Effects of 15‑nm Gold Nanoparticles on Proliferation, Apoptosis, and Spheroid Formation in HT29 Colon Carcinoma and SPEV Embryonic Kidney Cells
Abstract
Cell‑culture models provide a reliable platform to assess the potential toxicity of nanomaterials and to dissect their mechanistic impact on cancer biology. Here we evaluate how 15‑nm citrate‑stabilized gold nanoparticles (AuNPs) influence two epithelial cell lines of distinct origin: the continuous porcine embryonic kidney line SPEV and the human colorectal carcinoma line HT29. Exposure to AuNP concentrations ranging from 1 to 12 µg mL⁻¹ allowed us to monitor cell adhesion, proliferation, apoptosis/necrosis, and the capacity to form multicellular spheroids. We found that 6–12 µg mL⁻¹ AuNPs significantly reduced cell proliferation and increased early and late apoptotic/necrotic populations in both lines. Conversely, low AuNP doses (1–3 µg mL⁻¹) stimulated spheroid formation, whereas higher doses exerted cytotoxic and anti‑cohesive effects in suspension culture. These results highlight the dose‑dependent dual role of AuNPs and support their continued exploration as a targeted anticancer strategy.
Background
Gold nanoparticles (AuNPs) are prized for their biocompatibility, optical properties, and emerging therapeutic potential across a spectrum of malignancies, including breast, liver, gastric, colon, and lung cancers [5–6]. The unique physicochemical traits of AuNPs—size, shape, surface charge, and concentration—govern their cellular uptake, distribution, and biological effects [12–14]. While many studies focus on the anticancer efficacy of AuNPs, comprehensive toxicity profiling in clinically relevant models remains essential for safe translation.
Targeted delivery of AuNPs to tumor tissue remains a challenge; differential uptake rates have been reported among epithelial cells of various origins [15–16]. Variations in membrane composition, glycoprotein expression, and endocytic capacity likely underlie these differences, underscoring the need for comparative studies in normal and malignant epithelial lines [17].
Cell‑culture assays offer a controlled environment to dissect the cellular responses to AuNPs, informing both safety assessments and therapeutic optimization. In this study, we compared the effects of AuNPs on a normal‑like embryonic kidney line (SPEV) and a colorectal carcinoma line (HT29) using adhesion, proliferation, apoptosis/necrosis, and spheroid formation readouts.
Methods
Culture of SPEV Cells
SPEV cells were maintained in DMEM (Sigma, USA) supplemented with 5 % FCS (HyClone, USA), penicillin/streptomycin (PAA, Austria), and amphotericin B (5 µg mL⁻¹). Cells were seeded at 0.5–2 × 10⁴ cells cm⁻² and cultured in 5 % CO₂ at 95 % humidity. Medium was refreshed every 48 h, and passages were performed upon reaching confluence, preserving the monolayer morphology across passages.
Culture of HT29 Cells
HT29 cells were grown in RPMI‑1640 (Sigma, USA) with 10 % FCS, 2 mM L‑glutamine, and 40 mg mL⁻¹ gentamicin (Sigma, USA). The working density was 0.5–4 × 10⁴ cells cm⁻². Cells were obtained from the Bank of Cell Lines at the RE Kavetsky Institute of Experimental Pathology, Oncology and Radiobiology, NAS of Ukraine.
Manipulation with AuNPs
AuNPs were synthesized by the citrate reduction method [22] and supplied by the Institute of Biochemistry and Physiology of Plants and Microorganisms, Russian Academy of Sciences. The particles averaged 15 nm in diameter, with a stock concentration of 57 µg mL⁻¹. Dark‑field electron microscopy and extinction spectra confirmed the size distribution (Fig. 1). Cells were treated with 1, 3, 6, or 12 µg mL⁻¹ AuNPs for 24–48 h; untreated cultures served as controls.
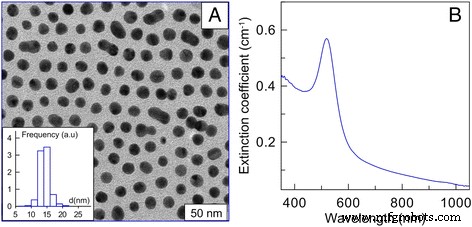
Figure 1. (a) Dark‑field TEM image of 15‑nm AuNPs (size distribution); (b) Extinction spectrum.
Adhesion and Proliferation Assays
Adhesion was quantified by counting flattened cells at 30, 60, 120, 180, and 1440 min after seeding, using an inverted microscope. Proliferation over 1–4 days was assessed by trypsin‑EDTA detachment and counting in a Goryaev chamber. Data were expressed as mean ± SE.
