Green Synthesis of Gold Nanoparticles from Artemisia capillaris, Portulaca oleracea, and Prunella vulgaris Extracts: Antioxidant Activity and Cytotoxicity Evaluation
Abstract
Three aqueous plant extracts—Artemisia capillaris, Portulaca oleracea, and Prunella vulgaris—were employed to biofabricate gold nanoparticles (AuNPs). Their antioxidant profiles, including free‑radical scavenging capacity, total phenolic content, and reducing power, were quantified and correlated with the efficiency of AuNP synthesis. Prunella vulgaris displayed the strongest antioxidant activity and served as the most effective reducing agent, yielding AuNPs (PV‑AuNPs) with a surface‑plasmon resonance (SPR) peak at 530 nm and diverse morphologies. High‑resolution X‑ray diffraction confirmed a face‑centred cubic crystal lattice, and inductively coupled plasma optical emission spectroscopy (ICP‑OES) estimated a 99.3 % reaction yield. Dynamic light scattering revealed a hydrodynamic diameter of 45 ± 2 nm and a zeta potential of –13.99 mV. PV‑AuNPs exhibited dose‑dependent antioxidant activity and significant cytotoxicity toward human colorectal adenocarcinoma (HT‑29) cells without fetal bovine serum (FBS), whereas pancreatic ductal adenocarcinoma (PANC‑1) cells showed maximal response in the presence of FBS. These findings highlight P. vulgaris extract as a potent, eco‑friendly reducing agent for producing bioactive AuNPs with therapeutic potential.
Introduction
The growing demand for metallic nanoparticles, particularly gold nanoparticles (AuNPs), stems from their versatility as drug carriers, catalysts, imaging agents, and biosensors [2]. Conventional synthesis routes—ion sputtering, reverse micelle formation, and chemical reduction—often involve costly reagents and raise toxicity concerns. Green synthesis, leveraging plant extracts as natural reducing, capping, and stabilizing agents, offers a cost‑effective, scalable, and biocompatible alternative [2]. Phytochemicals such as phenolics, terpenoids, and flavonoids not only reduce metal ions but also impart antioxidant properties and enhance nanoparticle stability.
Artemisia capillaris (family Asteraceae) has long been used in traditional medicine for its antibacterial, anti‑inflammatory, and hepatoprotective effects, attributed to constituents like capillin and scoparone [4‑6]. Portulaca oleracea (family Portulacaceae) possesses antibacterial, antifungal, and antioxidant activities, linked to alkaloids, fatty acids, and flavonoids [7‑8]. Prunella vulgaris (family Lamiaceae) exhibits antiviral, anticancer, immunomodulatory, and antioxidant effects, largely due to phenolic acids and triterpenoids such as rosmarinic acid and ursolic acid [9‑13].
The protein corona—an adsorption layer of serum proteins that forms upon nanoparticle entry into biological fluids—critically influences cellular uptake, biodistribution, and cytotoxicity [15‑17]. Surface charge, size, and morphology of AuNPs dictate corona composition, thereby modulating therapeutic efficacy. In this study, we examined how the antioxidant potency of the three plant extracts affects AuNP formation and how the resulting nanoparticles interact with cancer cells in the presence or absence of FBS.
Methods
Materials
All reagents were analytical grade. Potassium gold(III) chloride (KAuCl₄) and standard antioxidants (BHT, vitamin C) were sourced from Sigma‑Aldrich (St. Louis, MO, USA). Cell culture supplies were obtained from Gibco (Thermo Fisher Scientific) and GE Healthcare HyClone™ (Victoria, Australia). Detailed reagent specifications are listed in the supplementary table.
Instruments
UV‑visible spectra were recorded on a Shimadzu UV‑2600 spectrophotometer (λ = 300–800 nm). HR‑TEM imaging employed a JEM‑2100F (JEOL, 200 kV). X‑ray diffraction used a Rigaku Miniflex (CuKα, 1.54056 Å). ICP‑OES analysis was performed on a Perkin Elmer Optima 8300. Dynamic light scattering and zeta potential measurements were obtained with a NanoBrook 90 Plus analyzer. All instruments were calibrated according to manufacturer guidelines.
