Rapid Liver Hemostasis via Electric‑Field‑Enhanced Electrospun Medical Glue Fibers
Abstract
Precise deposition of electrospun fibers remains a major challenge for organ hemostasis. We present an electric‑field‑modified e‑spinning system that incorporates a metal cone at the nozzle, enabling controllable fiber placement. The deposition area is tunable by altering the cone size, a behavior confirmed by finite‑element simulations. In a rat liver resection model, we deposited N‑octyl‑2‑cyanoacrylate (NOCA) fibers directly onto the wound, achieving complete hemostasis within 10 s. Post‑operative histology showed markedly reduced inflammation and tissue adhesion compared with conventional airflow‑assisted e‑spinning. This handheld, battery‑powered device demonstrates that portable, field‑ready electrospinning can provide rapid, accurate hemostasis for emergency, clinical, and home care scenarios.
Background
Liver resection is a standard treatment for hepatic malignancies, yet massive bleeding is common due to the organ’s rich vasculature. Conventional hemostatic techniques—suture, ligation, electrocautery, and fibrin or gelatin sealants—have limitations such as delayed application, tissue damage, immunogenicity, short shelf life, or high cost. Electrospinning offers a low‑dose, surface‑coating approach capable of conforming to irregular wound topography. However, existing e‑spinning devices are bulky, lack precise deposition control, and depend on mains electricity, limiting their use in remote or emergency settings. Our recent airflow‑assisted e‑spinning design reduced deposition area but required an external power source for the air pump. We therefore sought a portable, electric‑field‑modified system that delivers oriented, precise fiber deposition without relying on external power beyond a small battery pack.
Metallic conductors placed within an electrostatic field can induce surface charges that reshape the field distribution. In electrospinning, the charged jet trajectory depends on the local field; thus, a strategically positioned metal cone can focus the jet, reducing its divergence and confining the deposition area. We selected NOCA—a clinically approved cyanoacrylate adhesive—as the spinning material because polymeric fiber mats are more flexible and require less volume than a conventional thick glue film.
In this study, we developed an electric‑field‑modified e‑spinning technique that allows controllable, precise deposition of NOCA fibers on a rat liver resection site, achieving rapid hemostasis. The technique’s portability and accuracy make it suitable for emergency, clinical, field, and home applications.
Methods
Materials
The NOCA adhesive (N‑octyl‑2‑cyanoacrylate with PMMA additive) was supplied by Guangzhou Baiyun Medical Adhesive Co., Ltd. Chloral hydrate (10 % solution) was used for rat anesthesia.
In Vivo Hemostatic Experiments
Forty adult male SD rats (300–350 g) were randomized into two groups (n = 20 each): airflow‑assisted e‑spinning and electric‑field‑modified e‑spinning. After anesthesia and midline laparotomy, a 50 % left‑lobe hepatectomy was performed. In the electric‑field group, a 2.5 cm‑side, 60°‑angled metal cone was mounted on the spinner, with a 10 cm needle‑to‑collector distance and 10 kV voltage. The airflow‑assisted group used a 1.2 mm outlet, 120 µl min^−1 flow, 10 kV, and 10 cm distance. The total procedure lasted ~20 min per rat. All experiments complied with institutional animal care guidelines.
Blood Test and Pathological Sectioning
Blood was collected by cardiac puncture on postoperative days 3 and 5 for WBC count and liver function tests. On day 7, rats were euthanized, livers excised, fixed in 4 % neutral formalin, paraffin‑embedded, and H&E stained.
Electric Field Simulation
Finite‑element analysis modeled a 12 kV supply, copper needle, copper cone, and aluminum collector in air. Needle length, cone diameter, and collector distance were set to 3 cm, 5 cm, and 10 cm, respectively.
Characterization
SEM imaging was performed on a Hitachi TM‑1000. FTIR spectra were recorded with a Nicolet In10 spectrometer. An optical microscope (Olympus BX51) assessed deposition boundaries, and a Casio Exilim camera captured the in‑vivo procedure.
