Green‑Synthesized Cerium‑Doped Carbon Nanoparticles Deliver Superior Hydroxyl‑Radical Scavenging for Antioxidant Therapy
Abstract
Recent advances in nanomedicine have highlighted cerium oxide nanoparticles as powerful antioxidants. Here, we present a straightforward, environmentally benign method that uses bull serum albumin (BSA) to biomineralise cerium‑doped carbonaceous nanoparticles (Ce‑doped CNPs). The resulting particles are monodisperse, averaging 14.7 nm in diameter, and display abundant hydrophilic surface groups that ensure long‑term dispersion in aqueous media. In vitro cytotoxicity studies (CCK‑8) confirm negligible cell toxicity across a wide dose range. When challenged with hydrogen‑peroxide‑induced reactive oxygen species, Ce‑doped CNPs exhibit exceptional hydroxyl‑radical scavenging, protecting vascular smooth‑muscle cells and 7721 tumour cells from oxidative damage in a dose‑dependent manner. These findings establish Ce‑doped CNPs as promising, low‑toxicity ROS scavengers for the treatment of oxidative‑stress‑related diseases.
Background
Oxidative stress, defined as an imbalance between reactive oxygen species (ROS) production and antioxidant defenses, underlies many chronic diseases, including atherosclerosis, cancer, and neurodegeneration. The highly reactive hydroxyl radical (•OH) is particularly damaging, initiating lipid peroxidation, protein modification, and DNA strand breaks that culminate in cell death and tissue injury. Therapeutic strategies that reduce ROS levels, especially •OH, are therefore of great clinical interest.
Cerium oxide nanoparticles (CeO₂ NPs) have emerged as potent nano‑enzymes owing to the reversible Ce³⁺/Ce⁴⁺ redox cycle. This allows them to mimic superoxide dismutase (SOD) and catalase (CAT) activities, scavenging •O₂⁻ and H₂O₂, respectively. Nevertheless, conventional CeO₂ NPs often require surface coatings (e.g., PVP) to achieve aqueous stability and biocompatibility, which can alter their intrinsic properties. Our study circumvents this limitation by embedding cerium ions within a carbonaceous framework derived from BSA, yielding Ce‑doped CNPs with intrinsic hydrophilicity and robust antioxidant performance.
Experimental Section
Materials and Reagents
Bull serum albumin (BSA), fluorescein diacetate (FDA), and propidium iodide (PI) were sourced from Sigma (New York, NY, USA). Fetal bovine serum (FBS) and Dulbecco’s Minimum Essential Medium (DMEM) were obtained from Invitrogen China Limited (Shanghai). Cerium nitrate hexahydrate (Ce(NO₃)₃·6H₂O), methyl violet (MV), ferrous sulfate heptahydrate (FeSO₄·7H₂O), and 30 % H₂O₂ were purchased from Aladdin Reagent Corporation (Shanghai) and Sinopharm Chemical Reagent Co., Ltd. (Beijing). All chemicals were analytical grade and used without further purification; de‑ionised water was employed throughout.
Synthesis of Cerium‑Doped Carbonaceous Nanoparticles
The Ce‑doped CNPs were prepared via a biomineralisation route adapted from the BSA‑templated synthesis reported by Wang et al. In brief, 1.25 g of BSA was dissolved in 50 mL de‑ionised water under continuous stirring to form a clear solution. A 300 mM Ce(NO₃)₃·6H₂O solution was then added dropwise while maintaining vigorous stirring. Simultaneously, 2.0 M NaOH was introduced slowly to raise the pH to 12. The mixture was stirred at 55 °C for 8 h, yielding a brown suspension that cooled to room temperature. The product was filtered through a 0.22‑µm PES membrane to remove large aggregates, dialysed against water (14 kDa MWCO) for 3 days, and freeze‑dried under vacuum. The resulting powder was stored for further characterisation.
Characterisation Techniques
Transmission electron microscopy (TEM, JEOL JEM‑2100) provided morphological data at 200 kV. Particle size distributions were calculated with ImageJ (NIH). Fourier‑transform infrared spectroscopy (FT‑IR, Nicolet Nexus 470) and X‑ray photoelectron spectroscopy (XPS, PHI 5000) identified surface functional groups and oxidation states. X‑ray diffraction (XRD, Rigaku D/MAX‑2000) confirmed crystalline phases. UV‑Vis absorption spectra (Shimadzu UV‑2550) were recorded for solution studies.