Preparation of Aqueous Extracts
Dried aerial parts of P. vulgaris and A. capillaris were sourced from Omniherb (Daegu, Republic of Korea); P. oleracea was purchased from Handsherb (Youngchun, Republic of Korea). Each plant (100 g) was sonicated in 1 L deionized water at 25 °C for 3 h, repeated thrice. The filtrate was freeze‑dried to yield a powder, stored at –20 °C until use.
Antioxidant Assays
Free‑radical scavenging (DPPH and ABTS), reducing power, and total phenolic content were quantified following standard protocols. All assays were conducted in triplicate, and results expressed as IC₅₀ (µg mL⁻¹) or mg GAE g⁻¹. Detailed methodologies are provided in the supplementary materials.
Biofabrication of AuNPs
Stock solutions of KAuCl₄ (10 mM) and plant extracts (2 % w/v) were prepared. After centrifugation (18,500 g, 30 min, 18 °C), supernatants were mixed to final concentrations of 0.5 mM Au and 0.05 % w/v extract in 2 mL vials. The reaction proceeded at 37 °C for 5 h. UV‑vis spectra were recorded to monitor SPR development.
Cell Culture
HT‑29 (colon), PANC‑1 (pancreas), and MDA‑MB‑231 (breast) cells were maintained in high‑glucose DMEM supplemented with 10 % FBS, 100 U mL⁻¹ penicillin, and 100 µg mL⁻¹ streptomycin at 37 °C, 5 % CO₂.
Cytotoxicity Assay
Cell viability was assessed via the EZ‑CYTOX WST assay. Cells (5 × 10³ cells mL⁻¹) were seeded in 96‑well plates, incubated 24 h, then treated with PV‑AuNPs (0–478.3 µg mL⁻¹ Au) ± 10 % FBS for 24 h. Absorbance at 450 nm was recorded, with background subtraction of PV‑AuNP intrinsic absorbance.
Statistical Analysis
Data are presented as mean ± SE. Significance was evaluated by one‑way ANOVA followed by Tukey’s post‑hoc test (p < 0.05). Analyses were performed with GraphPad Prism 5.02.
Results and Discussion
Antioxidant Profiles of the Extracts
The extraction yields were 14.1 % (A. capillaris), 39.12 % (P. oleracea), and 28.6 % (P. vulgaris). Antioxidant assays revealed that P. vulgaris exhibited the lowest IC₅₀ values for DPPH (50.35 ± 1.22 µg mL⁻¹) and ABTS (38.6 ± 0.44 µg mL⁻¹), surpassing BHT (71.37 ± 0.84 µg mL⁻¹). Reducing power and total phenolic content followed the same trend: 162.98 µg mL⁻¹ (P. vulgaris) vs. 235.38 µg mL⁻¹ (A. capillaris) vs. 396.16 µg mL⁻¹ (P. oleracea). Total phenolics were 90.53 ± 6.81 mg GAE g⁻¹ for P. vulgaris, 39.88 ± 4.55 mg GAE g⁻¹ for A. capillaris, and 18.32 ± 2.76 mg GAE g⁻¹ for P. oleracea. These results confirm that P. vulgaris harbors the most potent antioxidant constituents.
