Optical Properties and Growth Mechanism of Macroporous La₃Ga₅.₅Nb₀.₅O₁₄ Ceramic Synthesized via Pechini Process
Abstract
We investigated the optical characteristics and growth pathway of La3Ga5.5Nb0.5O14 (LGN) powders produced by the Pechini sol‑gel method. After calcination at 600–800 °C, X‑ray diffraction confirmed the emergence of a single‑phase orthorhombic LGN structure (JCPDS 47‑0533) at 800 °C. A schematic growth mechanism is proposed, detailing the transformation of precursor nano‑foams into macroporous crystalline grains. Photoluminescence measurements, excited at 327 nm, reveal a broad blue emission centered at 475 nm (77 K), attributed to the [NbO6]7– octahedral group. The 800 °C sample exhibits a sharp optical absorption edge at 320 nm, corresponding to a band‑gap energy of 3.95 eV and indicating minimal oxygen‑vacancy defects.
Introduction
Compounds with the Ca3Ga2Ge4O14 (CGG) structure—such as La3Ga5SiO14 (LGS), La3Ga5.5Nb0.5O14 (LGN), and La3Ga5.5Ta0.5O14 (LGT)—have been extensively explored for their piezoelectric and optical properties. These materials underpin high‑performance bulk acoustic wave (BAW) and surface acoustic wave (SAW) filters, oscillators, and nonlinear optical devices. LGN, in particular, exhibits a wide mid‑infrared transparency window (2–6 µm) and has been characterized for phase‑matching in second‑harmonic and difference‑frequency generation up to 6.5 µm. Recent studies have demonstrated Nd‑doped LGN lasers with tunable output under diode pumping.
Macroporous ceramics, defined by porosities of 5–90 % and pore sizes exceeding 100 nm, combine high mechanical strength, thermal conductivity, and chemical stability, making them attractive for filtration, catalysis, thermoelectric conversion, and biomedical applications. Their photonic band gaps enable advanced uses in optical communications and gas sensing. However, LGN‑based macroporous ceramics prepared via chemical routes remain underexplored. Traditional synthesis of LGN single crystals requires temperatures above 1500 °C (Czochralski method), whereas the Pechini process offers a low‑temperature alternative (~800 °C) with superior compositional homogeneity and particle size control.
In this work, we employ the Pechini method to synthesize a single‑phase, orthorhombic LGN ceramic and systematically investigate its growth mechanism, morphology, and optical properties.
Methods/Experimental
Materials Used
High‑purity lanthanum nitrate hexahydrate La(NO3)3·6H2O, gallium nitrate Ga(NO3)3, niobium pentachloride NbCl5, citric acid anhydrous (CA), and ethylene glycol (EG) were used.
Preparation of La3Ga5.5Nb0.5O14
The LGN precursor was prepared by dissolving stoichiometric amounts of La(NO3)3, Ga(NO3)3, and NbCl5 in water. NbCl5 reacts with ethanol to form niobium ethoxide (Nb(OC2H5)5) according to:
\[\mathrm{NbCl}_5 + 5\mathrm{C}_2\mathrm{H}_5\mathrm{OH} \rightarrow \mathrm{Nb}(\mathrm{OC}_2\mathrm{H}_5)_5 + 5\mathrm{HCl}\]
A chelating agent (CA) was added in a 2:1 molar ratio to the metal ions, followed by ethylene glycol to promote polymerization. The resulting gel was dried at 120 °C for 24 h and then sintered in air at 600–800 °C for 3 h to obtain the macroporous LGN ceramic.
Characterization/Phase Identification
DTA–TGA (PE–DMA 7) monitored the thermal decomposition of the precursor. X‑ray powder diffraction (Rigaku Dmax‑33) identified the crystal phases. High‑resolution TEM (HF‑2000, Hitachi) examined morphology and microstructure. Photoluminescence (Hitachi‑4500, xenon lamp) measured excitation/emission spectra at 300 K and 77 K. UV–vis absorption (Hitachi U‑3010) evaluated the band‑gap energy.
