Frequency‑Modulated Dielectrophoresis of Vesicles and Cells: Predictable U‑Turn Trajectories at the Crossover Frequency
Abstract
We have derived a comprehensive model for the dielectrophoretic (DEP) force exerted on micro‑ and nanoscopic particles when exposed to frequency‑modulated (FM) electric fields. By tuning the FM wave’s frequency band to encompass the crossover frequency fX of the real part of the Clausius–Mossotti factor, our theory predicts a systematic reversal of the DEP force each time the instantaneous frequency crosses fX. Experimentally, we observed periodic U‑turns in multilamellar vesicles (MLVs), human T‑cell leukemia (TL) cells, B‑cell leukemia (BL) cells, and red blood (RB) cells under FM‑DEP. Video tracking of these U‑turns provides a fast, <30‑second, and highly accurate method to determine fX, useful for both saline suspensions and nanoparticle suspensions where electric‑field‑induced solvent flow is minimized. The accuracy of fX was confirmed through two independent experiments: (1) measuring the DEP force on a single vesicle under alternating‑current (AC) DEP at various sinusoidal frequencies and comparing the resulting fX with that obtained from FM‑DEP; and (2) assessing how fX varies with the conductivity of the surrounding medium for three cell types. The second approach allowed simultaneous estimation of cytoplasmic conductivities and membrane capacitances using a refined single‑shell model. Cytoplasmic conductivities were slightly lower than previously reported ranges, while membrane capacitances matched literature values closely.
Background
Cellular polarizability is dominated by the membrane and cytoplasmic electrical properties, which vary with the applied field frequency. This frequency‑dependent dielectric spectrum enables label‑free discrimination of cell phenotypes via impedance spectroscopy, AC DEP, traveling‑wave DEP (twDEP), and electrorotation [1–16]. Compared with antibody‑based or adhesive labeling methods, these electrical techniques avoid cell modification and potential activation, making them attractive for noninvasive cell sorting.
DEP arises in a nonuniform electric field, generating a force proportional to the gradient of the field amplitude and the real part of the Clausius–Mossotti (CM) factor. The phase gradient gives rise to twDEP or electrorotation, which provide complementary dielectric information. Our work extends the conventional AC DEP by introducing FM electric fields, allowing continuous traversal of the crossover frequency fX and enabling rapid dielectric characterization.
In typical AC DEP setups, the field is applied between fixed microfabricated electrodes, limiting spatial flexibility. Electronic tweezers—where micromanipulated electrode needles deliver the field—offer a versatile, contact‑free alternative. However, dielectric measurements require short exposure times (<30 s) to reduce solvent flow effects, which is challenging with multi‑electrode systems. FM‑DEP addresses this by modulating the frequency while keeping the electrode geometry constant.
Our plug‑in system (Fig. 1) employs two tungsten needle electrodes, each 0.5 µm tip diameter, positioned 100 µm apart. The needles are driven by an arbitrary waveform generator and current amplifier, and the entire assembly is mounted on a patch‑clamp micromanipulator beneath an inverted microscope. This configuration permits precise, noncontact manipulation of individual vesicles or cells above the substrate.
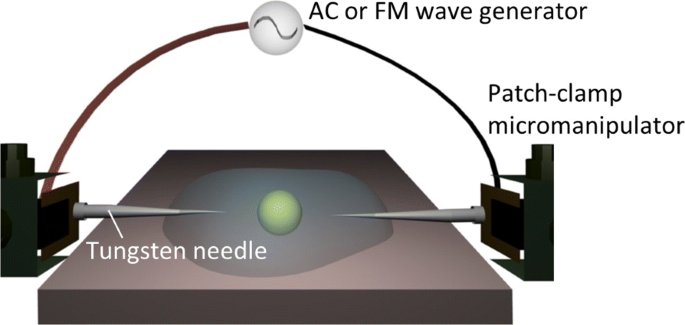
Experimental setup. A schematic of the dielectrophoretic manipulation system illustrating the AC or FM electric field applied to a target particle via a pair of electrode needles controlled by patch‑clamp micromanipulators.
The FM wave is defined by
𝐸(𝐫,t)=𝐴(𝐫)cosθ(t),
with the phase related to the instantaneous frequency f(t) by θ̇=2πf(t). We use a wide‑band FM modulation:
f(t)=fc+Δf cos(2πfmt),
where the modulation frequency fm is much smaller than the carrier frequency range, satisfying the wide‑band limit (WBL) condition Δf/fm≫1.
Methods
Materials
MLVs were prepared from 1,2‑dioleoyl‑sn‑glycero‑3‑phosphatidylcholine (DOPC; Avanti Polar Lipids). A 1 mL DOPC solution (20 mM) in chloroform/methanol (2:1 v/v) was dried under nitrogen and vacuum‑sealed for >12 h. The thin film was rehydrated with deionized water and incubated at 25 °C for several hours.
