Horseradish Peroxidase‑Encapsulated Hollow Silica Nanospheres for Intracellular ROS Sensing
Abstract
Reactive oxygen species (ROS) are pivotal in cell signaling, yet their overproduction drives oxidative damage. Precise, live‑cell quantification of ROS is therefore essential for both basic biology and clinical diagnostics. Traditional fluorogenic probes paired with horseradish peroxidase (HRP) suffer from HRP’s poor membrane permeability. Here, we present horseradish peroxidase‑encapsulated hollow silica nanospheres (HRP@HSNs) fabricated via a microemulsion‑templated sol‑gel approach. These nanoreactors retain robust enzymatic activity, readily cross cell membranes, and exhibit excellent biocompatibility. Coupling HRP@HSNs with the dihydrorhodamine 123 (DHR123) probe enabled qualitative and semi‑quantitative detection of intracellular hydrogen peroxide (H₂O₂) in activated RAW 264.7 macrophages. The platform can be readily functionalised with targeting ligands, offering organelle‑ or cell‑type‑specific ROS imaging capabilities.
Background
ROS—including superoxide, hydrogen peroxide, hydroxyl radicals, singlet oxygen, and peroxynitrite—are generated continuously during aerobic metabolism, primarily from the mitochondrial electron transport chain. Antioxidant defenses, both enzymatic (e.g., superoxide dismutase, catalase, peroxidases) and non‑enzymatic (vitamins A, C, E, urate, bilirubin), normally neutralise these species. Imbalance leads to oxidative stress, damaging DNA, lipids, proteins, and contributing to diseases such as diabetes, cancer, cardiovascular and neurodegenerative disorders (Alzheimer’s, Parkinson’s) [1–5].
Recent advances in fluorescent microscopy have enabled non‑invasive, single‑cell ROS imaging, largely through ratiometric or intensity‑based probes that become fluorescent upon oxidation of a non‑fluorescent precursor [6–8]. Specificity is critical; boronate‑based probes, for example, target H₂O₂ via a bioorthogonal oxidation [9]. However, many probes are unstable in vivo, poorly tissue‑penetrant, and rapidly cleared renally [12–14]. Strategies such as stabilising linkers, genetically encoded sensors, bioluminescent reporters, or PET tracers have been explored [15–18]. Nano‑formulations further enhance ROS imaging by improving specificity and sensitivity [19–22].
Enzymes with high catalytic activity are powerful biosensors, yet their free form lacks stability and membrane permeability, limiting intracellular use. Electrode‑based HRP sensors are unsuitable for live‑cell imaging. Encapsulation of HRP within nanoparticle shells can protect the enzyme and facilitate intracellular delivery, as demonstrated with hollow silica nanospheres (HSNs) [25–27]. This study evaluates HRP@HSNs for intracellular ROS detection, assessing loading, activity, cellular uptake, cytotoxicity, and functional imaging in macrophages.
Methods/Experimental
Chemicals and Reagents
Decane, n‑hexanol (98 %), ammonium hydroxide (35 % w/w), TEOS (98 %), APTMS (95 %), FITC, and other reagents were sourced from ACROS and Sigma‑Aldrich. HRP (type VI‑A), DHR123, PMA, and H₂O₂ (35 %) were purchased from Cayman and SHOWA Chemical Industry. All reagents were used as received.
Synthesis of Hollow Silica Nanospheres (HSNs)
HSNs were prepared by a water‑in‑oil microemulsion with selective etching, following protocols in our earlier work [25, 29]. Briefly, 20 mL decane, 1.63 mL CA‑520, and 550 µL n‑hexanol were stirred at 650 rpm, then 350 µL water was added to form the microemulsion. APTMS (25 µL, 200 µL in 1.4 mL ethanol) and TEOS (100 µL) were introduced, followed by 250 µL 35 % ammonia at 20 °C. After 10 h, 95 % ethanol destabilised the emulsion; solid silica nanoparticles (SSNs) were collected by centrifugation (11,000 rpm, 20 min). SSNs were etched in water at 40 °C for 40 min to yield hollow shells, then washed and stored in 99.5 % ethanol.
Synthesis of Horseradish Peroxidase‑Encapsulated Hollow Silica Nanospheres (HRP@HSNs)
The procedure mirrors HSN synthesis, substituting 350 µL water with 350 µL aqueous HRP (10 mg mL⁻¹). HRP@HSNs were stored in deionised water at 4 °C.
Synthesis of FITC‑HSNs and HRP@FITC‑HSNs
To introduce green fluorescence, FITC‑APTMS (10 mg FITC, 200 µL APTMS in 1.4 mL ethanol, 18 h, dark) replaced the ethanolic APTMS in the synthesis, yielding FITC‑HSNs and HRP@FITC‑HSNs.