Apoptosis/Necrosis Assessment
After 4 days of AuNP exposure, cells were stained with Annexin‑V and 7‑AAD and analyzed by flow cytometry (FACS Calibur, BD). Results were processed with WinMDI v2.8.
Multicellular Spheroid Formation
To generate spheroids, 5 × 10⁴ cells cm⁻² were seeded in 24‑well plates coated with 1 % agar and cultured in 0.24 % carboxymethyl‑cellulose (CMC) with 80 rpm rotation. Spheroids were allowed to form for 24 h, then maintained for an additional 48 h in the presence of AuNPs. Spheroid size and number were measured by dark‑field microscopy and analyzed with Axio Vision 4.7, calculating volume via V = 0.4 × a × b² (a, b = diameters). Aggregates were grouped by volume (1 × 10⁻⁴–1 × 10⁻² mm³) to determine median volumes and counts.
Statistical Analysis
Data were analyzed using one‑way ANOVA and Student’s t‑test (Statistica 8). Significance was set at p ≤ 0.05, presented as mean ± SE.
Results
Adhesion of SPEV and HT29 Cells
Figure 2 illustrates the impact of AuNPs on SPEV adhesion. After 1 h, 1, 3, and 6 µg mL⁻¹ AuNPs reduced adhered cell counts relative to control, with 12 µg mL⁻¹ causing a 1.8‑fold decrease that persisted over 24 h. HT29 cells showed a similar trend: low concentrations had negligible effects, whereas 6 and 12 µg mL⁻¹ reduced adhesion by 1.16‑ and 1.28‑fold, respectively (Fig. 3).
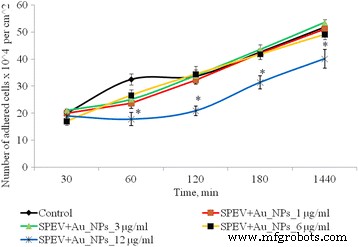
Figure 2. Adhesion dynamics of SPEV cells after AuNP exposure (*p ≤ 0.05 vs. control).
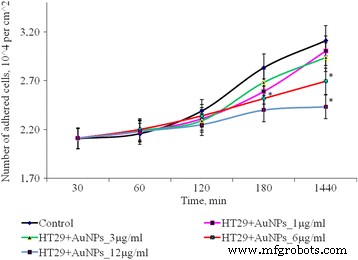
Figure 3. Adhesion dynamics of HT29 cells after AuNP exposure (*p ≤ 0.05 vs. control).
Proliferation of SPEV and HT29 Cells
Figure 4 shows that 1–6 µg mL⁻¹ AuNPs did not alter SPEV proliferation during days 2–4, whereas 12 µg mL⁻¹ reduced cell numbers by up to 1.5‑fold (Fig. 4). HT29 proliferation remained unchanged up to 3 days; at day 4, 6 and 12 µg mL⁻¹ lowered cell counts by 1.33‑ and 1.44‑fold, respectively (Fig. 5).
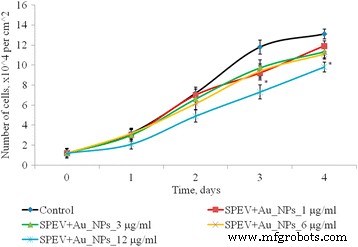
Figure 4. Proliferation of SPEV cells after AuNP exposure (*p ≤ 0.05 vs. control).
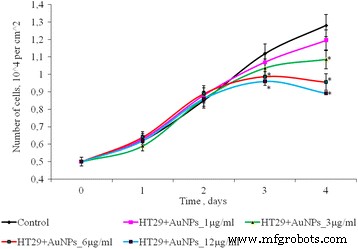
Figure 5. Proliferation of HT29 cells after AuNP exposure (*p ≤ 0.05 vs. control).
Apoptosis/Necrosis Induction
Low AuNP doses (1–3 µg mL⁻¹) did not significantly alter Annexin‑V/7‑AAD profiles in either line (Tables 1–2). At 6–12 µg mL⁻¹, both SPEV and HT29 showed increased early and late apoptotic/necrotic populations. For SPEV, 12 µg mL⁻¹ raised Annexin‑V⁺/7‑AAD⁺ cells by 7.8 % (p ≤ 0.05); for HT29, 6 µg mL⁻¹ and 12 µg mL⁻¹ increased this fraction by 3.2 % and 4.8 %, respectively.