Results and Discussion
The amorphous precursor undergoes pyrolysis to form crystalline LGN. The overall decomposition reaction can be summarized as:
\[3\mathrm{La(NO}_3)_3 + 5.5\mathrm{Ga(NO}_3)_2 + 0.5\mathrm{Nb(OC}_2\mathrm{H}_5)_5 \xrightarrow{\text{CA}} \mathrm{La}_3\mathrm{Ga}_{5.5}\mathrm{Nb}_{0.5}\mathrm{O}_{14} + \mathrm{NO}_2\uparrow + \mathrm{H}_2\mathrm{O}\uparrow + \mathrm{CO}_2\uparrow + \mathrm{C}_2\mathrm{H}_5\mathrm{OH}\uparrow\]
Figure 1 shows XRD patterns at 600, 700, and 800 °C. At 600 °C, weak micro‑crystalline peaks appear; by 700 °C, crystallization initiates; at 800 °C, a sharp, single‑phase orthorhombic LGN pattern (JCPDS 47‑0533) emerges, confirming full crystallization.
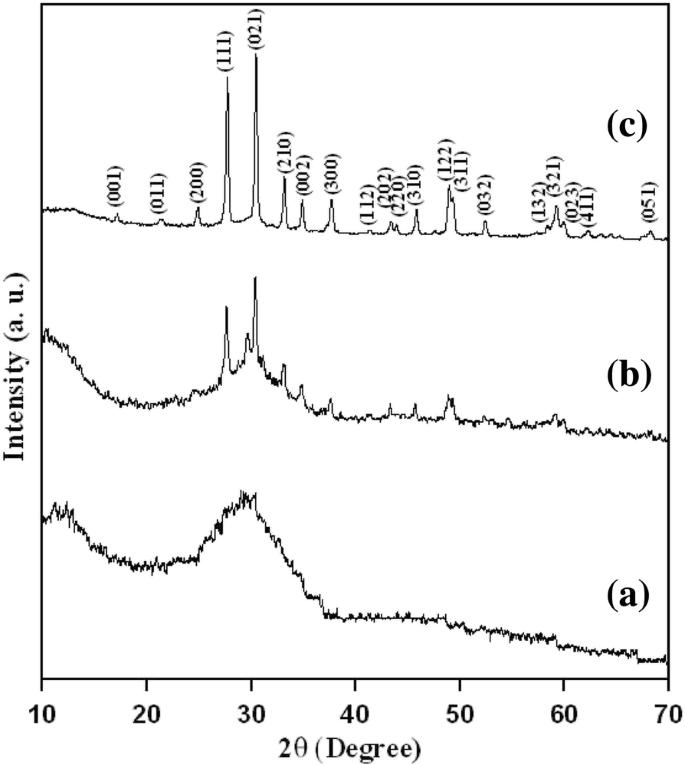
X‑ray diffraction patterns of LGN precursor powders annealed at (a) 600 °C, (b) 700 °C, and (c) 800 °C for 3 h.
FT‑IR spectra (Figure 2) reveal strong CO2 stretching at 2300 nm and nitrate absorption at 1500 nm at 600–700 °C. New peaks between 500–600 nm appear at 800 °C, indicating the formation of LGN nanocrystals and the presence of residual organics.
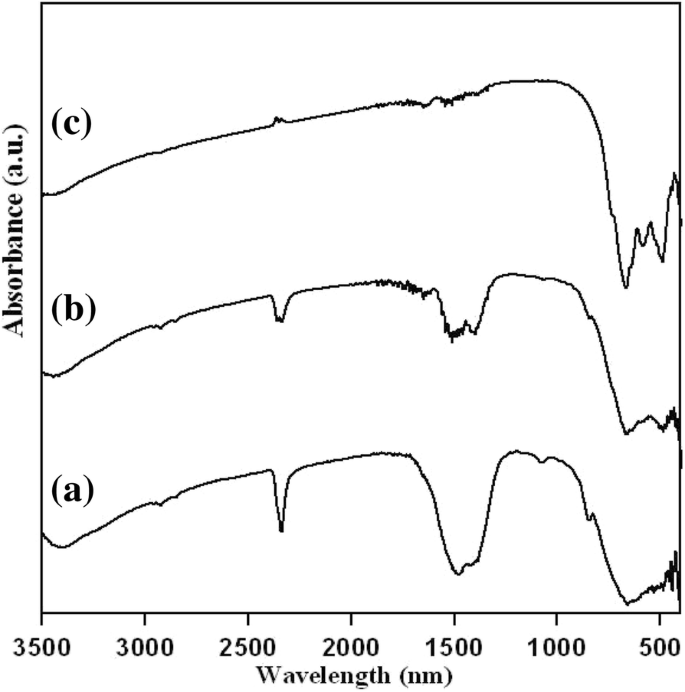
FT‑IR spectra of dried powders at (a) 600 °C, (b) 700 °C, and (c) 800 °C.