HRP Entrapment Efficiency and Loading Capacity
HRP was labelled with RITC to create RITC‑HRP, which was encapsulated to form RITC‑HRP@HSNs. After dissolving in 1 M NaOH, fluorescence was measured (Hitachi F‑4500, λ_ex = 543 nm, λ_em = 550–650 nm) and compared to a calibration curve. Entrapment efficiency (%) = (mass of RITC‑HRP in nanospheres / initial RITC‑HRP) × 100; loading capacity (µg HRP mg⁻¹ HSNs) was calculated accordingly.
HRP Activity Assay
HRP activity was quantified using the chromogenic substrate TMB. Enzyme solutions (native HRP and HRP@HSN) were mixed with 20 µM TMB and 20 µM H₂O₂ in phosphate/citrate buffer (pH 5.2). Absorbance at 655 nm was recorded with a BioTek Synergy reader; activity was derived from the native HRP calibration curve.
Reactivity Assay with Various ROS
DHR123 (20 µM) with or without HRP@HSNs (50 µg mL⁻¹) was incubated with 100 µM of ROS (H₂O₂, TBHP, superoxide, •OH, •OtBu) in DMEM (pH 7.4). Fluorescence (λ_ex = 488 nm, λ_em = 530 nm) was measured every 5 min over 120 min.
Cell Culture and Viability Assay
RAW264.7 macrophages were cultured in DMEM + 10 % FBS + antibiotics at 37 °C, 5 % CO₂. For viability, 2 × 10⁵ cells/well were seeded in 24‑well plates; after 24 h, cells were exposed to 0–200 µg mL⁻¹ of nanoparticles in serum‑free DMEM for 2 h. WST‑1 reagent (Clontech) was used to assess viability (450 nm, reference 650 nm) with a Bio‑Rad 680 reader.
Cell Uptake Analysis
1 × 10⁶ RAW264.7 cells/well were incubated with 0–200 µg mL⁻¹ of FITC‑labelled nanoparticles for 2 h. After washing and trypsinisation, uptake was quantified by flow cytometry; trypan blue quenched extracellular fluorescence.
Flow Cytometry of ROS Production in PMA‑Stimulated Macrophages
Following 2 h nanoparticle exposure and 30 min DHR123 loading, cells were stimulated with 1 µg mL⁻¹ PMA for 1 h before analysis on a FACS Canto II.
Quantitative Analysis
96‑well plates were seeded with 3 × 10⁴ RAW264.7 cells/well. After 2 h nanoparticle treatment and 1 h PMA (varied) + DHR123 (20 µM) incubation, fluorescence (λ_ex = 488 nm, λ_em = 530 nm) was measured. A calibration curve (H₂O₂ + HRP@HSN) allowed conversion of fluorescence to H₂O₂ concentration.
Characterization
TEM (JEOL JEM‑1200 EX II, 100 kV) confirmed 45 nm hollow cores; uranyl acetate staining verified internal HRP. Dynamic light scattering (Malvern Zetasizer Nano ZS) assessed hydrodynamic size and zeta potential; cells were imaged with a Zeiss Axio Observer Z1.
Results and Discussion
Design and Synthesis of HSNs and HRP@HSNs
HRP@HSNs were produced via ammonia‑catalysed sol‑gel chemistry within a w/o microemulsion (Scheme 1). TEM (Fig. 1) shows monodisperse 45 nm spheres; uranyl acetate staining confirms HRP encapsulation inside the hollow cavity. DLS reveals hydrodynamic diameters of 188 ± 4 nm (HSNs) and 184 ± 6 nm (HRP@HSNs) in water, which increase to 1.8–1.7 µm in serum‑free DMEM due to aggregation. Bovine serum albumin (BSA) reduces this aggregation, yielding stable 195–197 nm particles.