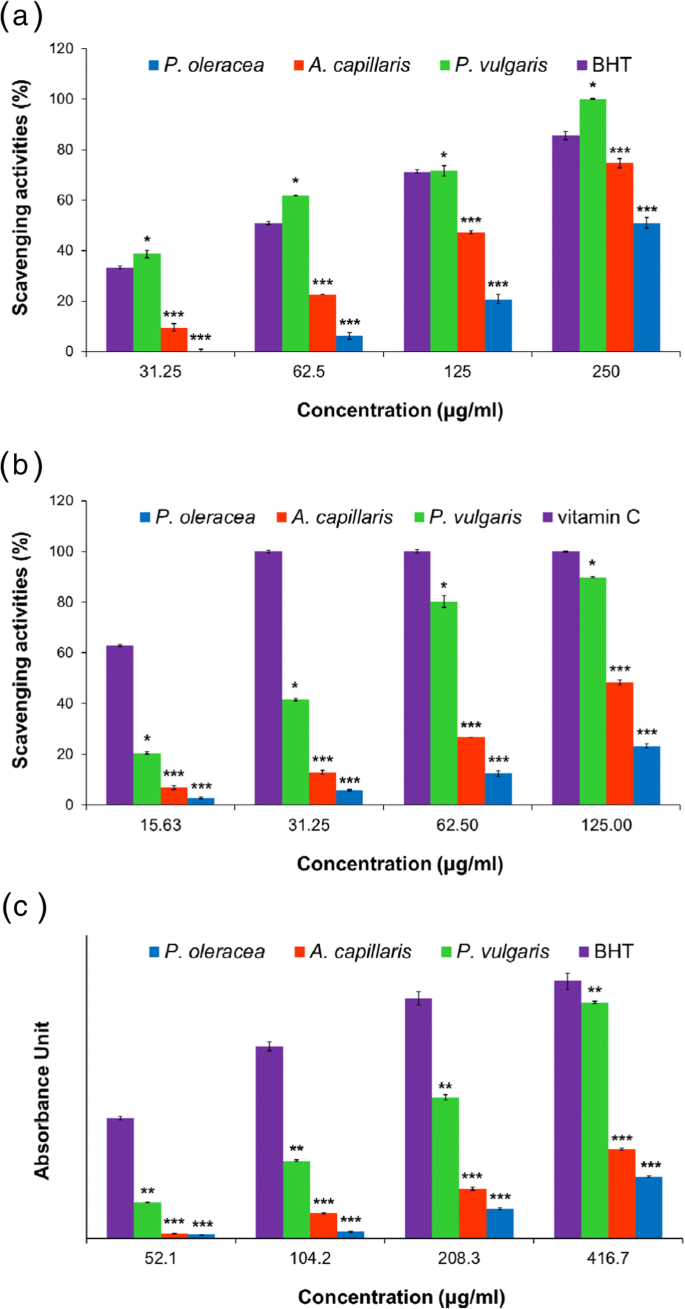
DPPH, ABTS, and reducing‑power activities of the three extracts. a DPPH scavenging; b ABTS scavenging; c Reducing power. Data are mean ± SEM; *p < 0.05, **p < 0.01, ***p < 0.001 versus standard.
Green Synthesis of AuNPs
Each extract facilitated AuNP formation, as evidenced by distinct SPR peaks and visible color changes: PO‑AuNPs (dark blue, broad 500–700 nm band), AC‑AuNPs (brown, 555 nm peak), and PV‑AuNPs (wine‑red, 530 nm peak). The superior antioxidant activity of P. vulgaris translated into higher SPR intensity, narrower bandwidth, and improved colloidal stability. PO‑AuNPs aggregated after 30 min at 25 °C, indicating the lowest stability among the three.

Photographs of PO‑AuNPs, AC‑AuNPs, and PV‑AuNPs.
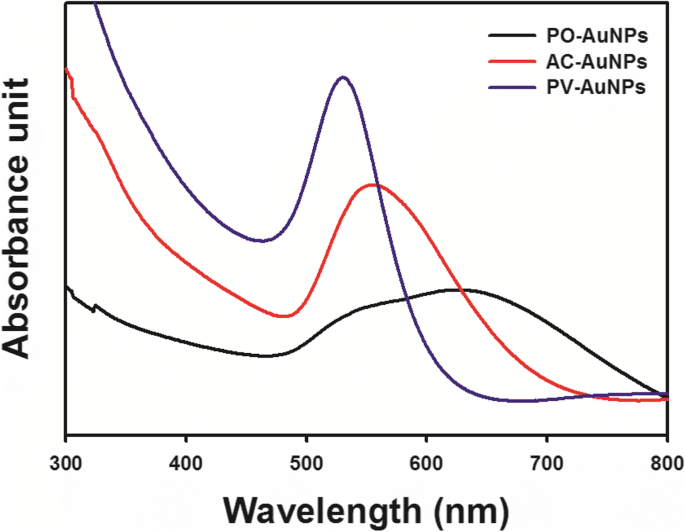
UV‑vis spectra of PO‑AuNPs (black), AC‑AuNPs (red), and PV‑AuNPs (blue).