UV‑Vis Photometric Assay of Radical Scavenging
Ce‑doped CNPs (10 µM) were dispersed in 0.1 M Tris‑HCl (pH 5.0) and sonicated to enhance solubility. A reaction mixture containing 3.0 × 10⁻⁵ M MV, 0.15 mM FeSO₄·7H₂O, 1.0 M H₂O₂, and the Ce‑doped CNP suspension (0.17 mM) was prepared in a 10 mL volume. After 5 min at room temperature, absorbance at 582 nm was measured to quantify MV recovery, reflecting hydroxyl‑radical scavenging.
Cell Viability (CCK‑8) Assay
VSMC and 7721 cells (1.5 × 10⁴ cells/well) were seeded in 96‑well plates and cultured to ~80 % confluence. After 24 h, media were replaced with DMEM containing 0–400 µg/mL Ce‑doped CNPs, and cells were incubated for 24 h. CCK‑8 reagent (10 µL/well) was added and incubated for 4 h. Absorbance at 490 nm was read on a Synergy HT microplate reader; viability was expressed as % of untreated control.
Hydrogen‑Peroxide‑Induced Oxidative Stress Model
To model oxidative injury, VSMC and 7721 cells were exposed to 30 % H₂O₂ at concentrations ranging from 50 µmol mL⁻¹ to 800 µmol mL⁻¹ for 4 h. Post‑treatment, cell viability was assessed by CCK‑8 as described above, enabling determination of the LD₅₀ of H₂O₂ (≈560 µM).
Apoptosis and Live‑Dead Assays
Cells pre‑treated with Ce‑doped CNPs (0–400 µg/mL) were subsequently challenged with 560 µM H₂O₂ for 4 h. Live‑dead staining (Calcein‑AM/PI) was performed on 7721 cells cultured in 6‑well plates, and fluorescence images were captured under a confocal microscope.
Flow Cytometry Analysis of Apoptosis
Following H₂O₂ exposure, 7721 cells were harvested, washed, and stained with Annexin V‑FITC and PI. Samples were analysed on a Beckman Coulter Epics XL MCL cytometer; percentages of early (Annexin V⁺/PI⁻) and late (Annexin V⁺/PI⁺) apoptotic cells were calculated using FlowJo 7.6.2.
Statistical Analysis
Data represent mean ± SEM. One‑way ANOVA followed by Tukey’s post‑hoc test was applied; p < 0.05 was considered statistically significant.
Results and Discussion
Nanoparticle Synthesis and Morphology
The biomineralisation strategy yielded highly monodisperse Ce‑doped CNPs, as shown by TEM (Figure 1a). The particles displayed a polygonal, non‑aggregated morphology with an average diameter of 14.7 ± 0.8 nm (Figure 1b). The aqueous dispersion remained clear for several days, indicating excellent colloidal stability—a key advantage for biomedical delivery.
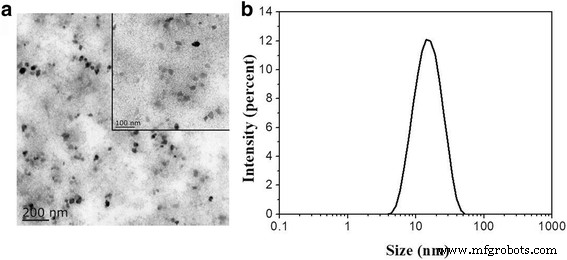
a TEM images of Ce‑doped CNPs. b Size distribution.
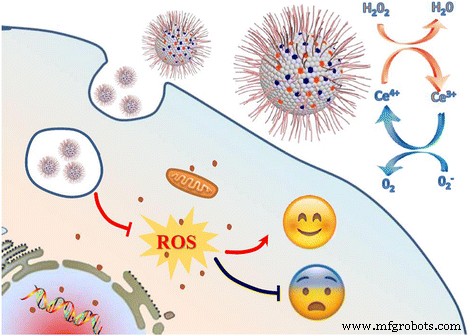
Schematic of the Ce‑doped CNP design.