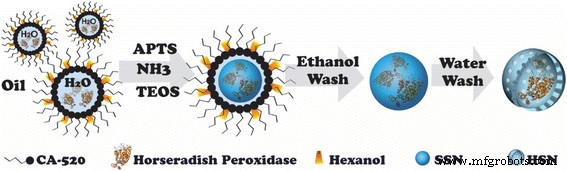
Flow chart of the synthesis of horseradish peroxidase‑encapsulated hollow silica nanospheres (HRP@HSNs). APTMS, 3‑aminopropyltrimethoxysilane; TEOS, tetraethyl orthosilicate; SSN, solid silica nanoparticle
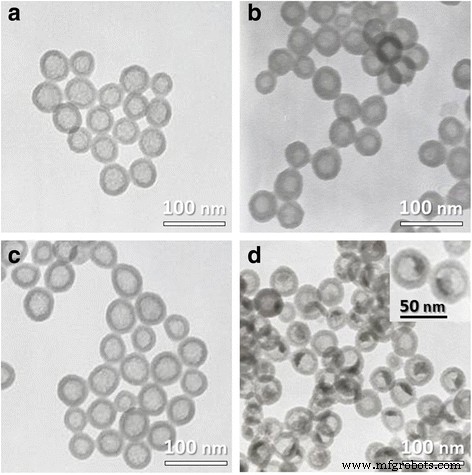
TEM images of (a) HSNs, (b) uranyl‑acetate‑stained HSNs, (c) HRP@HSNs, (d) uranyl‑acetate‑stained HRP@HSNs. Inset: enlarged view
HRP Entrapment Efficiency and Loading Capacity
RITC‑HRP encapsulation yielded ~6 % entrapment efficiency across 11–33 nmol loading; the highest loading capacity reached 12.5 ± 1.2 µg HRP mg⁻¹ HSN at 33.3 nmol input. While the efficiency is modest, it demonstrates that HRP can be retained within the nanocavity, and the loading can be tuned by enzyme concentration.
Cytotoxicity and Cellular Uptake
WST‑1 assays show no significant reduction in RAW264.7 proliferation after 2 h or 26 h exposure to up to 200 µg mL⁻¹ HSNs or HRP@HSNs, indicating low cytotoxicity. FITC‑labelled particles achieved >80 % labeling efficiency at >50 µg mL⁻¹ after 2 h, confirming efficient intracellular delivery without compromising cell viability.
Reactivity of HRP@HSNs to Various ROS
HRP activity assay shows ~40 % retention of native activity after encapsulation, likely due to diffusion limitations through the silica shell. In cell‑free assays, DHR123 alone is unresponsive to H₂O₂ or superoxide but reacts strongly with •OH and •OtBu. HRP or HRP@HSNs markedly amplify fluorescence for all tested ROS, with a higher signal from native HRP correlating with its greater activity. Relative fluorescence ratios at 60 min reveal that both HRP variants enhance detection of H₂O₂ and superoxide, confirming that the nanocavity preserves catalytic function while permitting substrate diffusion.
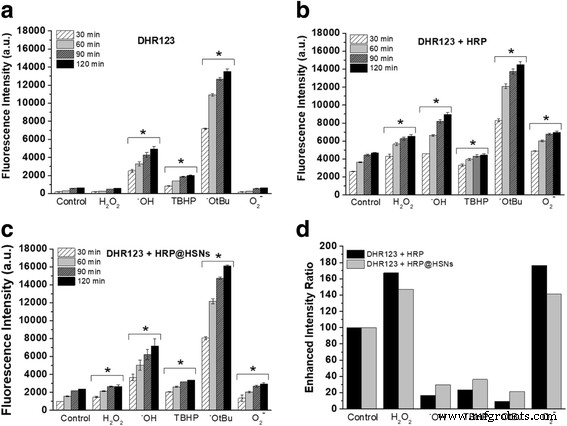
(a–c) Time‑dependent fluorescence of DHR123 alone (a), with HRP (b), or with HRP@HSNs (c). (d) Enhanced intensity ratio at 1 h for each ROS (20 µM DHR123, 400 ng mL⁻¹ HRP, 50 µg mL⁻¹ HRP@HSNs, 100 µM ROS). *p < 0.05 versus control.
Intracellular ROS Detection with HRP@HSNs
RAW264.7 cells pre‑treated with HRP@HSNs (2 h) and loaded with DHR123 (30 min) displayed markedly higher fluorescence after PMA stimulation compared to controls. Flow cytometry shows a >2‑fold increase in R123 fluorescence in stimulated cells; HRP@HSNs deliver additional catalytic activity, whereas plain HSNs produce a modest fluorescence rise, likely reflecting cellular stress responses. Fluorescence intensity increased dose‑dependently with both PMA (0.1–2 µg mL⁻¹) and HRP@HSNs (50–200 µg mL⁻¹). Representative confocal images confirm enhanced intracellular ROS imaging (Fig. 3d).
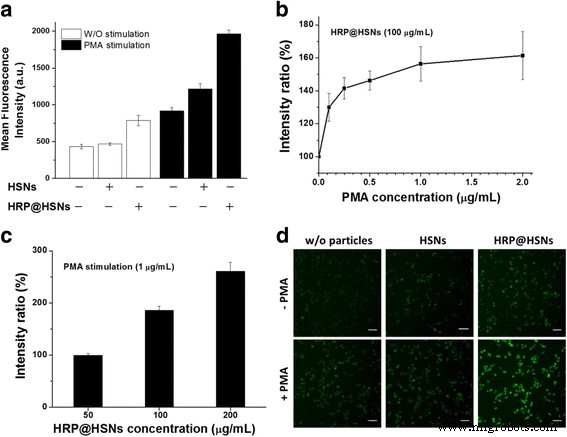
(a) Flow cytometry of RAW264.7 cells with/without PMA and nanoparticles. (b) PMA concentration‑dependent fluorescence. (c) HRP@HSN concentration‑dependent fluorescence. (d) Fluorescence images (scale 50 µm).