Morphology and Size Distribution (PV‑AuNPs)
HR‑TEM revealed a diverse morphology: spheres (23.8 ± 1.3 nm), equilateral triangles (25.7 ± 2.2 nm), rods (28.4 ± 0.4 nm, aspect ratio = 2.4), and rhombi (long diagonal = 22.0 ± 0.6 nm, short diagonal = 15.4 ± 0.3 nm). All particles were crystalline, as confirmed by lattice fringes. Size histograms displayed Gaussian distributions for each shape.
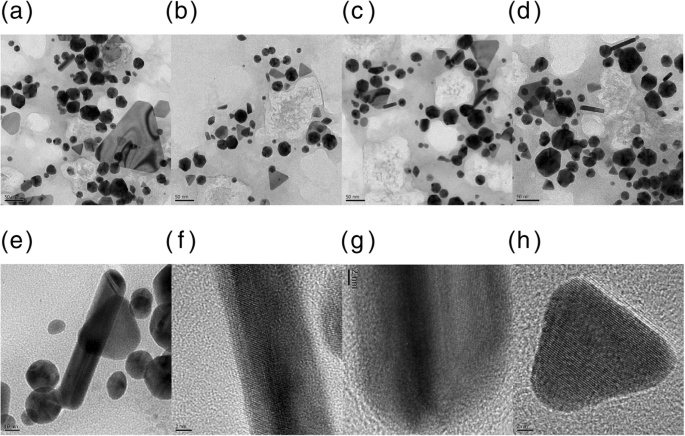
HR‑TEM images of PV‑AuNPs (scale bars: a 50 nm, b 50 nm, c 50 nm, d 50 nm, e 10 nm, f 2 nm, g 2 nm, h 2 nm).
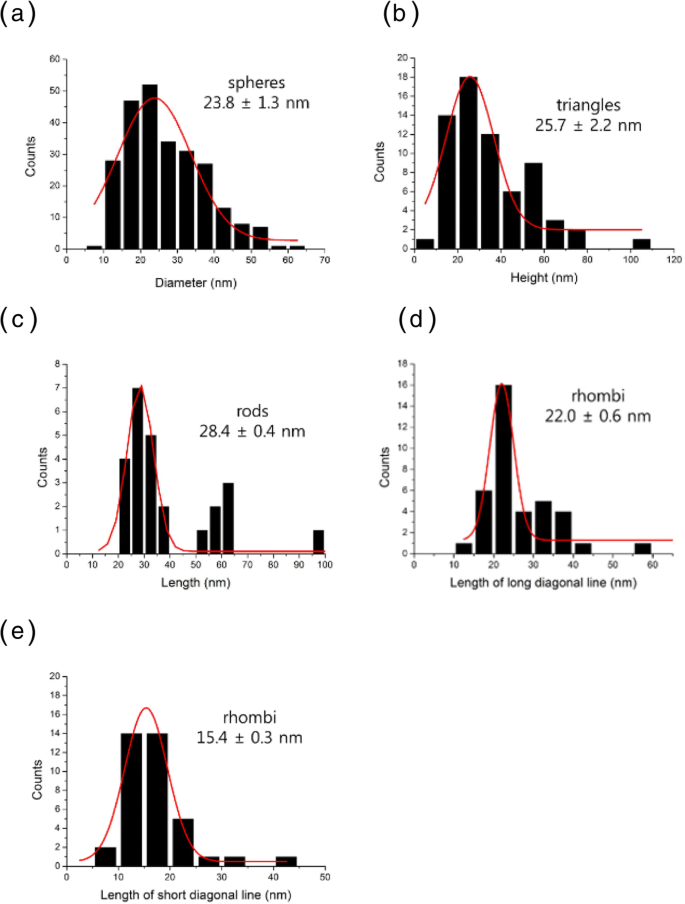
Size distributions of PV‑AuNPs: (a) spheres, (b) triangles, (c) rods, (d) rhombi (long diagonal), (e) rhombi (short diagonal).