Surface Chemistry and Oxidation State
XPS analysis confirmed the coexistence of Ce³⁺ (≈15.99 %) and Ce⁴⁺, underpinning the redox‑active nature of the particles (Figure 2b). The C 1s spectrum (Figure 2c) revealed peaks at 285.0, 287.8, and 288.9 eV, corresponding to C–C, C=O, and C–O groups, while the O 1s spectrum (Figure 2d) displayed peaks at 530.5 and 531.8 eV, indicative of Ce–O bonds and surface hydroxyls. FT‑IR spectra (Figure 3A) displayed a broad O–H stretch at 3400 cm⁻¹ and nitrate‑related peaks at 1510 and 1450 cm⁻¹, confirming the presence of hydrophilic functional groups and successful incorporation of cerium ions.
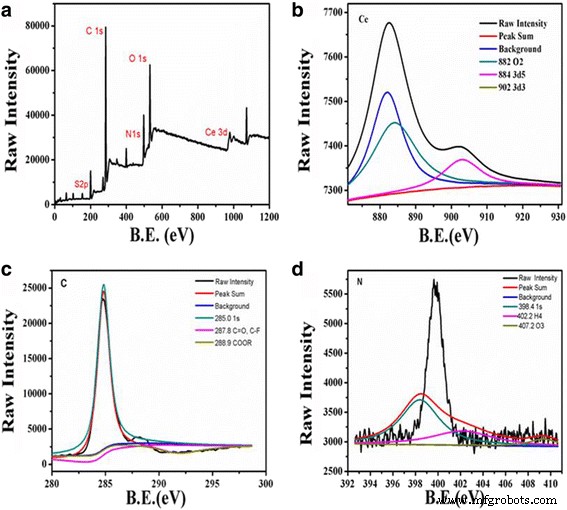
a XPS survey spectrum. b Ce 3d spectrum. c C 1s spectrum. d O 1s spectrum.
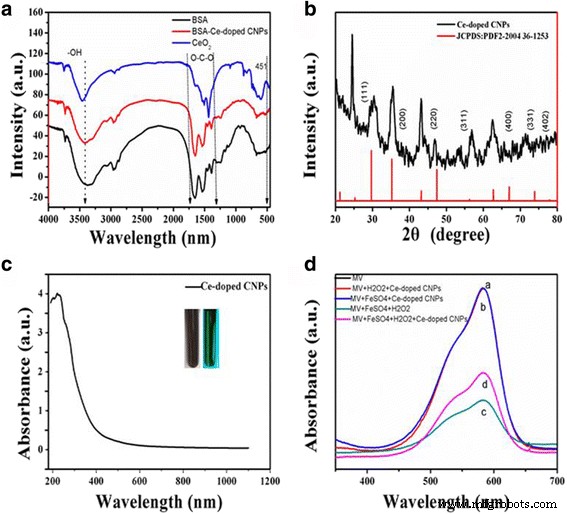
A FT‑IR spectrum of BSA and Ce‑doped CNPs (a). XRD pattern (b). UV‑Vis absorption of Ce‑doped CNPs (c). MV absorbance after various treatments (d). a MV alone. b MV + H₂O₂ + Ce‑doped CNPs. c MV + FeSO₄ + H₂O₂. d MV + FeSO₄ + H₂O₂ + Ce‑doped CNPs after 5 min.
XRD patterns (Figure 3B) revealed characteristic peaks of a hybrid carbon‑cerium lattice, with carbon‑related peaks near 24.4° and ceria reflections at 30.3°, 35.23°, and 43.2°, confirming successful integration of Ce into the carbon framework.
In Vitro Cytotoxicity
CCK‑8 assays demonstrated that Ce‑doped CNPs did not compromise cell viability at concentrations up to 400 µg/mL (Figure 4a). Even at the highest dose, viability remained above 90 %, underscoring the excellent biocompatibility of the particles.
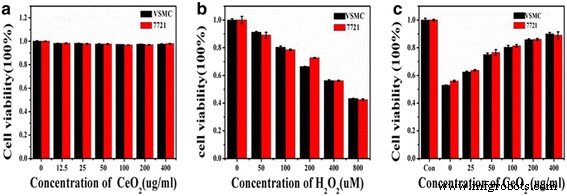
a Cell viability after Ce‑doped CNP exposure. b VSMC viability under 560 µM H₂O₂ (CCK‑8). c 7721 cell viability under the same oxidative stress.
