Electrochemical Deposition of Amorphous Co‑Ni Hydroxide for Highly Sensitive Non‑Enzymatic Glucose Sensing
Abstract
Among transition‑metal hydroxides, cobalt‑ and nickel‑based compounds are celebrated for their exceptional electrochemical performance, especially in non‑enzymatic sensing. Binary Co‑Ni hydroxides combine the redox versatility of both metals, yielding outstanding activity for glucose detection. We present a single‑step, chemically clean electrochemical deposition that produces three‑dimensional amorphous Co‑Ni hydroxide nanostructures with a homogeneous 1:1 Co:Ni ratio. The amorphous phase offers a high specific surface area, efficient electron transfer, and abundant Ni^2+/Ni^3+ and Co^2+/Co^3+/Co^4+ redox couples, which together enable superior glucose sensing. In 0.5 M NaOH the sensor delivers a sensitivity of 1911.5 µA mM^−1 cm^−2 at low concentrations, a linear range from 0.00025 to 5 mM, a detection limit of 0.127 µM, long‑term stability, and excellent selectivity against common interferents. These attributes establish amorphous Co‑Ni hydroxide as a promising material for non‑enzymatic glucose monitoring.
Introduction
Carbohydrates are vital energy sources, and monitoring blood glucose is essential for diagnosing and managing diabetes mellitus—a global epidemic that affects millions worldwide. Accurate, rapid, and continuous glucose monitoring demands sensors that are highly sensitive, selective, cost‑effective, and stable. Electrochemical glucose sensors dominate the field because of their simplicity, low cost, portability, and excellent accuracy.
Glucose sensors are traditionally classified into enzyme‑based and non‑enzymatic types. Enzyme‑based sensors suffer from enzyme denaturation, high cost, and limited stability, driving research toward non‑enzymatic alternatives that rely on transition‑metal catalysts to oxidize glucose directly. Noble‑metal catalysts are expensive; therefore, non‑noble metals such as Ni and Co have attracted attention for their abundance and catalytic activity.
Single‑metal or single‑oxide materials often exhibit limited performance due to a narrow set of redox couples. Bimetallic systems, especially Co‑Ni, enhance electrocatalytic activity by combining complementary redox behavior and creating synergistic effects. Although most bimetallic hydroxides are crystalline, recent studies suggest that amorphous phases can offer higher surface area, more defect sites, and improved charge transfer, potentially surpassing crystalline counterparts in sensing performance.
In this work, we develop a facile electrochemical deposition method to fabricate amorphous Co‑Ni hydroxide nanostructures and evaluate their performance as a non‑enzymatic glucose sensor in alkaline media.
Methods
Synthesis of Amorphous Co‑Ni Hydroxide Nanostructures on Graphite
The nanostructures were prepared in a single step by electrochemical cathodic deposition. A graphite sheet (15 mm × 7 mm) served as the working electrode while a second identical sheet acted as the counter. The deposition bath comprised de‑ionized water and a Ni/Co alloy target (1:1 molar ratio) placed centrally. A constant potential of 90 V was applied for 12 h, yielding uniform Co‑Ni hydroxide coverage.
Characterization of the Amorphous Co‑Ni Hydroxide
Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) (JSM‑7600F and FEI Tecnai G2 F30) assessed morphology and crystallinity. Energy‑filtered TEM mapped elemental distribution. X‑ray photoelectron spectroscopy (XPS) and Raman spectroscopy (Renishaw inVia, 785 nm) examined surface chemistry and bonding states.
Electrochemical Measurements
All measurements used a CHI‑760E workstation in a conventional three‑electrode cell: 0.5 M NaOH as electrolyte, Co‑Ni hydroxide/graphite as working, saturated calomel electrode (SCE) as reference, and platinum wire as counter. Cyclic voltammetry (CV) and amperometry were performed to evaluate glucose sensing performance.