Hydrodynamic Size and Surface Charge
DLS measurements indicated a hydrodynamic diameter of 45 ± 2 nm (polydispersity = 0.258) and a negative zeta potential of –13.99 mV, suggesting effective steric and electrostatic stabilization by surface‑bound phytochemicals.
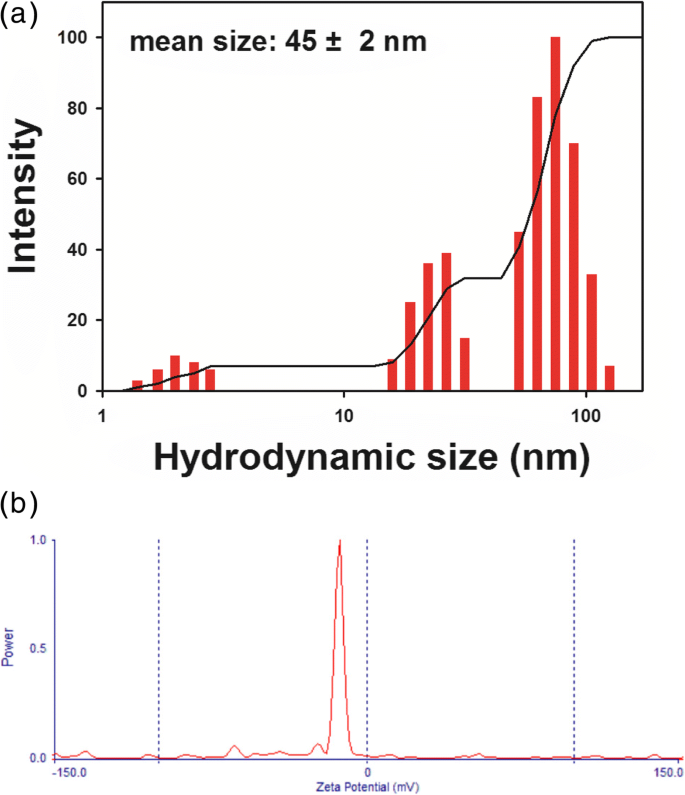
Hydrodynamic size (a) and zeta potential (b) of PV‑AuNPs.
Crystallinity (HR‑XRD)
HR‑XRD patterns displayed characteristic peaks at 2θ = 38.1°, 44.5°, 64.8°, and 77.4°, corresponding to the (111), (200), (220), and (311) planes of a face‑centred cubic lattice. Scherrer analysis of the (111) peak yielded an approximate crystallite size of 16.7 nm.
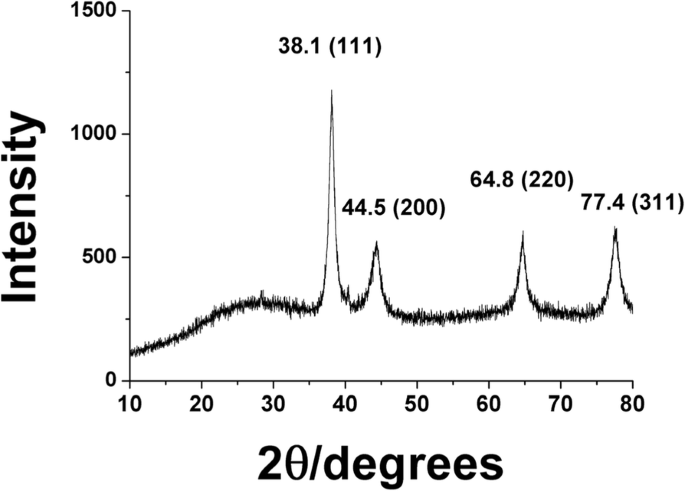
HR‑XRD analysis of PV‑AuNPs.
Reaction Yield
ICP‑OES quantified 96.337 ppm Au in the colloidal solution and 0.679 ppm in the supernatant, yielding a 99.3 % conversion efficiency.
Antioxidant Activity of PV‑AuNPs
PV‑AuNPs displayed dose‑dependent DPPH scavenging, with an IC₅₀ of 165.0 µg mL⁻¹ Au (≈49.4 µM vitamin C). This activity parallels that reported for plant‑mediated AuNPs and is attributed to surface‑bound phenolics.