Antioxidant Efficacy
Hydrogen peroxide at 560 µM was identified as the LD₅₀ for both VSMC and 7721 cells. Pre‑treatment with Ce‑doped CNPs significantly improved survival in a dose‑dependent manner, with 400 µg/mL restoring viability to ~88 % (Figure 4c). Live‑dead staining (Figure 5) showed a marked reduction of red (PI‑positive) cells with increasing Ce‑doped CNPs, confirming protection against oxidative apoptosis. Flow cytometry (Figure 6) quantified apoptosis: H₂O₂ alone induced 46.4 % late apoptosis, which dropped to 24.2, 20.0, 15.1, and 11.2 % with 50, 100, 200, and 400 µg/mL Ce‑doped CNPs, respectively.
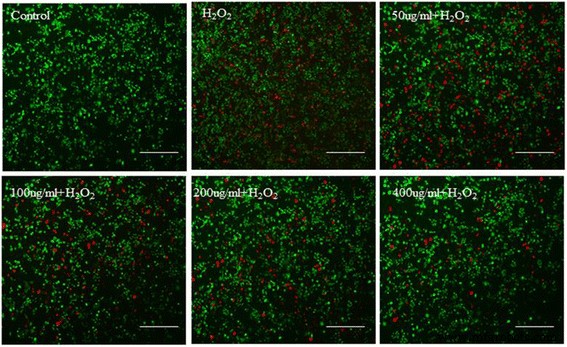
Live‑dead staining of 7721 cells after H₂O₂ exposure and Ce‑doped CNP treatment (scale bars = 50 µm).
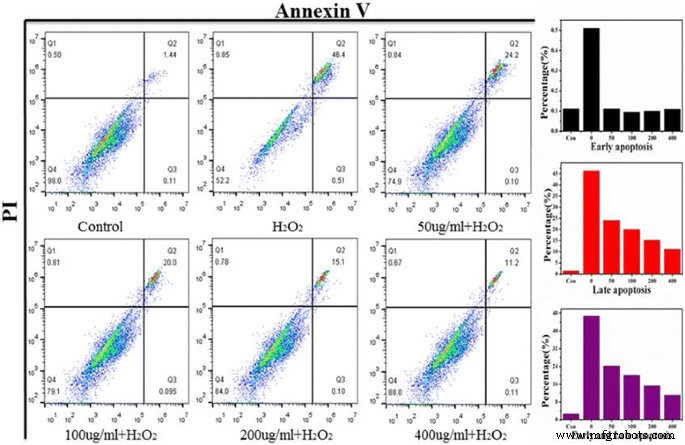
Flow cytometry profiles of 7721 cells: early (Annexin V⁺/PI⁻) and late (Annexin V⁺/PI⁺) apoptosis under various treatments.
Conclusions
We have developed a green, one‑step synthesis of cerium‑doped carbonaceous nanoparticles that are highly dispersible, biocompatible, and exhibit potent hydroxyl‑radical scavenging. The particles retain intrinsic Ce³⁺/Ce⁴⁺ redox cycling, enabling them to mimic SOD and CAT activities and protect vascular and tumour cells from H₂O₂‑induced oxidative injury. Their dose‑dependent antioxidant efficacy and minimal cytotoxicity position Ce‑doped CNPs as attractive candidates for the prevention and treatment of oxidative‑stress‑related pathologies.
Nanomaterials
- Sol‑Gel Fabricated SiO₂@C/MWNT Nanocomposites Deliver Superior Li‑Ion Battery Anodes
- Engineering Monodisperse GFP‑Doped Silica Nanoparticles for Efficient Intracellular Protein Delivery and Bioimaging
- Optimized Post‑Treatment Strategy for Monodisperse FePt–Fe3O4 Binary Nanoparticles
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Efficient Solution-Scale Synthesis of Red Phosphorus Nanoparticles for High‑Performance Lithium‑Ion Battery Anodes
- High‑Sensitivity Fe³⁺ Detection Using Ag‑Functionalized TiO₂ Nanotube Arrays via Anodic Stripping Voltammetry
- Carbon‑Coated Molybdenum Phosphide Nanoparticles: A Low‑Cost Catalyst with 131 mV Overpotential for Alkaline Hydrogen Evolution
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy
- In‑Situ Synthesis of Silver Nanoparticles on Amino‑Grafted Polyacrylonitrile Fibers for Enhanced Antibacterial Performance
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy



