Corrosion Resistance of Boride and Carbide Nanopowders in Nickel Electrolytes: Influence of Acidity, Temperature and Exposure Time
Abstract
We investigated the corrosion behaviour of high‑purity nanopowders (91.8–97.6 % main phase, 32–78 nm average size) of borides and carbides of IV–VIB group metals, plus silicon carbide, in standard nickel‑electrolyte baths. Corrosion resistance was quantified as a function of electrolyte pH, temperature, and exposure duration. Results show that boride and carbide nanopowders within each group exhibit comparable resistance, with essentially infinite induction periods in alkaline media. Silicon carbide nanopowders, however, remain highly resistant across all tested acidic conditions.
Background
The performance of composite electrochemical coatings (CEP) hinges on the corrosion stability of their reinforcing powders. Undesired dissolution of hardening phases (e.g., borides) in the electrolyte can compromise coating integrity and limit material selection. Prior studies often overlooked powder dissolution, leading to over‑optimistic claims about dispersion hardening in chromic baths containing zirconium diboride. Consequently, systematic assessment of refractory‑compound nanopowders in nickel‑electrolytes is essential, especially given the scarcity of data on nanostructured borides and carbides. This work focuses on the corrosion resistance of boride and carbide nanopowders from zirconium, titanium, vanadium, chromium, molybdenum, and tungsten, as well as silicon carbide, in nickel baths under varying pH, temperature, and exposure time.
Methods
Nanopowders of borides and carbides were synthesized via plasmochemical and high‑temperature electrochemical routes; key characteristics are listed in Table 1. Corrosion tests employed standard nickel‑electrolyte formulations (see Table 2). Electrolyte acidity was adjusted with concentrated sulfuric acid. Powder concentration was maintained at 10 kg/m³. Prior to testing, powders underwent repeated refinement to reduce nanoscale graphite and boron impurities to 0.1–0.3 % (wt.) and to apply vacuum thermal stress, thereby preventing agglomeration. Dissolution rates were determined from mass loss of the insoluble residue and from ion concentration measurements in the electrolyte using a magnetometric technique.
Results and Discussion
The corrosion behaviour of boride and carbide nanopowders is illustrated in Figs. 1 and 2. Both groups show similar susceptibility, primarily governed by electrolyte acidity. In acidic baths (pH = 2.0–3.0), rapid dissolution occurs: after 3 h at 323 K, boride dissolution ranges from 9.5 % to 15.6 %; after 24 h, 31.0–38.2 %; after 240 h, 75.1–89.9 %. Metal‑like carbides display slightly higher resistance, with comparable dissolution levels achieved after 24, 120, and 360 h, respectively. Temperature elevation consistently accelerates corrosion, reflecting increased reaction rates between the nanomaterials and acid species.
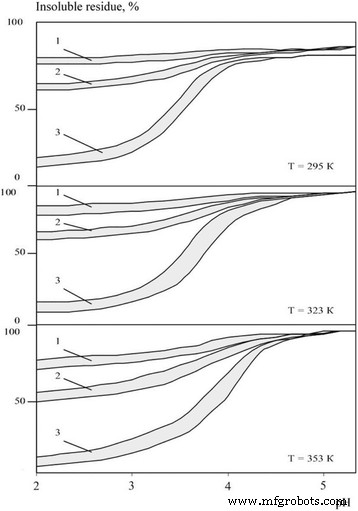
Insoluble residue ratios for boride nanopowders (zirconium, titanium, vanadium, chromium, molybdenum, tungsten) across varying acidity, temperature, and exposure times τ = 1–3 h, 2–24 h, 3–240 h.