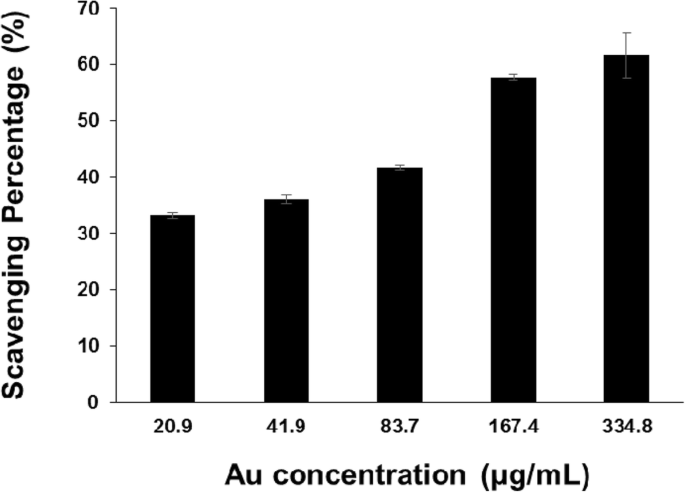
DPPH radical scavenging by PV‑AuNPs (Au concentration expressed in µg mL⁻¹).
Cytotoxicity in Cancer Cell Lines
In serum‑free conditions, HT‑29 cells were most sensitive to PV‑AuNPs (65.6 % inhibition at 478.3 µg mL⁻¹). PANC‑1 and MDA‑MB‑231 showed lower responses. With 10 % FBS, PANC‑1 cells exhibited enhanced cytotoxicity (37.5 % at 478.3 µg mL⁻¹), whereas HT‑29 cytotoxicity decreased, highlighting a protein‑corona‑dependent, tissue‑specific effect. The negative zeta potential likely promotes selective protein adsorption (e.g., fibrinogen), influencing cellular uptake and toxicity.
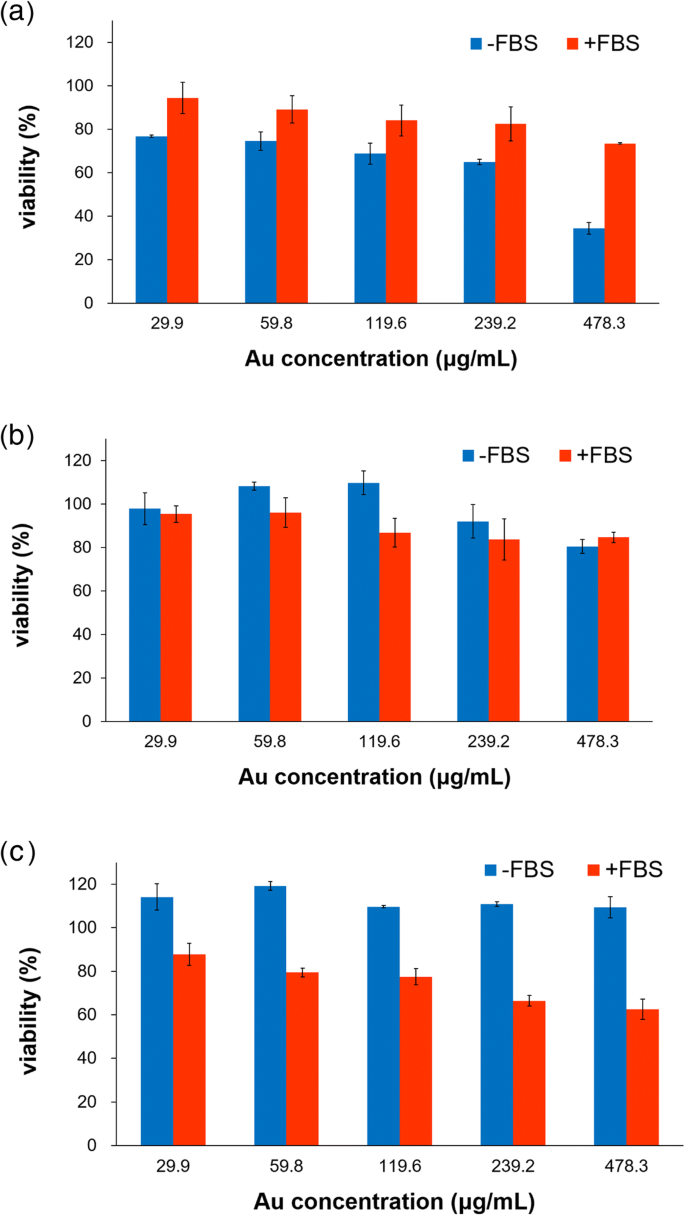
Cell‑viability of HT‑29 (a), MDA‑MB‑231 (b), and PANC‑1 (c) after PV‑AuNP treatment ± 10 % FBS.