Application of HRP@HSNs for Quantitative H₂O₂ Analysis
Using an external H₂O₂ calibration curve (0.625–15 µM), intracellular H₂O₂ generated by PMA‑stimulated RAW264.7 cells was quantified. Cells treated with 100 µg mL⁻¹ HRP@HSNs and 20 µM DHR123, then exposed to 0.1–2 µg mL⁻¹ PMA, yielded endogenous H₂O₂ concentrations of ~10 µM at ≥0.25 µg mL⁻¹ PMA. This semi‑quantitative measurement demonstrates the platform’s utility for assessing oxidative stress in live cells.
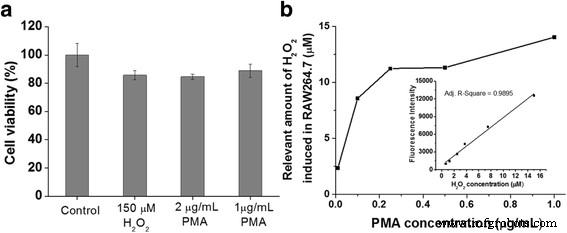
(a) WST‑1 viability of RAW264.7 after H₂O₂ or PMA treatment (1 h). (b) Endogenous H₂O₂ quantification using HRP@HSNs and DHR123. Inset: calibration curve.
Conclusions
We have engineered hollow silica nanospheres that encapsulate active HRP, creating robust intracellular ROS sensors. The porous silica shell allows small substrates to reach the entrapped enzyme while protecting HRP from proteolysis. Coupled with DHR123, HRP@HSNs provide enhanced, semi‑quantitative detection of endogenous H₂O₂ in macrophages under oxidative stress. Surface functionalisation with antibodies or peptides will enable organelle‑ or tumour‑cell‑specific targeting, and alternative payloads (e.g., nanocatalysts or boronate probes) can broaden the sensing repertoire. These nanoreactors represent a versatile platform for simultaneous ROS imaging, quantification, and targeted therapeutic delivery in biomedical research and clinical diagnostics.
Abbreviations
- APTMS
3‑Aminopropyltrimethoxysilane
- BSA
Bovine serum albumin
- DHR123
Dihydrorhodamine 123
- DLS
Dynamic light scattering
- FITC
Fluorescein isothiocyanate
- HRP
Horseradish peroxidase
- HSNs
Hollow silica nanospheres
- Igepal CA‑520
Polyoxyethylene (5) isooctylphenyl ether
- mETC
Mitochondrial electron transport chain
- PET
Positron emission tomography
- PMA
Phorbol 12‑myristate 13‑acetate
- R123
Rhodamine 123
- RITC
Rhodamine B isothiocyanate
- ROS
Reactive oxygen species
- SSN
Solid silica nanoparticle
- TBHP
Tert‑butyl hydroperoxide
- TEM
Transmission electron microscopy
- TEOS
Tetraethyl orthosilicate
- TMB
3,3′5,5′‑Tetramethylbenzidine
Nanomaterials
- Harnessing Reactive Oxygen Species Toxicity for Advanced Tumor Therapy: Nanoparticle Strategies and Clinical Outlook
- Engineering Monodisperse GFP‑Doped Silica Nanoparticles for Efficient Intracellular Protein Delivery and Bioimaging
- Highly Sensitive Non‑Enzymatic Glucose Sensor Based on Mesoporous NiO Nanopetals Grown on FTO
- Enhanced Reactive Oxygen Species Generation by GdVO4:Eu³⁺ Nanoparticles and Methylene Blue Complexes under UV–Vis and X‑ray Irradiation
- Synergistic Antioxidant and Anti‑Amyloid Action of Chlorogenic Acid–Selenium Nanoparticles in Vitro
- Eco‑Friendly Starch‑Paper Triboelectric Nanogenerator for Real‑Time Human Sweat Sensing
- ROS‑Responsive Piperlongumine‑Eluting GI Stent: A Novel Nanofiber Strategy to Suppress Cholangiocarcinoma
- Rh-Doped MoTe₂ Monolayer: A Powerful DFT-Validated Sensor and Scavenger for SF₆ Decomposition Products
- Chitosan‑Capped, Enzyme‑Responsive Hollow Mesoporous Silica Nanoplatforms for Targeted Colon Drug Delivery
- Nanoparticle Toxicity in Biomedicine: Reactive Oxygen Species Impact