Human T‑cell leukemia (JKT‑β‑del) and B‑cell leukemia (CCRF‑SB) cell lines were cultured in RPMI 1640 supplemented with 10 % fetal bovine serum and 100 mM sodium pyruvate at 37 °C, 5 % CO₂. After 1 week, cells were pelleted (370 g, 3 min, twice) and resuspended in 1 mL RPMI 1640. Suspensions were then diluted in isotonic 200 mM sucrose to achieve desired conductivities.
Human RB cells were isolated from whole blood of healthy volunteers (early twenties). Cells were suspended in a 3.1 % hematocrit RPMI 1640 mix and diluted with 200 mM sucrose. All RB experiments were performed within 10 min of blood draw.
Experimental Setup
Conductivity of suspensions was measured with a SevenMulti meter (Mettler‑Toledo). The FM or AC field was generated by an Agilent 33220A waveform generator and amplified by an F30PV current amplifier. Two 0.5 µm tungsten needles, separated by 100 µm, delivered the field. The needle pair was positioned 50 µL of suspension on a heated stage (25 °C). Imaging was performed with a Nikon TE2000‑U microscope and a Retiga Exi CCD camera (25 fps). The FM waveform’s frequency resolution was verified to be within experimental error.
Additional Movie S1–S3 demonstrate AC DEP manipulation of diatom cells in deionized water: (S1) rotation at 30 kHz, (S2) pushing toward a glass wall at 100 kHz, and (S3) retrieval at 20 MHz.
Results and Discussion
Experimental Observation of a Leukemia Cell Experiencing FM‑DEP
The plug‑in needles allow field application above the substrate, facilitating precise cell selection. Movie S4 shows a triangular diatom cell responding to a 100–500 kHz frequency jump at 0.5 s intervals. Movies S5 and S6 present typical U‑turn trajectories of TL cells under FM‑DEP (0.25 Hz modulation, 200 kHz ≤ f(t) ≤ 3 MHz). Fig. 2 displays a 3‑D trajectory (x, y, t) where the origin corresponds to the needle tip. The x‑axis aligns with the tangent to the electrode surface; the y‑axis reflects the U‑turn amplitude.
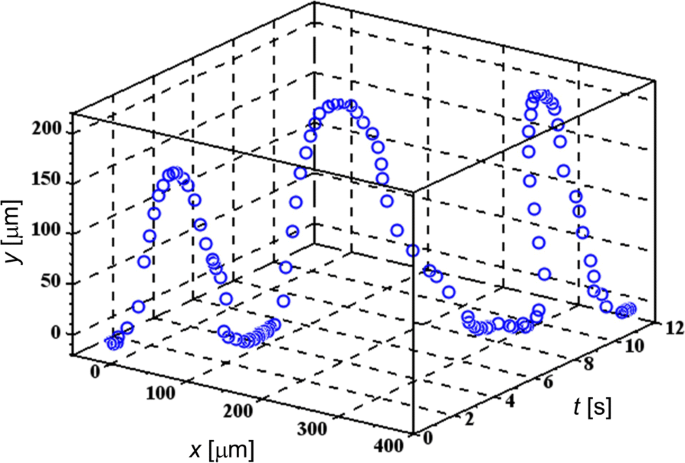
3‑D trajectory of a targeted TL cell. Periodic U‑turns due to the frequency modulation are demonstrated for the TL cell undergoing the FM‑DEP.
U‑turns comprise three phases: leaving the needle, approaching after a U‑turn, and staying on the electrode surface. Solvent flow often prevents exact return to the same spot, but the timing of leaving and U‑turn initiation is clear. The observed U‑turn period (≈4 s) matches the modulation period of 0.25 Hz.
Theoretical Study on the FM‑DEP
For a spherical particle with interior permittivity εin and conductivity σin and exterior εout, σout, the DEP force is
𝐹DEP(𝐫,t)=𝐩(𝐫,t)·∇𝐸(𝐫,t),
with the dipole moment
𝐩(𝐫,t)=4πR3εoutKH{𝐸(𝐫,t)+ (τ/Δτ) Ẽ(𝐫,t)},
where KH=(εin–εout)/(εin+2εout), Δτ–1=τ0–1–τ–1, and τ0 and τ are the characteristic times defined in the text. Averaging over an AC cycle yields the mean DEP force
⟨𝐹DEP⟩=χ(fAC)∇𝐴RMS2,
where χ(f) is proportional to the real part of the CM factor.
Replacing the AC field with an FM wave and invoking the WBL, the averaged DEP force becomes
⟨𝐹DEP(𝐫,t)⟩=χ{f(t)}∇𝐴RMS2,
showing that the force direction changes whenever f(t) crosses fX. This explains the periodic U‑turns observed experimentally.
Solving for the instant when f(t)=fX gives
fX=fc–Δf cos(πfmΔtn),
where Δtn is the measured time from leaving the electrode to the U‑turn. Thus, the FM‑DEP method provides a rapid determination of fX from the observed trajectory.