Conclusions
The aqueous extracts of A. capillaris, P. oleracea, and P. vulgaris displayed measurable antioxidant activity. P. vulgaris emerged as the most potent reducing agent, yielding highly crystalline, wine‑red AuNPs (PV‑AuNPs) with a 99.3 % conversion rate and a hydrodynamic diameter of 45 ± 2 nm. PV‑AuNPs exhibited dose‑dependent antioxidant capacity and selective cytotoxicity toward colorectal, pancreatic, and breast cancer cells, with the protein corona modulating efficacy. These findings underscore the value of high‑antioxidant plant extracts in green nanoparticle synthesis and suggest PV‑AuNPs as promising candidates for targeted drug delivery, especially when combined with cardioprotective phytochemicals from P. vulgaris. Future work will focus on loading chemotherapeutics onto PV‑AuNPs to harness both anticancer and protective effects.
Abbreviations
- ABTS:
2,2′‑Azino‑bis(3‑ethylbenzothiazoline‑6‑sulfonic acid)
- AC‑AuNPs:
Gold nanoparticles synthesized from A. capillaris extract
- AgNPs:
Silver nanoparticles
- AuNPs:
Gold nanoparticles
- BHT:
Butylated hydroxytoluene
- DMEM:
Dulbecco’s modified Eagle’s medium
- DPPH:
2,2‑Diphenyl‑1‑picrylhydrazyl
- FBS:
Fetal bovine serum
- GAE:
Gallic acid equivalent
- HR‑TEM:
High‑resolution transmission electron microscopy
- HR‑XRD:
High‑resolution X‑ray diffraction
- HT‑29:
Human colorectal adenocarcinoma cell line
- IC50:
Half‑maximal inhibitory concentration
- ICP‑OES:
Inductively coupled plasma optical emission spectroscopy
- MDA‑MB‑231:
Human breast adenocarcinoma cell line
- PANC‑1:
Human pancreas ductal adenocarcinoma cell line
- PO‑AuNPs:
Gold nanoparticles synthesized from P. oleracea extract
- PV‑AuNPs:
Gold nanoparticles synthesized from P. vulgaris extract
- SPR:
Surface plasmon resonance
- WST assay:
Water‑soluble tetrazolium assay
Nanomaterials
- Gold Nanoparticles as Advanced Chemosensors: Enhancing Electrochemical Detection
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Amphiphilic Hyperbranched Polyglycerol Enables Size‑Controlled, Stable Gold Nanoparticles for Hydrocarbon‑Based Nanofluids
- Spherical Gold–Cockle Shell Calcium Carbonate Nanoparticles: Fabrication, Characterization, and Cytotoxicity for Biomedical Use
- Green Production of Copper Nanoparticles: Catalytic, Antibacterial, Cytotoxic, and Antioxidant Potentials
- Green Synthesis of Gold and Silver Nanoparticles with Platycodon Saponins from Platycodi Radix
- Shape-Dependent Cytotoxicity & Cellular Uptake of Green Tea‑Reduced Gold Nanoparticles in Cancer Cells
- Green Synthesis of Gold Nanoparticles with Mimosa tenuiflora Bark Extract: Cytotoxicity, Cellular Uptake and Catalytic Degradation of Methylene Blue
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy



