Tin: From Bronze Age to Modern Industry – Uses, Production, and Future
Background
Tin (Sn, from the Latin stannum) is a silvery‑white metal prized for its corrosion resistance and its ability to coat other metals. In everyday life it appears as the protective layer on steel cans, the alloy in bronze (tin + copper), solder (tin + lead), and the fluoride compound in many toothpastes.
Historical records trace tin use to roughly 3500 B.C. in modern Turkey, where early metalworkers discovered that adding tin to copper produced a harder alloy—bronze—capable of sharper, longer‑lasting tools and weapons. This breakthrough inaugurated the Bronze Age, lasting about 2,000 years, and spurred a worldwide search for tin sources. England’s rich deposits were quietly traded across the Mediterranean, and in 310 B.C. Greek explorer Pytheas located mines near what is now Cornwall. The Roman conquest of Britain in 43 A.D. was driven in part by the desire to control tin trade.
Tin’s reach extended beyond Europe. By 2500‑2000 B.C. artisans in Thailand’s Khorat Plateau produced bronze, and by 1600 B.C. Vietnam already used bronze plows. In the Americas, tin was known in Mexico and Peru long before the Spanish arrived in the 16th century.
Roman engineers first coated copper vessels with tin to preserve their brightness, a practice that spread to iron in the 14th century. The mid‑17th‑century introduction of tinplate—thin sheets of tin‑coated iron—enabled mass production of metal containers. In 1810 Pierre Durand patented the sealed tin‑can, and by the mid‑1800s tin cans had largely replaced glass bottles for food storage.
American inventor Isaac Babbitt’s 1839 alloy of tin, antimony, and copper—known as Babbitt metal—became the standard for bearings, driving advances in high‑speed machinery and transportation.
In 1952 Pilkington’s float glass method, which relies on molten tin as a cooling bath, revolutionized the glass‑making industry by producing ultra‑flat sheet glass without traditional grinding.
Today, Malaysia, Bolivia, Indonesia, Thailand, Australia, Nigeria, and England supply the bulk of global tin. The United States has no major deposits.
Raw Materials
The earth’s crust contains nine tin‑bearing ores, but only cassiterite is mined commercially. Processing often requires limestone, silica, salt, and carbon (coal or fuel oil). Higher impurity levels may necessitate additional reagents.
The Manufacturing Process
Extraction methods vary by deposit type. Deep‑underground mines in Bolivia and England exploit ore with 0.8‑1.0 % tin, while surface gravel deposits in Southeast Asia yield ore with as little as 0.015 % tin. Over 80 % of the world’s tin comes from these low‑grade gravel sources.
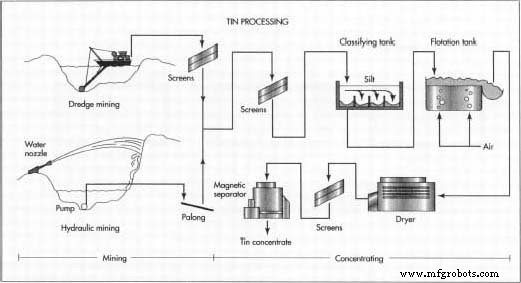
Mining
- Gravel below water level is dredged from an artificial pond using floating dredges equipped with chain‑driven buckets or rotating cutters. The material is screened on board to separate soil, sand, and stones before being transferred ashore.
- Dry deposits are broken with high‑pressure water jets; the resulting slurry is stored in a pond and pumped into a wooden trough (palong) where heavier tin ore settles behind riffles. Periodic dumping collects the ore for further processing.
Concentrating
- Ore passes through vibrating screens, a classifying tank, and optionally a flotation tank to separate fine silt and floatable particles.
- After drying and magnetic separation, the tin concentrate reaches 70‑77 % purity, predominantly cassiterite.
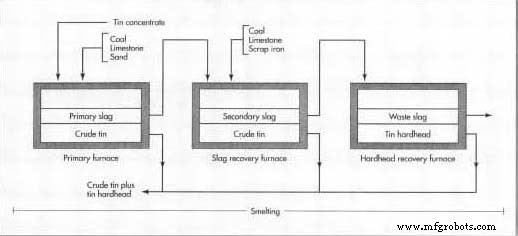
Smelting
- The concentrate is heated with carbon (coal or oil) to ~2550 °F (1400 °C). Carbon monoxide reduces cassiterite to crude tin, while limestone and sand form slag with silica or iron impurities.
- Because tin can bond with slag, a second furnace re‑melts the slag with additional carbon, scrap iron, and limestone to recover more tin. A third pass extracts “hardhead” tin from the residual slag.
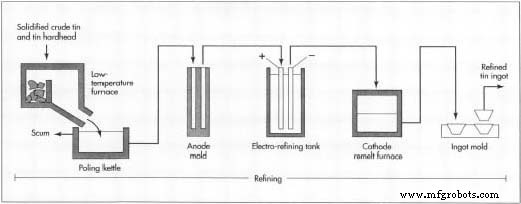
Refining
- Crude tin, hardhead, and slag tin are melted in a low‑temperature furnace where only tin liquefies. The liquid is collected in a poling kettle, separating it from solid impurities (liquidation).
- Boiling—agitation with steam or green wood poles—removes surface scum, yielding tin of ~99.8 % purity.
- Electrolytic refining further purifies tin to >99.98 % by depositing it onto cathodes in an electrolytic cell. Impurities settle as slime at the bottom.
- Final refined tin is cast into ingots or bars: lower purity grades into 25‑100 lb (11‑45 kg) ingots; higher purity into ~2 lb (1 kg) bars.
Quality Control
Consistent 99 %+ purity is achieved through routine sampling at critical stages, allowing real‑time adjustments. In the U.S., ASTM B339 defines commercial tin grades: AAA (99.98 % tin) for research, A (99.80 %) for tinplate, and B–E (down to 99 %) for general alloys.
Byproducts & Waste
While no useful byproducts emerge, waste includes rejected soil, sand, and stones, whose environmental impact depends on local disposal practices. Smelting slag may contain arsenic, lead, and other hazardous elements, but tin itself poses no known harm to humans or ecosystems.
The Future
Demand for tin is projected to rise as it replaces more toxic metals like lead, mercury, and cadmium. Emerging applications include tin‑silver solders for electronics and tin shot for ammunition, offering less environmental impact. Research is also exploring tin‑based compounds that bind heavy metals in landfill sites, preventing leaching into soil and groundwater.
Images

A tin bonnet was a common 10th‑anniversary gift in the 1800s, displayed as a decorative piece.
Manufacturing process
- Titanium: Key Properties, Applications, and Industrial Uses
- Castanets: History, Craftsmanship, and Performance
- The History, Production, and Future of Glue: From Ancient Adhesives to Modern Innovations
- Thread: Types, History, Production, and Quality Control
- Acetylene: Production, Uses, and Handling – A Comprehensive Overview
- Comprehensive Overview of Asbestos: From Ancient Uses to Modern Health Risks
- The History, Design, and Manufacturing of Dice: From Ancient Tools to Modern Gaming
- Plywood: History, Production, and the Path Ahead
- Silver: History, Properties, and Modern Applications
- The Evolution and Production of Toothpaste: From Ancient Remedies to Modern Fluoridated Formulas