Results and Discussions
Morphology and Structure of the Amorphous Co‑Ni Hydroxide
SEM images (Fig. 1a,b) reveal nanostructures with an average diameter of ~400 nm and a wrinkled, three‑dimensional surface. TEM (Fig. 1c,d) confirms the absence of lattice fringes, indicating an amorphous phase; the corresponding SAED pattern shows a broad halo ring. Elemental mapping (Fig. 1f‑h) demonstrates uniform distribution of O, Co, and Ni, confirming a homogeneous 1:1 Co:Ni composition.

Morphology and structure of the products. a, b SEM images of the amorphous Co‑Ni hydroxide deposited on a graphite sheet. c, d TEM images of the sample (insets show SAED and HRTEM). e STEM image. f–h Element mappings of O, Co, and Ni.
XPS analysis (Fig. 2a‑c) confirms Ni and Co in the +2 oxidation state, consistent with Ni(OH)_2 and Co(OH)_2. The O 1s peak at 531 eV indicates metal‑hydroxyl bonding. Raman spectroscopy (Fig. 2d) displays characteristic bands of Co(OH)_2 and Ni(OH)_2, further supporting the amorphous Co‑Ni hydroxide composition.
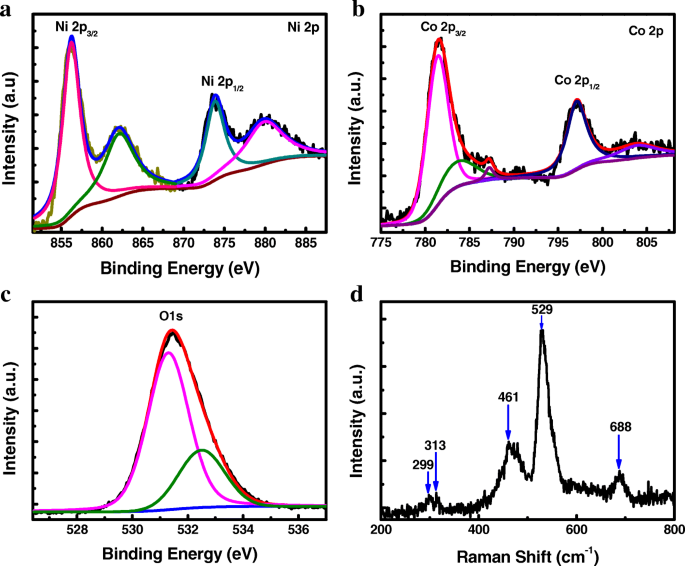
Chemical states of bonded elements. a–c XPS spectra of Ni 2p, Co 2p, and O 1s. d Raman spectrum of the amorphous Co‑Ni hydroxide.
Electrochemical Behavior Toward Glucose Oxidation
CV in 0.5 M NaOH (Fig. 3a) shows pseudocapacitive behavior with well‑defined redox peaks attributable to Ni^2+/Ni^3+ and Co^2+/Co^3+/Co^4+ transitions. The anodic peak at ~0.36 V vs SCE corresponds to the oxidation of Ni^2+ and Co^2+ to higher states. Scan‑rate dependence confirms a diffusion‑controlled process (I_pa ∝ ν^1/2, R^2 = 0.999).
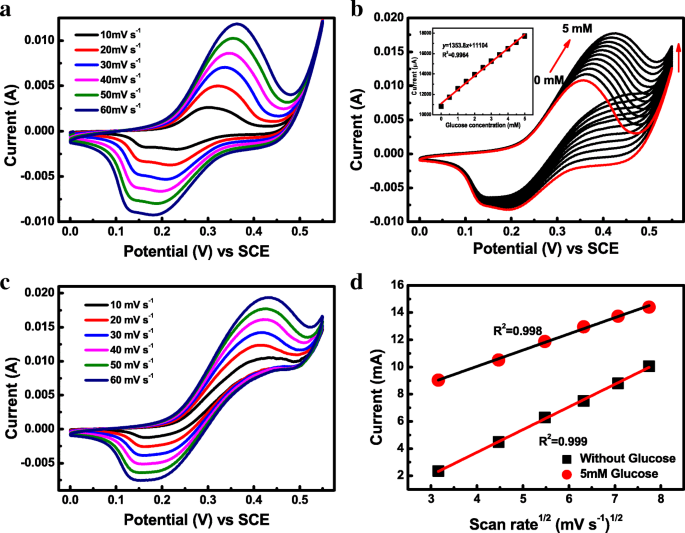
Electrochemical behavior of the amorphous Co‑Ni hydroxide toward glucose oxidation in 0.5 M NaOH. a CV curves at various scan rates without glucose. b CV curves with different glucose concentrations (0–5 mM) at 50 mV s^−1. c CV curves at scan rates 10–60 mV s^−1 (5 mM glucose). d I_pa versus ν^1/2 plots (absence and presence of glucose).
