Polystyrene Core–Shell Nanocomposites with Lauryl Sulfate‑Doped Polyaniline: Synthesis, Characterization, and Sensing Potential
Abstract
We report the design, synthesis, and comprehensive evaluation of polystyrene (PS) core–shell nanoparticles coated with polyaniline (PANI) doped with lauryl sulfuric acid (LSA). PS nanoparticles (15–30 nm) were prepared via microemulsion polymerization of styrene in aqueous SLS solution. Subsequent in‑situ oxidative polymerization of aniline in the PS latex, in the presence of LSA, yielded a uniform PANI shell. The actual PANI loading was quantified by UV‑Vis spectroscopy, while FTIR confirmed the doping level and oxidation state. TEM and SEM revealed the core–shell architecture and a shell thickness ranging from 10–20 nm. The resulting composites exhibited a non‑linear dependence of conductivity and thermal stability on PANI content, attributable to the dual role of LSA as dopant and plasticizer. Importantly, the NC15 composite (≈15 wt % PANI) displayed superior ammonia‑sensing performance (up to 73 % response at 20 ppm) compared with pure PANI‑LSA, underscoring its potential for gas‑sensing and antistatic applications.
Background
Polyaniline (PANI) is renowned for its electrical conductivity, chemical stability, and low cost, enabling applications in electronics, energy storage, and sensing.1,2 However, PANI’s inherent brittleness and poor processability limit its practical deployment. Incorporating PANI into polymer matrices such as polystyrene (PS) can create core–shell nanocomposites that combine PS’s mechanical robustness with PANI’s conductivity, facilitating fabrication of complex devices.3–5
The core–shell approach typically involves in‑situ oxidative polymerization of aniline within a latex or dispersion of the core polymer.6–10 This method yields a uniform PANI shell around the core particles. Traditionally, HCl or commercial anilinium chloride salts are used as dopants to protonate aniline. However, surfactant‑based acids like dodecylbenzenesulfonic acid (DBSA) have been adopted to simultaneously act as surfactant, dopant, and plasticizer, eliminating the need for additional acid.11,12
Previous studies have demonstrated that PS/PANI core–shell particles can serve in electrostatic accelerators, electrorheological fluids, and potentially in sensing devices.13–20 Yet, systematic investigations of PS/PANI composites as gas‑sensors remain scarce. Recent work blending PS with camphorsulfonic acid‑doped PANI showed moderate ammonia responses, suggesting that direct core–shell architectures might enhance sensitivity by exposing PANI to analytes.23
In this study, we employ lauryl sulfuric acid (LSA) as both dopant and plasticizer during PANI shell formation. LSA’s long dodecyl tail provides surfactant properties, while its sulfate group enables protonation of aniline. This dual functionality is expected to promote uniform shell growth and improve interfacial interactions between PS and PANI.
Methods
Materials
Aniline (Merck), styrene (reagent grade), potassium persulfate (KPS), sodium lauryl sulfate (SLS), and LSA (prepared via ion‑exchange of SLS with KU‑2‑8 resin) were used without further purification. All reagents were stored under argon at 3–5 °C.
Preparation of PS latexes
PS nanoparticles were synthesized by radical polymerization of styrene in an aqueous SLS micellar solution (0.2 g SLS, 0.01 g NaH₂PO₄, 0.01 g KPS) at 70 °C under nitrogen. 2 g of styrene was added over 1.5 h, then the reaction was stirred for 3 h at 70 °C and 1 h at 90 °C. The mixture was cooled, dialyzed (MWCO 3500 Da) against distilled water for 48 h, and the resulting PS latex was used immediately.
Preparation of PS/PANI‑LSA nanoparticles
Aniline (1 mol : 1.5 mol LSA) and KPS (1 mol : 1.25 mol aniline) were added to the PS latex at 10 °C. The initial aniline weight was adjusted to target PANI contents of 1–10 wt % in the final composite. The mixture was stirred for 1 h to form anilinium salts, cooled to 10 °C, and KPS was added dropwise. Polymerization proceeded for 24 h at 10 °C, after which the latex was dialyzed (cellophane membrane) for 3 days and dried under vacuum at 60 °C. Pure PANI‑LSA was synthesized identically but without PS particles.