Spheroid Formation
Figure 6 and 7 depict how AuNPs modulate spheroid number and volume. Control HT29 spheroids averaged 5.19 × 10⁻³ mm³, whereas SPEV spheroids averaged 0.79 × 10⁻⁵ mm³. 1–3 µg mL⁻¹ AuNPs amplified SPEV spheroid volume by 9.7‑ and 7.4‑fold, and HT29 volume by 1.4‑ and 1.2‑fold. Increasing AuNP concentration to 12 µg mL⁻¹ reduced spheroid volume in both lines (HT29: 7.18 → 4.24 × 10⁻³ mm³; SPEV: 7.69 → 4.58 × 10⁻⁵ mm³) while increasing the number of HT29 spheroids from 3 to 10 per field and decreasing SPEV spheroids from 32 to 19.
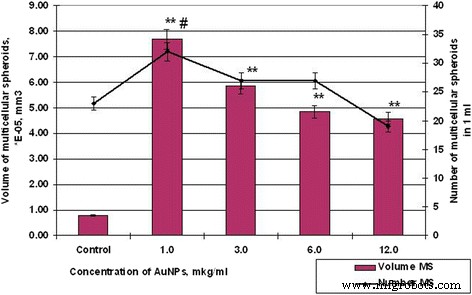
Figure 6. Number and volume of SPEV spheroids after AuNP exposure (#p ≤ 0.01 for number; **p ≤ 0.01 for volume).
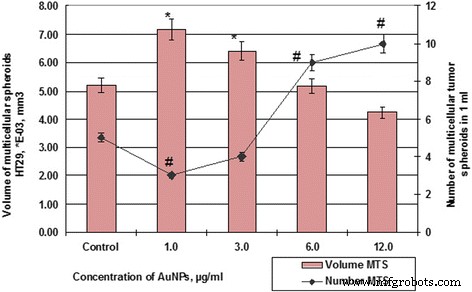
Figure 7. Number and volume of HT29 spheroids after AuNP exposure (#p ≤ 0.01 for number; **p ≤ 0.01 for volume).
Discussion
Our data confirm a dose‑dependent cytotoxic profile of 15‑nm AuNPs in both SPEV and HT29 cells. Concentrations of 6–12 µg mL⁻¹ inhibited proliferation, heightened apoptosis/necrosis, and disrupted cell cohesion in suspension, as evidenced by altered spheroid size and number. The heightened sensitivity of HT29 (6 µg mL⁻¹) relative to SPEV (12 µg mL⁻¹) aligns with previous reports of differential uptake and cytoskeletal disruption by AuNPs [27–30].
Mechanistically, AuNPs may interfere with focal adhesion complexes, modulate actin dynamics, and trigger mitochondrial oxidative stress—pathways that culminate in cell cycle arrest and programmed cell death [31–34]. The observed anti‑cohesive effect at higher doses likely reflects disruption of cell–cell junctions, leading to increased spheroid fragmentation.
While in vitro findings are promising, in vivo validation is essential to confirm therapeutic efficacy and safety. Nonetheless, our study provides a rigorous benchmark for the design of AuNP‑based anticancer strategies that exploit their dual capacity to inhibit tumor growth and modulate cellular microenvironments.
Conclusions
15‑nm AuNPs exhibit clear, concentration‑dependent effects on both normal‑like SPEV and malignant HT29 cells: reduced proliferation, increased apoptosis/necrosis, and altered spheroid dynamics. Low doses (1–3 µg mL⁻¹) promote spheroid formation, whereas higher doses (6–12 µg mL⁻¹) exert cytotoxic and anti‑cohesive actions. These findings support further investigation of AuNPs as a targeted anticancer modality, with attention to dose optimization for maximal therapeutic benefit and minimal off‑target toxicity.
Nanomaterials
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- How Elastic Stiffness and Surface Adhesion Govern Nanoparticle Bouncing Behavior
- Assessing Nanoparticle Aggregation Impact on Polymer Nanocomposite Stiffness with a Two-Step Micromechanical Approach
- Green‑Synthesized Protein‑Coated Gold Nanoparticles from Tricholoma crassum: Antimicrobial, Apoptotic, and Gene‑Delivery Potentials
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- Magnesium‑Doped Zinc Oxide Nanoparticles: Optimizing Photocatalytic Degradation and Antibacterial Efficacy
- Dual‑Mode MRI/Fluorescence Nanoprobe cRGD‑Gd‑Cy5.5 for Targeted Imaging of Integrin αvβ3‑Positive Tumors
- Controlling Silver Nanoparticle Morphology via CTAB‑Capped Seeds and Aging Time
- Comparative Toxicity of PEG-Coated Cobalt Ferrite Nanoparticles and Nanospheres