Glucose addition shifts the anodic peak positively and increases I_pa, indicating glucose oxidation via Ni^3+ and Co^4+ mediated pathways (Eqs. 2–6). The anodic peak current exhibits a linear relationship with glucose concentration from 0.00025 to 5 mM, with a sensitivity of 1911.5 µA mM^−1 cm^−2 in the low‑concentration range (Fig. 4b). The detection limit, calculated as 3σ/S, is 0.12 µM.
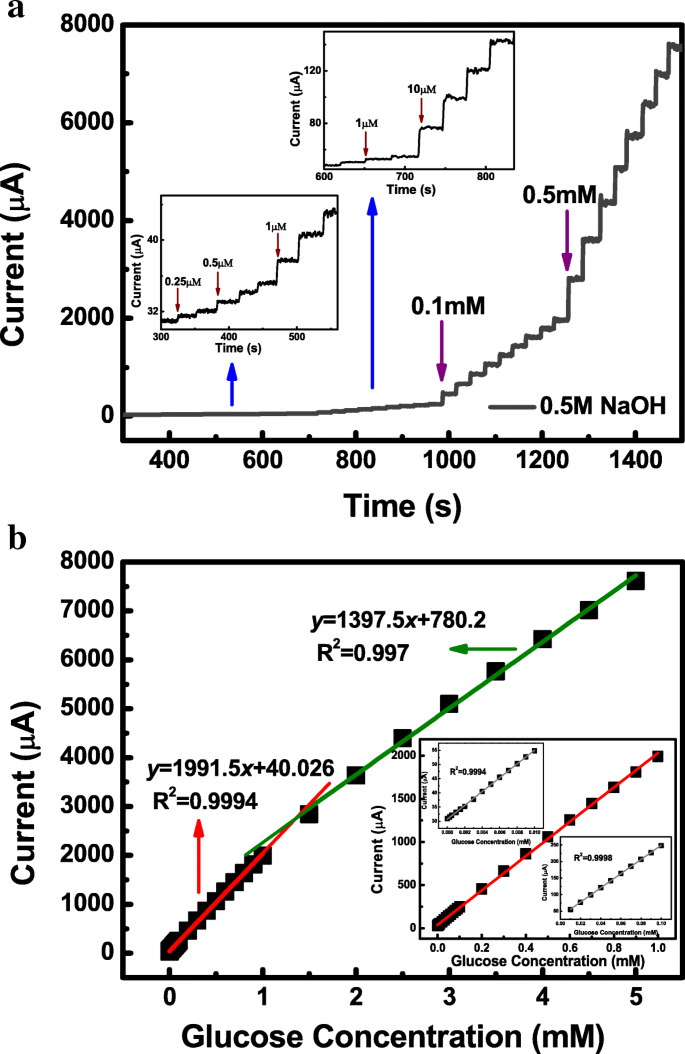
Amperometric detection of glucose. a i‑t curve (0.36 V vs SCE). b Calibration plot showing two linear regimes: 0.00025–1 mM (R^2 = 0.9994) and 1–5 mM (R^2 = 0.997).
Effect of NaOH Concentration and Stability
Amperometric response was tested in 0.1, 0.5, and 1 M NaOH. The 0.5 M solution yields the highest sensitivity, lowest LOD, and widest linear range (Fig. 5b), making it the optimal electrolyte for glucose detection.