Results and Discussion
Electric Field‑Modified E‑spinning for Precise Deposition
The handheld device (Fig. 1) uses two mercury‑free AAA batteries and a high‑voltage converter, eliminating dependence on mains electricity. A tunable metal cone replaces the previous airflow module, altering the field distribution without extra power. Safety is ensured by maintaining low current despite high voltage.
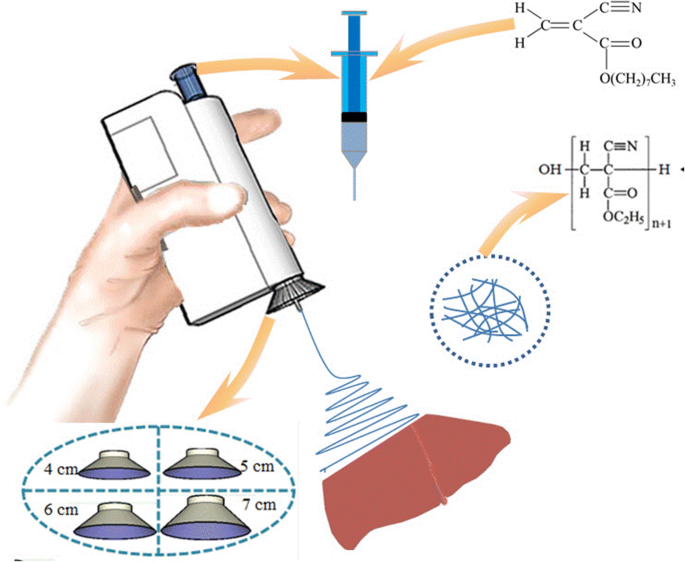
Schematic of the electric‑field‑modified e‑spinning NOCA device for liver hemostasis.
SEM revealed continuous NOCA fibers of 1–3 µm diameter (Fig. 2a). FTIR confirmed polymerization: peaks at 714, 2761, and 1732 cm^−1 (–CH_2–, –C≡N, –C=O) and disappearance of the 3127 cm^−1 (=CH–) band. By varying the cone diameter, the deposition area shrank proportionally (Fig. 2c), indicating tighter jet confinement. Increasing the needle‑collector distance broadened the area (Fig. 2d), but the cone still produced a smaller spread than conventional e‑spinning. This demonstrates the cone’s ability to focus the electric field and enhance deposition precision.
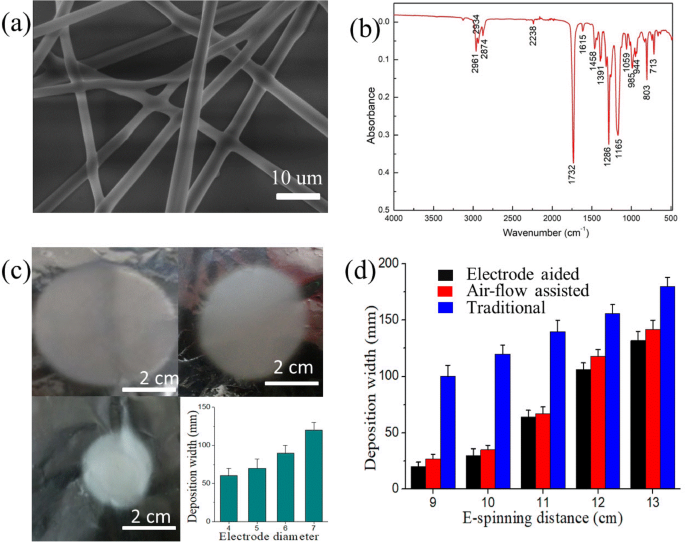
a SEM of NOCA fibers; b FTIR spectrum; c Deposition area vs. cone diameter; d Deposition area vs. distance.
