Rapid, Low‑Cost Microfluidic Electrochemical Sensor Fabricated Directly on Screen‑Printed Electrodes for Ultra‑Sensitive PSA Detection
Abstract
We present the first demonstration of a microfluidic platform fabricated directly onto a commercially available screen‑printed electrode (SPE) using a simple, rapid, and inexpensive process. The device—termed CASPE‑MFD (Commercially Available SPE‑Based Microfluidic Device)—combines polydimethylsiloxane (PDMS) microchannels with a glass‑coated SPE substrate. In human serum, the CASPE‑MFD achieves a limit of detection (LOD) of 0.84 pg/mL (25.8 fM) for prostate‑specific antigen (PSA) and displays excellent linearity across a 0.001–10 ng/mL range. This platform offers a scalable, low‑cost solution for point‑of‑care diagnostics, environmental monitoring, and food safety analysis.
Background
Microfluidic systems manipulate volumes from nanoliters to picoliters within micrometer‑scale channels, enabling high‑throughput, reagent‑efficient analyses. Their integration with electrochemical sensors confers remarkable sensitivity, rapid response, and minimal instrumentation requirements, making them ideal for on‑site biomedical diagnostics.
Electrochemical readouts are inherently compatible with microfluidics, eliminating bulky optics and simplifying fabrication. The synergy of microfluidics and SPEs—already widely used in clinical point‑of‑care devices—promises a fully integrated, disposable sensor platform.
In this work, we fabricate CASPE‑MFDs by bonding PDMS microchannels onto a glass‑coated SPE via oxygen plasma activation. The device is tested for PSA quantification in phosphate buffer and human serum using chronoamperometry (CA) and square‑wave voltammetry (SWV).
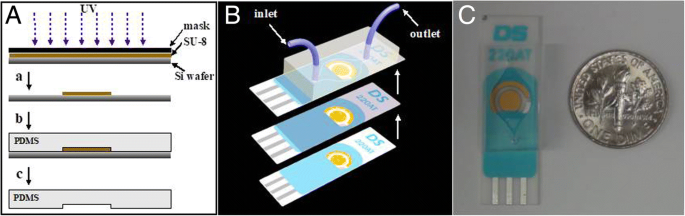
a Fabrication of PDMS microchannels via SU‑8 photolithography.b Assembly of the CASPE‑MFD: PDMS channels bonded to a glass‑coated SPE containing two gold working/counter electrodes and a silver pseudo‑reference electrode.c Photograph of the completed CASPE‑MFD.
Materials and Methods
Reagents and Materials
PSA, HRP‑conjugated anti‑PSA, biotinylated anti‑PSA, streptavidin magnetic beads, BSA, hydroquinone, Tween‑20, H2O2, and ferrocenecarboxylic acid were sourced from commercial suppliers (Petsec Energy Ltd., Fisher Scientific, Sigma‑Aldrich). PDMS pre‑polymer and curing agent were obtained from Dow Corning. All buffers were prepared with ultrapure water (Millipore Milli‑Q).
Instrumentation
Fluorescence imaging: Olympus U‑CMAD3. Plasma cleaning: Harrick PDC‑32G. Electrochemical measurements: CHI 760B potentiostat (three‑electrode setup: gold working/counter, silver pseudo‑reference).
Microfluidic Chip Fabrication
PDMS channels were patterned on a silicon wafer coated with SU‑8 2075, exposed to UV light, and cured. The SPE substrate was coated with a thin glass layer via a sol‑gel mixture (TEOS:MTES:ethanol:water = 1:1:1:1) and cured at 65 °C overnight. Both PDMS and SPE surfaces were activated by 30 s O2 plasma before alignment and bonding, forming a hermetic seal.
Chronoamperometric Experiments
Measurements were performed in 1× pH 7.4 PBS containing 4.5 mM hydroquinone and 0.1 mM H2O2. The CASPE‑MFD was first activated by cycling between 0.5–1.5 V in 0.5 M H2SO4. Then, 50 µL of magnetic‑bead‑conjugated anti‑PSA antibody was introduced at 50 µL min−1, followed by blocking, PSA incubation, HRP‑anti‑PSA addition, and finally the electroactive solution. Chronoamperometry was carried out at –2.0 mV versus the silver pseudo‑reference, recording the steady‑state current after 30 s.
Results and Discussion
Device Characterization
Fluorescent microbeads injected into the CASPE‑MFD filled the channels uniformly without bubbles, confirming robust fluidic performance at flow rates up to 100 µL min−1 (Figure 2).

a Fluorescent microbead infiltration on the SPE. b Bright‑field image of the filled CASPE‑MFD. c Enlarged view of the fluorescence distribution.
Cyclic voltammetry with 0.5 mM ferrocenecarboxylic acid confirmed reversible electron transfer (Epa–Epc = 62 mV) and diffusion‑controlled kinetics (ipa ∝ √v). The currents in the CASPE‑MFD matched those in bulk solution, indicating that the microfluidic architecture does not compromise sensitivity (Figure 3).