Comparing Crossover Frequencies of a Single MLV Determined from the FM‑ and AC‑DEPs
Using a salt‑free MLV suspension, we compared fX obtained from FM‑DEP (10–50 kHz FM range, fm=0.1 Hz, 10 s period) and AC‑DEP (30–100 kHz sinusoid). From the FM trajectory, the mean leaving time was 5.8 ± 0.2 s, yielding fX=35 ± 1 kHz. AC‑DEP force measurements via a moving needle yielded a force–frequency curve that fitted the model
FDEP(f)= [L+(2πfτ)2H]/[1+(2πfτ)2],
with fitted parameters L=–21.02 pN, H=19.03 pN, τ=4.9 µs, giving fX=34.15 kHz, in excellent agreement with the FM result.
Conductivity Dependencies of the Crossover Frequencies for Biological Cells
We applied FM‑DEP to TL, BL, and RB cells in conductivity ranges 60–160 mS m–1 (Δf≈700 kHz, fm=0.25 Hz). For each cell type, we measured the leaving times of 10 cells per drop, averaged over two drops, and derived fX using Eq. (10). Figure 6 plots the resulting fX versus σout. The data were fitted to the extended single‑shell model
fX= [σout–(σout2)/(2σcyt)]/(√2 π R Cm) + fX0,
yielding membrane capacitances Cm and cytoplasmic conductivities σcyt that agree with literature values (Cm≈0.5–1.5 µF cm–2, σcyt≈0.3–0.8 S m–1). The consistency across cell types confirms the reliability of FM‑DEP for dielectric characterization.
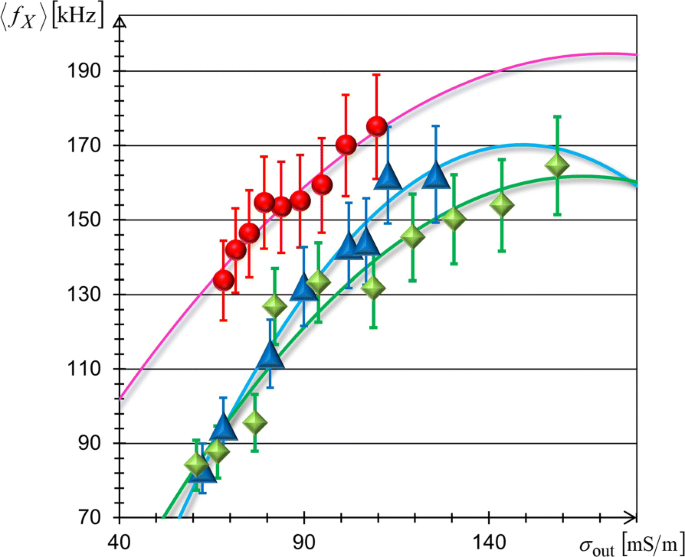
Conductivity dependence of crossover frequencies. Mean crossover frequency, ⟨fX⟩, of TL cells (blue triangles), BL cells (green diamonds), and RB cells (red circles) versus solution conductivity σout. The best‑fit curves of Eq. (15) are shown as solid lines.
Conclusions
Our WBL‑based theory and experiments confirm that FM‑DEP produces predictable U‑turns at the crossover frequency, enabling rapid, <30‑second determination of fX. The FM‑DEP measurements of MLVs match direct AC‑DEP force measurements, while the conductivity‑dependent fX data yield membrane capacitances that align with literature. FM‑DEP therefore offers a robust, noncontact method for dielectric spectroscopy of vesicles and cells, and can be extended to sub‑micron and nanoscopic particles in on‑chip configurations for real‑time electrokinetic spectroscopy.
Abbreviations
- AC:
Alternating current
- BL:
B cell leukemia
- CM:
Clausius–Mossotti
- DEP:
Dielectrophoresis
- DOPC:
1,2‑Dioleoyl‑sn‑glycero‑3‑phosphatidylcholine
- FM:
Frequency modulated
- MLV:
Multilamellar vesicle
- RB:
Red blood
- RMS:
Root mean squared
- TL:
T cell leukemia
- twDEP:
Traveling wave dielectrophoresis
- WBL:
Wide band limit
Nanomaterials
- Optimizing Gold Nanoparticle Placement in TiO₂ Enhances Dye‑Sensitized Solar Cell Efficiency
- High‑Performance CoFe/C Core–Shell Nanocomposites for Broadband Microwave Absorption
- Hydrophilic HBP‑Modified rGO Enables Dual pH/NIR‑Triggered DOX Release for Synergistic Chemo‑Photothermal Tumor Therapy
- Quaternized PVA/Graphene Oxide Composite Membrane Enhances Ethanol Barrier and Ionic Conductivity for Passive Alkaline DEFCs
- Optimizing Niobium-Doped Titanium Oxide Layers for Enhanced Silicon Quantum Dot Solar Cells
- SOX2 Modulates the CCAT1/miR‑185‑3p/FOXP3 Pathway to Control Proliferation and Self‑Renewal of Cervical Cancer Stem Cells
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy
- How the Pandemic is Accelerating Automation: Robots, Cobots, and Smart Manufacturing
- Periodic vs. Predictive Maintenance: Choosing the Right Strategy for Your Business
- Speed, Accuracy, and Safety: Proven Benefits of Automated Welding Cells