Mechanism Analysis of Precise Deposition
Finite‑element results (Fig. 3) show that the metal cone intensifies the field strength and narrows the divergence angle. The field line near the cone reaches 4 × 10^5 V/m (vs. 3 × 10^5 V/m without the cone) and the angle drops from 20° to 6°, confining the charged jet to a smaller trajectory. This explains the reduced deposition area observed experimentally.
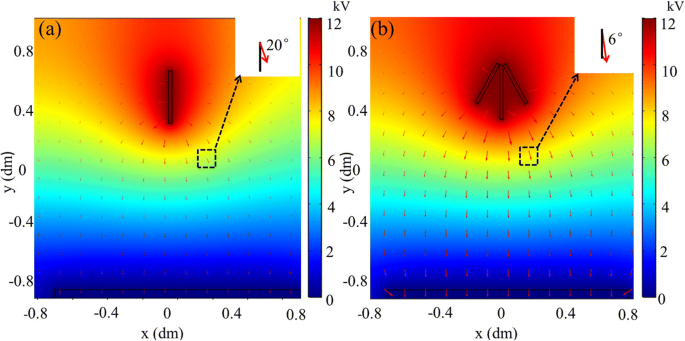
Electric‑field distribution: a without cone; b with cone. Insets show field line angles (6° vs. 20°).
In Vivo Rapid Hemostasis and Analysis
In the rat model, the electric‑field‑modified device achieved complete hemostasis within 10 s (Fig. 4a–c), faster than airflow‑assisted e‑spinning. SEM of the liver surface (Fig. 4d) revealed a uniform 50 µm‑thick fiber membrane that conformed to the irregular wound topography, highlighting the technique’s adaptability.
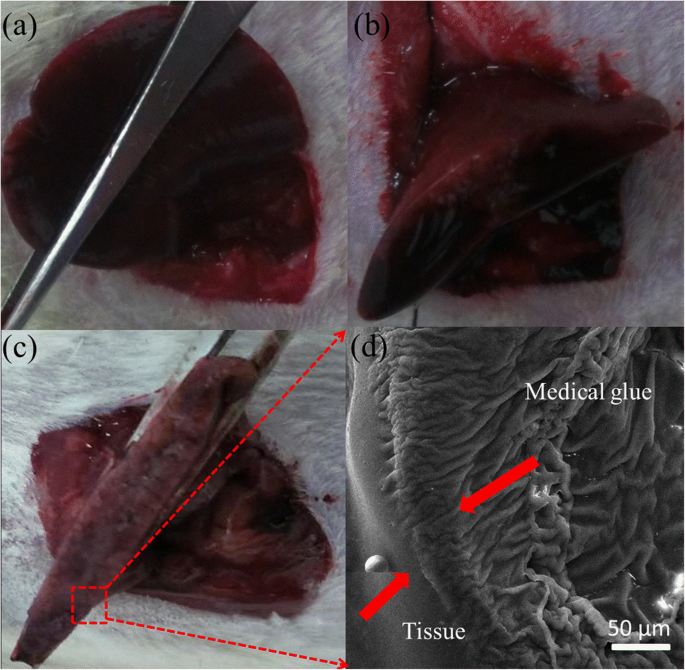
Hemostasis in a rat liver resection model: a liver exposed; b temporary suturing; c NOCA deposition; d cross‑sectional SEM of the fiber membrane.
WBC counts (Fig. 5a) showed significantly lower inflammation in the electric‑field group on day 5 (p < 0.05), comparable to sham controls. Liver enzymes (ALT, AST, GGT) were also markedly reduced (Fig. 5b–d), indicating minimal hepatic injury. Histology (Fig. 6) revealed a clearer tissue boundary, thinner capsule, and fewer inflammatory cells in the electric‑field group versus airflow‑assisted controls, underscoring reduced tissue adhesion and inflammatory response.