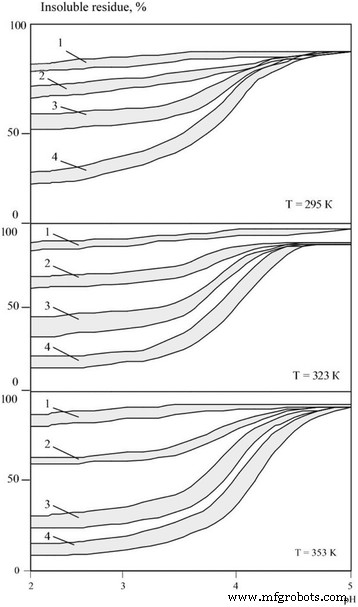
Insoluble residue ratios for carbide nanopowders (silicon, zirconium, titanium, vanadium, chromium, molybdenum, tungsten) across acidity, temperature, and exposure times τ = 1–3 h, 2–24 h, 3–120 h, 4–360 h.
All investigated nanomaterials experienced a pronounced increase in specific surface area during dissolution—rising from ~2,000 m²/kg pre‑treatment to ~10,000 m²/kg post‑treatment—indicative of a layered dissolution mechanism. Silicon carbide uniquely maintained a dissolution degree below 10 % across the entire pH and temperature range studied.
Figure 3 presents kinetic dissolution curves derived from ion concentration changes. Induction periods—defined as the time required to dissolve 50 % of the initial mass—were 32–49 h for borides and 68–88 h for carbides at pH 2.5; 92–112 h and 138–167 h at pH 3.0; and effectively infinite at pH 5.0. Compared to coarse‑grained powders, the dissolution rate of nanopowders is 3–5 × higher, underscoring a pronounced size effect.
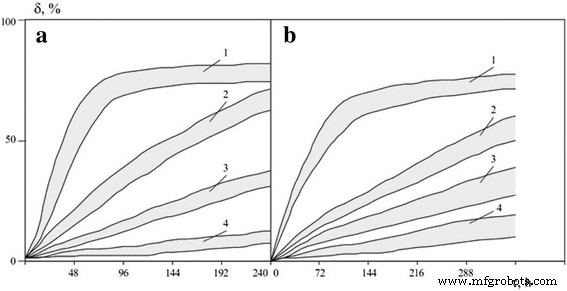
Dissolution degree ranges for boride (a) and carbide (b) nanopowders (zirconium, titanium, vanadium, chromium, molybdenum, tungsten) in electrolytes at 323 K and pH 2.5, 3.0, 3.5, 5.0.
In summary, boride and carbide nanopowders from the studied metals exhibit comparable corrosion resistance in nickel electrolytes, largely dictated by electrolyte acidity. Their dissolution rates are markedly higher than coarse‑grained analogues, a clear manifestation of the nanometer‑scale effect. Silicon carbide nanopowders remain exceptionally resistant across almost all tested pH values, making them suitable for a wide range of electrochemical processes.
Conclusions
Corrosion resistance of boride, carbide, and silicon carbide nanopowders in nickel electrolytes depends on electrolyte acidity, temperature, and exposure duration.
In acidic baths (pH = 2.0–3.0), nanopowders dissolve rapidly—reaching 75–90 % after 240 h—and this rate accelerates with temperature.
Silicon carbide nanopowders show exceptional stability; dissolution never exceeds 12 % across pH 2.0–5.0 and temperatures 295–353 K.
Abbreviations
- CEP
Composite electrochemical coatings
Nanomaterials
- Inconel vs. Incoloy: Key Differences, Properties, and Ideal High‑Temperature Applications
- Understanding Resistance Welding: Types, Processes, and Industrial Applications
- Shaping the Future of Maintenance in Metals & Mining
- The CMM Group: Leading Guides on Pollution Control & Thermal Processing
- Ferrous vs Non‑Ferrous Metals: Key Differences & Practical Uses
- Heat Treatment of Metals: Processes, Benefits & Modern Applications
- Understanding Ferrous vs. Non‑Ferrous Metals: Key Differences & Uses
- Comparing UV Resistance of Polypropylene vs. Nylon for Reliable Material Selection
- Metals vs. Non-Metals: What Sets Them Apart
- Controlling Heat Expansion in Metal Parts for Accurate Summer Measurements