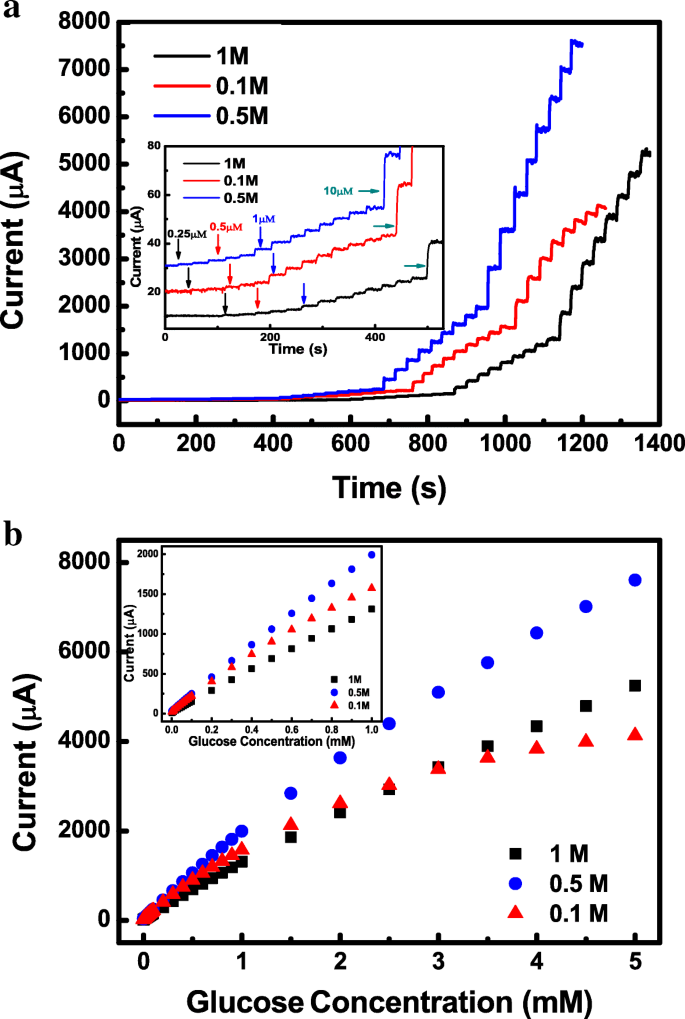
Current response to glucose at different NaOH concentrations. a i‑t curves for 0.1, 0.5, and 1 M NaOH. b Current–glucose concentration plots.
Long‑term stability was evaluated over 2 months; the sensor retains 103 % of its initial response, demonstrating exceptional durability (Fig. 6a). Comparative analysis (Table 1) shows that the amorphous Co‑Ni hydroxide outperforms many reported Ni‑ or Co‑based non‑enzymatic sensors in sensitivity, linear range, and stability.
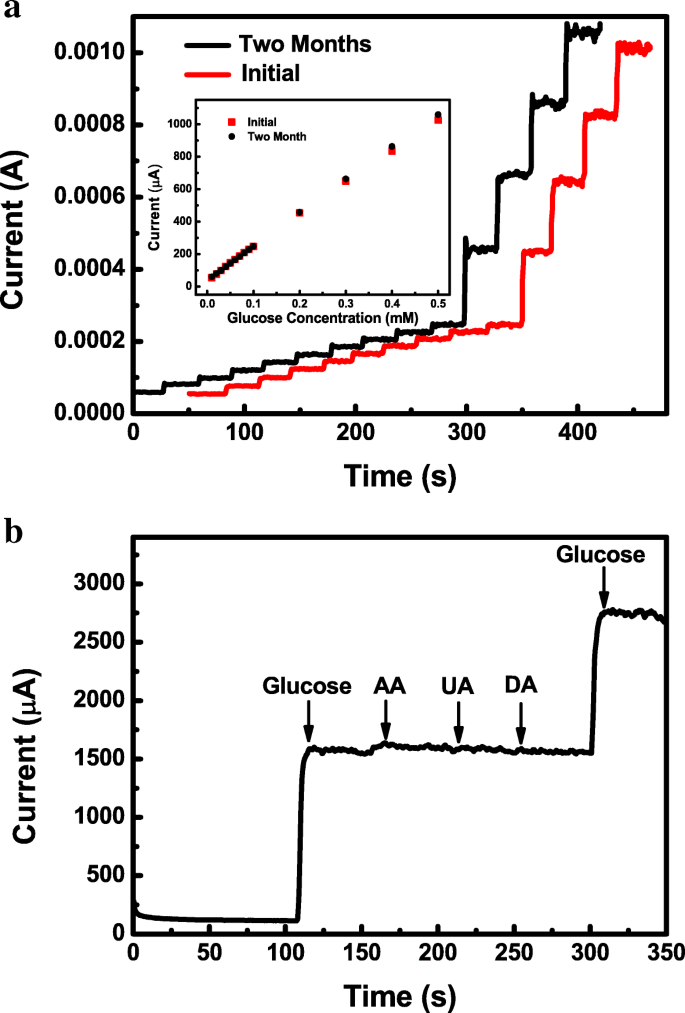
Stability and selectivity of the amorphous electrode. a i‑t curve over 2 months in 0.5 M NaOH. b Selectivity test against 1 mM glucose and 0.1 mM AA, UA, DA.
