photoluminescence and Catalase‑Mimicking Activity
Abstract
We report a 50‑second microwave‑assisted synthesis of nitrogen and sulfur co‑doped carbon quantum dots (N,S‑CQDs) that exhibit an unprecedented pyridinic‑rich structure. This design yields both a high photoluminescence quantum yield (23.6 %) and robust enzyme‑mimic catalytic activity, outperforming conventional CQDs.
Background
Carbon quantum dots (CQDs) have emerged as a versatile zero‑dimensional material, prized for their chemical stability, low cytotoxicity, and unique electronic and optical properties [1]–[3]. Their surface functional groups (–OH, –COOH) enable integration with a wide range of organic and inorganic systems, facilitating applications in bio‑imaging [4], optoelectronics, and photocatalytic degradation or hydrogen production [6]–[8]. Recent studies confirm that heteroatom doping—particularly with nitrogen and sulfur—significantly enhances CQD performance [9]–[10]. N‑doped or N,S‑co‑doped CQDs routinely surpass pristine dots in fluorescence quantum efficiency and photocatalytic activity [11]–[12], with performance correlated to the dopant concentration [13], [14]. However, conventional hydrothermal routes struggle to incorporate high dopant loads due to the high solubility of inorganic precursors, resulting in low N‑doping levels in the final CQDs.
Here, we present a rapid microwave‑assisted protocol that delivers N,S‑co‑doped CQDs (N,S‑CQDs) with nitrogen and sulfur contents of 12.8 % and 7.2 % (wt.), respectively—approximately five‑fold and three‑fold higher than previously reported N‑ or N,S‑co‑doped CQDs [11], [14]—by using citric acid (CA) as the carbon source and thiourea as both a heteroatom source and a weak base.
Methods
In a porcelain crucible, 0.42 g (2 mmol) of citric acid monohydrate and 0.46 g (6 mmol) of thiourea were mixed and subjected to 50 s of microwave irradiation (445 W). The resulting brownish‑yellow powder was dispersed in 30 mL of deionized water, centrifuged at 9 000 rpm for 20 min, filtered (0.22 µm), dialyzed (1 kDa MWCO) for 24 h, and finally freeze‑dried under vacuum. Pristine CQDs were synthesized from citric acid alone following the same post‑processing steps.
The peroxidase‑like activity of N,S‑CQDs was evaluated in a 30 mL citric acid–disodium hydrogen phosphate buffer (pH ≈ 3.5, 35 °C) containing 1 µg mL⁻¹ N,S‑CQDs and 8 × 10⁻⁴ M tetramethylbenzidine (TMB). After adding 160 µL of 30 % H₂O₂, the blue TMB oxidation product was monitored at 652 nm every 2 min to calculate reaction rates. Reusability was tested in a 60 mL buffer with 2 µg mL⁻¹ N,S‑CQDs and 5 × 10⁻³ M TMB; after each 1‑h cycle, fresh H₂O₂ (0.3 %, 320 µL) was added for a total of four cycles.
Structural and optical characterizations were performed using TEM (JEM‑2100, 200 kV), HRTEM, SAED (FEI T20, 200 kV), UV/vis (Shimadzu UV‑3600), fluorescence (Hitachi F‑7000, 700 V), time‑resolved PL (Horiba TCSPC), XRD (Bruker D8 Advance, Cu Kα), FT‑IR (Nicolet iS10), XPS (PHI 5000 Versa), and TG‑MS (Netzsch STA 449C) under N₂/air flow.
Results and Discussions
TEM images (Fig. 1a) reveal uniformly dispersed N,S‑CQDs with an average diameter of 2.0 nm. HRTEM shows a clear lattice fringe of 0.24 nm corresponding to the (1120) facet of graphene, confirming a graphitic core structure. SAED (Fig. 1b) further confirms crystallinity with a d‑spacing of 0.312 nm, matching reported N‑ or N,S‑co‑doped CQDs. XRD displays a broad peak at 2θ ≈ 25.5°, indicative of interlayer spacing (0.33 nm) typical for graphitic carbon. Compared with g‑CNQDs or β‑C₃N₄ synthesized via long hydrothermal treatments, the rapid 50‑s microwave route preserves a graphitic framework while incorporating high heteroatom loads.
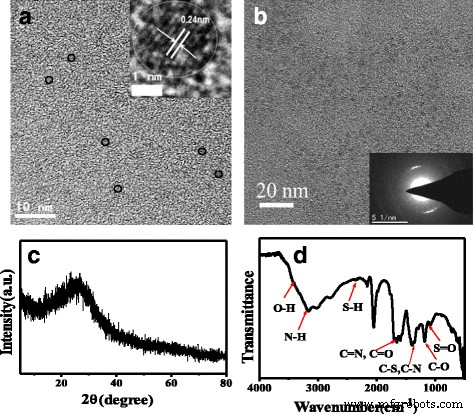
a TEM (HRTEM inset) and b SAED of N,S‑CQDs; c XRD and d FT‑IR spectra.