Characterization
PANI content was quantified by UV‑Vis spectroscopy of leuco‑emeraldine base (LB) solutions in N‑methyl‑2‑pyrrolidone (NMP). FTIR spectra were recorded on KBr pellets (1 cm⁻¹ resolution) using a Bruker Vertex 70. TEM (JEOL JEM‑1400) and SEM (Hitachi S4800) were used to visualize morphology. Thermogravimetric analysis (TGA) was performed under air (10 °C min⁻¹) on a MOM Q‑1500 D system. Conductivity was measured on cast and compression‑molded films (240 °C, 5 MPa, 2 min). Ammonia sensing was evaluated using gold interdigitated electrodes on glass–ceramic substrates, exposing sensors to 19–152 ppm ammonia in air at 50 % RH.
Results and discussion
Morphology of the synthesized PS/PANI‑LSA nanocomposites
TEM images (Fig. 1a) show monodisperse PS cores (15–30 nm), among the smallest reported to date. The PANI‑LSA shell thickness increases with loading, reaching 10–20 nm in NC15 (Fig. 1f). Low‑loading composites (NC2, NC3, NC6) display faint shells due to the large, flexible LSA anions, whereas higher loadings (NC11, NC15) exhibit distinct, irregular shells.

TEM (a–f) and SEM (g–o) images of pure PS and PS/PANI‑LSA composites: a, g – pure PS; b, h – NC2; c, i – NC3; d, m – NC6; e, n – NC11; f, o – NC15.
SEM reveals that pristine PS nanoparticles agglomerate into 30–150 nm clusters. Incorporation of low PANI‑LSA content reduces agglomeration, likely due to the steric stabilization from LS¯ anions. At higher loadings, thicker shells and increased LS¯ concentration promote interparticle interactions, leading to larger agglomerates.
FTIR measurements
FTIR spectra of PS (Fig. 2) confirm aromatic C–H and C=C vibrations. PANI‑LSA shows characteristic bands at 1565, 1492, 1294, 1133, and 818 cm⁻¹, indicative of doped, partially quinoid‑rich PANI. The intense 1180 cm⁻¹ S=O band confirms residual SLS. In NC15, the PANI bands dominate, and the 1133 cm⁻¹ intensity surpasses that of pure PANI‑LSA, reflecting higher oxidation and conductivity.
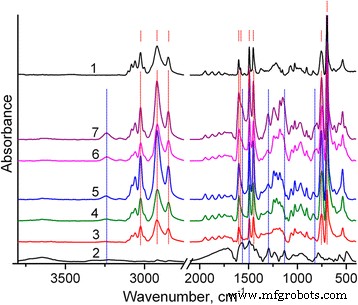
FTIR spectra: PS (1), PANI (2), and composites NC3 (3,4), NC3.5 (5), NC11 (6), NC15 (7). Red dashed lines mark PS peaks; blue lines mark PANI‑LSA peaks.
Thermal stability
TGA (Fig. 4) shows that PS nanoparticles exhibit three weight‑loss stages (up to 505 °C) due to residual SLS. Low‑loading composites (NC2, NC3, NC11) retain PS‑like stability up to ~208 °C. NC15, with the highest PANI‑LSA content, loses weight already at 120 °C, likely from unbound LSA and monomer residues. Above 290 °C, all composites intersect the PS curve, indicating complete dopant loss and transformation of PANI to its dedoped state. Notably, NC2 and NC3 remain thermally superior to PS beyond 290 °C, suggesting a stabilizing effect of the thin PANI shell. In contrast, NC11 and NC15, with thicker shells and more LSA, display reduced stability due to plasticization.
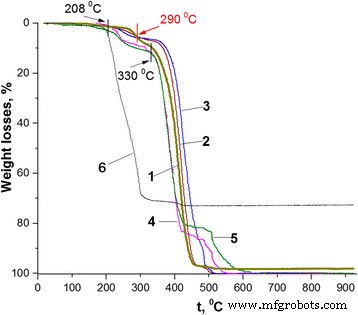
Thermogravimetric curves: 1 – PS; 2 – NC2 (1.84 wt %); 3 – NC3 (3.01 wt %); 4 – NC11 (11.27 wt %); 5 – NC15 (14.82 wt %); 6 – SLS.