Interference studies confirm negligible response to ascorbic acid, uric acid, and dopamine, underscoring the sensor’s high selectivity for glucose in physiological conditions.
Real‑Sample Analysis
Glucose concentrations in human serum were measured using the amperometric method. Recoveries ranged from 97.92 % to 100.33 % with a relative standard deviation of 2.66–3.99 % (Table 2), validating the sensor’s practical applicability.
Conclusions
We demonstrate a facile, clean electrochemical deposition strategy to synthesize amorphous Co‑Ni hydroxide nanostructures with a uniform 1:1 Co:Ni ratio. The resulting material exhibits exceptional non‑enzymatic glucose sensing performance in 0.5 M NaOH: a sensitivity of 1911.5 µA mM^−1 cm^−2, a detection limit of 0.12 µM, a linear range of 0.00025–5 mM, a 5‑s response time, and outstanding long‑term stability. These findings highlight amorphous Co‑Ni hydroxide as a promising platform for next‑generation glucose sensors.
Abbreviations
- 3D:
Three‑dimensional
- AA:
Ascorbic acid
- CV:
Cyclic voltammetry
- DA:
Dopamine
- Epa:
Potential of anodic peak
- Epc:
Potential of cathodic peak
- HRTEM:
High‑resolution transmission electron microscopy
- Ipa:
Anodic peak current
- RSD:
Relative standard deviation
- SAED:
Selected area electron diffraction
- SCE:
Saturated calomel electrode
- SEM:
Scanning electron microscopy
- TEM:
Transmission electron microscopy
- UA:
Uric acid
- XPS:
X‑ray photoelectron spectroscopy
Nanomaterials
- Smart Lock & Sensor Design: Expert Circuit Protection & Sensing Strategies for Secure Smart Homes
- Gold Nanoparticles as Advanced Chemosensors: Enhancing Electrochemical Detection
- Nanodiamonds: Ultra‑Precise Magnetic Sensors for Biomedical and Quantum Applications
- Highly Sensitive Non‑Enzymatic Glucose Sensor Based on Mesoporous NiO Nanopetals Grown on FTO
- Optimized α‑NiS Nanosphere Films for Long‑Term, Non‑Enzymatic Glucose Sensing
- Eco‑Friendly Starch‑Paper Triboelectric Nanogenerator for Real‑Time Human Sweat Sensing
- Rapid, Low‑Cost Microfluidic Electrochemical Sensor Fabricated Directly on Screen‑Printed Electrodes for Ultra‑Sensitive PSA Detection
- Ultra‑Sensitive Room‑Temperature CNTs/SnO₂/CuO Sensor for Rapid H₂S Detection
- Advanced Dual Electro‑Optical Sensing with Layer‑by‑Layer Electroactive Dye/LDH Nanoplatelet Films
- Efficient Synthesis of Gold Nanoplates with Ortho‑Carbonyl Capping Agents for Sensitive Lead Ion Electrochemical Detection