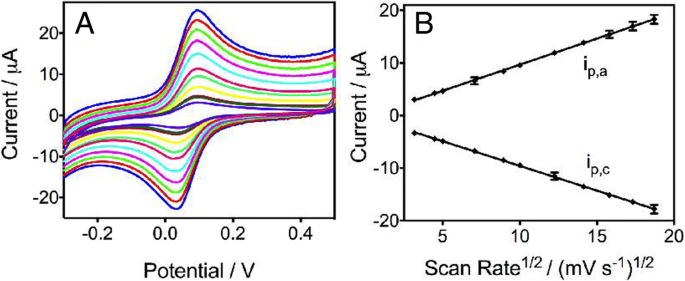
a CVs at scan rates 10–350 mV s−1. b Calibration plots of ipa and ipc versus √scan rate.
PSA Detection Performance
Chronoamperometric curves (Figure 5a) show a monotonic increase in current with PSA concentration (0–10 ng mL−1). The linear range (0.001–10 ng mL−1) yields a regression of I (µA) = 14.87 + 3.927 × log CPSA (R2 = 0.9985). The LOD of 0.84 pg mL−1 surpasses commercial assays (0.1 ng mL−1) by >100‑fold and outperforms comparable microfluidic sensors.
Square‑wave voltammetry (SWV) produced parallel results, confirming the robustness of the electrochemical readout (Figure 5c).
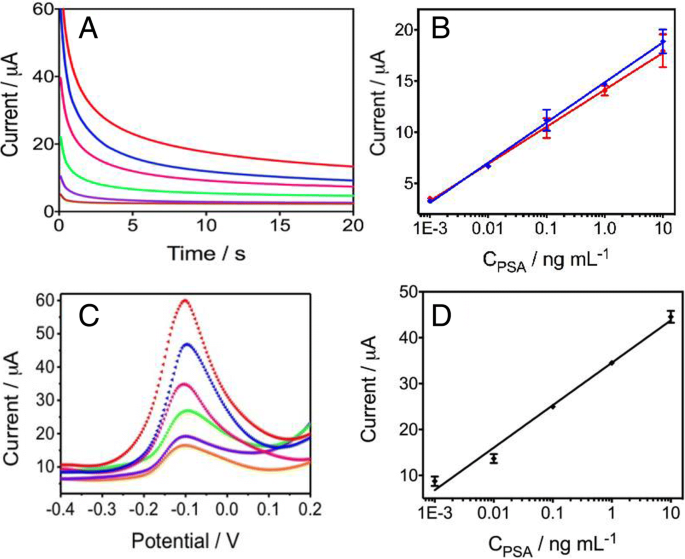
a Chronoamperometry for 0–10 ng mL−1. b Linear plots in buffer (blue) and human serum (red). c SWV curves. d Linear fit for SWV (R2 = 0.9884).
Selectivity and Real‑Sample Analysis
In human serum, the CASPE‑MFD maintained the same linearity and LOD, demonstrating compatibility with complex biological matrices. No significant interference was observed from potential matrix components, underscoring the assay’s selectivity.
Conclusions
The CASPE‑MFD combines low‑cost fabrication, microfluidic control, and SPE electrochemistry to deliver ultra‑sensitive PSA detection (<0.85 pg mL−1) in a portable format. Its ease of assembly, reproducibility, and potential for multiplexing make it a promising platform for point‑of‑care diagnostics of cancer biomarkers, electrolytes, and nucleic acids.
Abbreviations
- MFDs
Microfluidic devices
- CASPE-MFDs
Screen‑printed electrode‑based microfluidic devices
- PDMS
Polydimethylsiloxane
- PSA
Prostate‑specific antigen
- CA
Chronoamperometry
- SWV
Square wave voltammetry
- LOD
Limit of detection
- HRP
Horseradish peroxidase
- TEOS
Tetra‑ethoxy silane
- MTES
Metyl‑triethoxysilane
- BNP
B‑type natriuretic peptide
Nanomaterials
- Ultra‑Sensitive Electrochemical DNA Biosensor for Vibrio cholerae Detection Using Polystyrene‑Acrylic Acid Nanoparticles and Gold Nanoparticles
- Highly Sensitive Non‑Enzymatic Glucose Sensor Based on Mesoporous NiO Nanopetals Grown on FTO
- A Molecularly Imprinted CdSe@SiO₂ Core–Shell Nanohybrid: A Ratiometric Fluorescent Probe for Ultra‑Sensitive 4‑Nitrophenol Detection
- Ultra-Stable Pt–Ni Jagged Nanowire Sensor for Highly Sensitive Caffeic Acid Detection
- Electrochemical Deposition of Amorphous Co‑Ni Hydroxide for Highly Sensitive Non‑Enzymatic Glucose Sensing
- Carbon Dots: Advanced Fluorescent Platforms for Sensitive Metal‑Ion Detection
- Rapid Electrochemical Sensor for Polychlorinated Biphenyls Using β‑Cyclodextrin/Tin Disulfide‑Modified Screen‑Printed Carbon Electrodes
- Ultrasensitive Room‑Temperature Formaldehyde Sensor Using Ag‑Doped In₂O₃ Nanograins
- Portable Plasmonic Thermal Device for Accurate Quantification of Lateral Flow Assays
- Graphene‑Based Ultrasensitive Biosensor Enhances Protein Disease Detection