FT‑IR (Fig. 1d) confirms abundant surface functionalities: N–H and O–H stretching (3163 and 3416 cm⁻¹), C=O and C=N (1704 cm⁻¹), amide I/II (1656 and 1582 cm⁻¹), C–S (1405 cm⁻¹), C–N (1345 cm⁻¹), and S=O (1177 cm⁻¹). UV/vis (Fig. 2a) shows a strong π–π* absorption at 234 nm and a weaker n–π* band at 340 nm, reflecting a high density of aromatic sp² domains and successful pyridinic N incorporation.
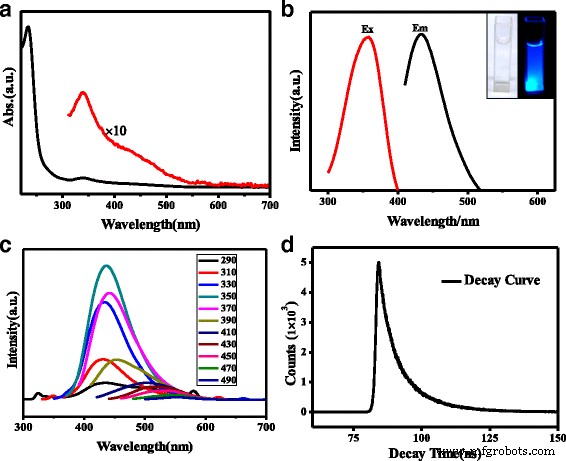
a UV/vis absorption; b PL spectrum (exitation 358 nm, emission 436 nm) and visual under 365 nm; c PL at varying excitation wavelengths; d PL decay (τ₁ = 3.48 ns, τ₂ = 11.05 ns, τ_avg = 6.72 ns).
Photoluminescence quantum yield (PLQY) of N,S‑CQDs reaches 23.6 % (excitation 358 nm), a three‑fold improvement over N‑ or N,S‑doped CQDs reported in the literature. In contrast, pristine CQDs exhibit only 1.15 %. The elevated PLQY correlates with the high pyridinic N content (12.8 % N) and the strong electron‑rich graphitic framework, which promote radiative recombination. XPS confirms the presence of N and S: C 1s shows sp² (284.4 eV), sp³ (285.6 eV), and C=O/C=N (288.1 eV) components; N 1s displays pyridinic (399.5 eV), pyrrolic (400.3 eV), and amidic (401.0 eV) peaks, with pyridinic N dominating (ratio ≈ 1.5:1). S 2p peaks at 163.3 and 164.4 eV confirm thiophenic S incorporation at the edges.
Thermal analysis (TG‑DTA) shows that the presence of thiourea lowers the melting and carbonization temperatures of citric acid by ~30 °C, facilitating rapid in‑situ doping and preventing sublimation. The resulting material retains a high residual mass (≈ 21 %) compared to pure citric acid, confirming efficient heteroatom integration.
Enzyme‑mimic assays demonstrate that N,S‑CQDs catalyze H₂O₂‑mediated oxidation of TMB with an initial rate of 2.16 × 10⁻³ µmol µmol⁻¹ L⁻¹ s⁻¹—twice that of pristine CQDs and comparable to natural horseradish peroxidase under identical conditions. The superior activity is attributed to the abundant pyridinic N, which serves as active sites for H₂O₂ decomposition. Reusability tests over four cycles show negligible loss of activity, underscoring the stability of the catalytic sites.
Conclusions
We have developed a microwave‑solid‑phase protocol that, in just 50 s, produces pyridinic‑rich N,S‑co‑doped CQDs with 12.8 % N and 7.2 % S content. Thiourea acts as both a heteroatom source and a weak base, enabling rapid dehydration and multi‑step carbonization at lower temperatures, which promotes high dopant incorporation and a dominant pyridinic N framework. The resulting N,S‑CQDs exhibit an exceptional PLQY of 23.6 % and catalase‑mimicking activity twice that of pristine CQDs, while maintaining excellent stability across repeated catalytic cycles. This work offers a scalable, high‑performance route to multifunctional CQDs for optoelectronic and biocatalytic applications.
Nanomaterials
- High‑Efficiency, Excitation‑Independent Blue‑Emitting Carbon Dots with Tunable Photoluminescence
- Hydrothermal Synthesis of Blue‑ and Green‑Emitting Carbon Quantum Dots from Tofu Wastewater
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- Blue‑Emitting Mn‑Alloyed CdTe Quantum Dots: Synthesis, Structural Insight, and Electrochemical Characterization
- One‑Pot Room‑Temperature Synthesis of 10‑nm 2D Ruddlesden–Popper Perovskite Quantum Dots with Tunable Emission
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- Synthesis, Structure, and Superior Capacitance of Sulfur‑Nitrogen Co‑Doped Graphene Quantum Dots
- How Edge Functionalization Alters Fluorescence in Carbon Quantum Dots
- Carbon Dots: Advanced Fluorescent Platforms for Sensitive Metal‑Ion Detection