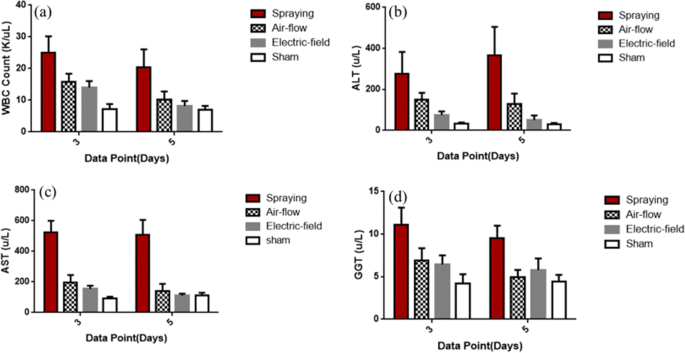
Blood tests: a WBC; b–d liver enzymes (ALT, AST, GGT).
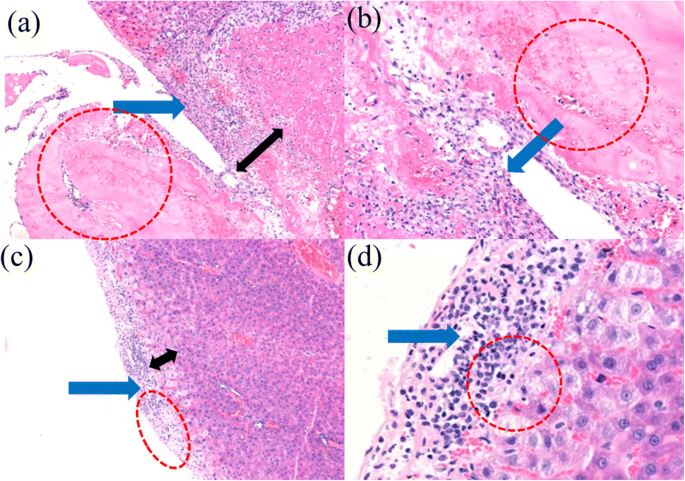
H&E histology (×100 and ×200). Inflammation and liver injury were evident in both groups, but less pronounced in the electric‑field group.
Conclusions
We have demonstrated that an electric‑field‑modified e‑spinning system, featuring a tunable metal cone, delivers precise, oriented deposition of NOCA fibers for rapid liver hemostasis. The deposition area is controllable by cone size, and simulations confirm that the focused field reduces jet divergence. In vivo, the technique achieves hemostasis in 10 s, with lower inflammation and tissue adhesion compared to airflow‑assisted e‑spinning. The handheld, battery‑powered design makes this approach suitable for emergency, clinical, field, and home care settings.
Abbreviations
- ALT:
Alanine aminotransferase
- AST:
Aspartate aminotransferase
- CA:
Cyanoacrylate
- E‑spinning:
Electrospinning
- FTIR:
Fourier transform infrared
- GGT:
Glutamyltransaminase
- HE:
Hematoxylin and eosin
- NOCA:
N‑Octyl‑2‑cyanoacrylate
- PMMA:
Polymethyl methacrylate
- SEM:
Scanning electron microscope
- WBC:
White blood cell
Nanomaterials
- Innodisk: Advanced AIoT Memory & Storage for Medical Innovation
- IBM 5 in 5: On‑Chip Medical Labs Detect Disease at the Nanoscale
- Rapid, Targeted Magnetic Hyperthermia with Auxiliarily‑Guided γ‑Fe₂O₃/Polyurethane Electrospun Fibers
- In Situ Electrospinning of Iodine‑Based Fibrous Meshes for Advanced Antibacterial Wound Dressings
- Controlling Secondary Surface Morphology of Electrospun PVDF Nanofibers via Solvent Choice and Relative Humidity
- Advanced In‑Situ FT‑IR/QMS Monitoring of CpZr(NMe2)3 Decomposition for Reliable ALD Processes
- Sustainable Activated Carbon from Kusha Grass (Desmostachya bipinnata) Enhances Supercapacitor Performance
- Top Rapid Prototyping Methods for Medical Device Innovation
- Choosing the Best Materials for Medical Device Prototypes
- Top 4 Digital Transformation Challenges for Medical Device OEMs