TEM images (Figure 3) illustrate the precursor morphology at 600 °C: nano‑foam structures with diameters <100 nm are evident. As the temperature rises, these foams collapse, creating microporous voids and promoting grain growth. Higher‑magnification TEM (Figure 4) confirms the emergence of nanocrystalline domains.

Morphology of as‑synthesized LGN at 600 °C. Inset: TEM image of nano‑foam at the precursor surface.

(a) TEM at 700 °C; (b) high‑resolution TEM; (c) lattice image; (d) electron diffraction.
A schematic growth mechanism (Figure 6) describes the evolution from nano‑foams at 600 °C to macroporous grains at 800 °C. During 600–700 °C, foams expand and collapse, forming pores <100 nm. Concurrently, micro‑crystals nucleate and coalesce, reducing interfacial energy and yielding well‑defined macroporous grains.
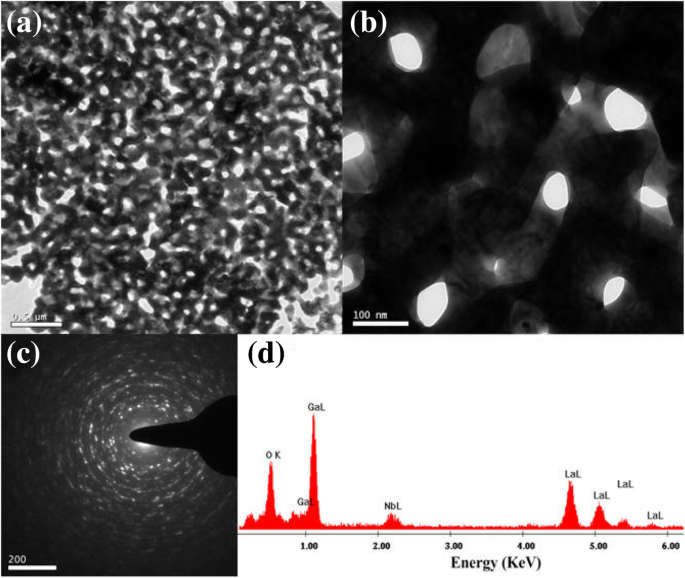
(a) TEM at 800 °C; (b) high‑resolution TEM; (c) electron diffraction; (d) EDX composition.
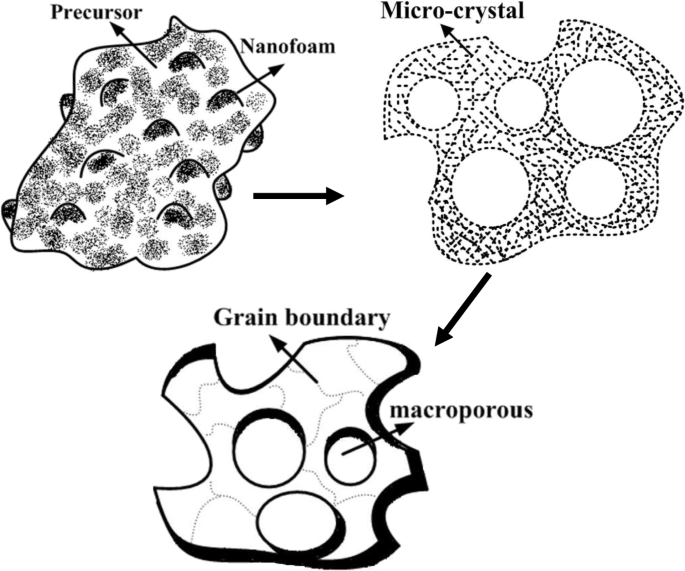
Schematic of macroporous LGN growth via the sol‑gel route.
Photoluminescence spectra (Figure 7) show a prominent blue emission at 475 nm under 327 nm excitation at 77 K, attributable to the [NbO6]7– octahedral group. At 300 K, the emission intensity diminishes due to thermal quenching via non‑radiative phonon coupling and defect trapping.