Conductivity and sensing properties of the synthesized nanocomposites
Conductivity measurements (Fig. 5) reveal a percolation threshold of 1.26 % vol for both cast and compression‑molded films, far below the theoretical 15–30 % expected for random spheres, confirming efficient phase‑separated PANI networks. The cast films exhibit a higher intrinsic PANI conductivity (2.3 × 10⁻⁴ S cm⁻¹) than compression‑molded films (8.9 × 10⁻⁵ S cm⁻¹), attributable to partial thermal degradation during molding. Critical exponents (1.14 for cast, 2.62 for molded) indicate distinct cluster geometries. Despite lower conductivities compared to larger PS/PANI composites, the values are adequate for antistatic applications.
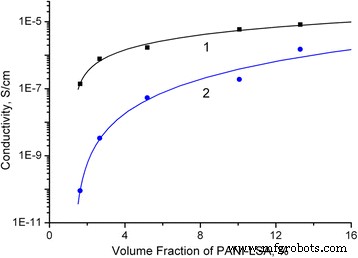
DC conductivity vs. PANI‑LSA volume fraction: cast (1) and compression‑molded (2).
Ammonia sensing (Fig. 6) demonstrates that NC15 outperforms pure PANI‑LSA at 19–114 ppm (up to 73 % response at 20 ppm) due to the high surface area of the core–shell architecture. At higher concentrations (> 114 ppm), the thicker PANI‑LSA shells in pure PANI limit further response, whereas NC15’s thin shells maintain sensitivity. These results confirm the suitability of PS/PANI‑LSA composites for gas‑sensing devices.
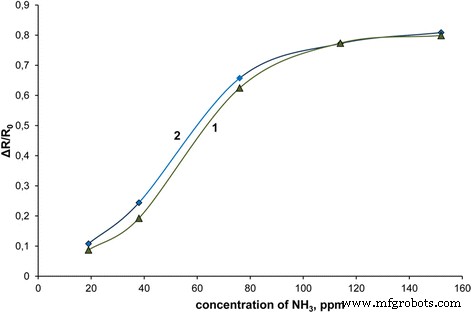
Sensor responses of cast PANI‑LSA (1) and NC15 (2) to ammonia (ppm).
Conclusions
We have successfully synthesized PS core–shell nanoparticles (25–50 nm) coated with 10–20 nm PANI‑LSA shells, the smallest such composites reported. The use of LSA as both dopant and surfactant yielded uniform shells and minimized the need for additional acids. Low PANI loadings suppress agglomeration via LS¯ stabilization, whereas higher loadings promote agglomeration through enhanced LS¯–PS interactions. Thermal analysis shows a dual effect: thin shells stabilize PS, while thick shells introduce plasticization, reducing stability. Conductivity studies reveal a low percolation threshold and a significant conductivity difference between cast and molded films. Ammonia sensing tests confirm that NC15 offers superior sensitivity at low concentrations, validating the core–shell design for gas‑sensing and antistatic applications.
Nanomaterials
- How 3D Printing Is Revolutionizing Aerospace Design and Production
- OSM Standard Sets New Benchmark for Miniaturized Computer‑on‑Modules
- CatalystEX 4.5: New Features, Enhanced Compatibility, and GrabCad Print Integration
- Highly Compressible Graphene/Polyaniline Aerogel: Superelasticity Meets 713 F g⁻¹ Capacitance for All‑Solid‑State Supercapacitors
- Graphene Oxide–Silver Nanoparticle Nanocomposites: A Potent Antibacterial and Antifungal Agent
- Silver‑Embedded Polymer Microfibers for Enhanced Up‑Conversion Optical Sensing
- Discover Unmatched Precision with the New Hi-TECH 750 CNC Lathe
- 5G & IoT: Driving the Next Wave of Digital Transformation
- Pioneering Wire EDM Solutions for Complex Mold and Die Production
- Select Asset Tags with Confidence Using Our New Resources