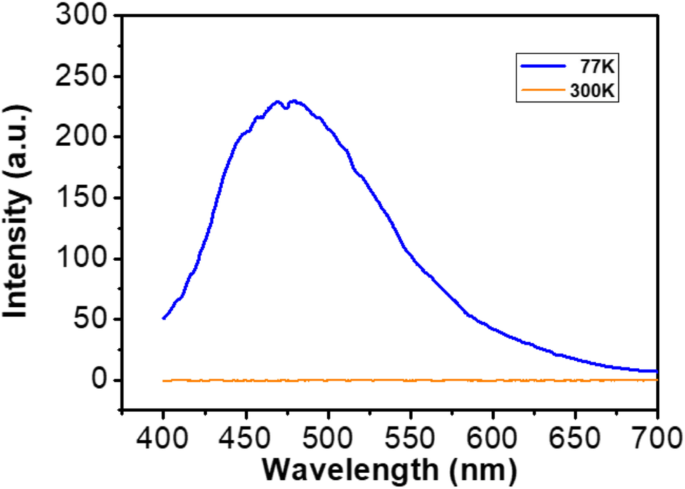
Emission spectra at 300 K and 77 K (λex = 327 nm) for LGN annealed at 800 °C.
UV‑vis absorption (Figure 8) reveals an onset at 320 nm, corresponding to a band‑gap energy of 3.95 eV. Minor absorption bumps near 320 nm at lower temperatures indicate oxygen‑vacancy defects, which diminish as the sintering temperature increases.
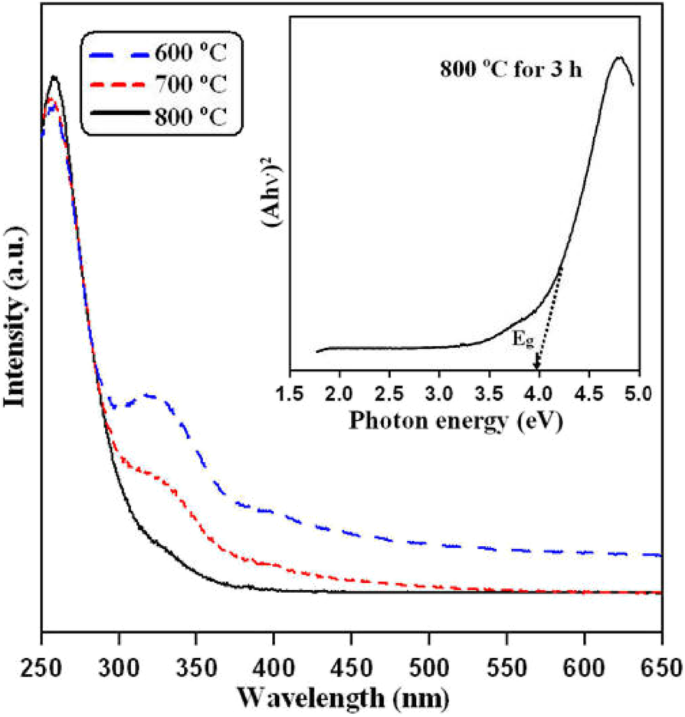
UV‑vis absorption of LGN powders annealed at 600–800 °C. Inset: (αhν)2 vs. hν plot showing Eg = 3.95 eV.
Conclusions
We successfully synthesized macroporous LGN ceramics via the Pechini route, achieving a single‑phase orthorhombic structure at 800 °C. The material exhibits a 327 nm excitation band arising from Nb5+–O2– charge transfer and a strong blue photoluminescence at 475 nm (77 K) linked to [NbO6]7–. The 800 °C sample shows a clear absorption edge at 320 nm, confirming a band gap of 3.95 eV and indicating minimal oxygen vacancies. The proposed growth mechanism highlights the transformation of nano‑foams into macroporous grains during sintering.
Availability of data and materials
All data and materials generated in this study are available to interested readers without restrictive transfer agreements.
Abbreviations
- BAW:
Bulk acoustic wave
- CA:
Citric acid
- CGC:
Calciumgallogermanate
- DTA:
Differential thermal analysis
- EDX:
Energy dispersive X‑ray analysis
- Eg:
Band gap
- EG:
Ethylene glycol
- FTIR:
Fourier‑transform infrared spectroscopy
- hν:
Photon energy
- PL:
Photoluminescence
- SAW:
Surface acoustic wave
- TEM:
Transmission electron microscopy
- TGA:
Thermogravimetry analysis
- UV‑vis:
Ultraviolet‑visible spectroscopy
- XRD:
X‑ray powder diffraction
Nanomaterials
- Hydrogen‑Assisted Atomic Rearrangement in GaN‑Based Multiple Quantum Wells: Enhancing Structural Uniformity and Optical Efficiency at 750 °C
- Tuning Electronic and Optical Anisotropy in Monolayer GaS via Vertical Electric Fields
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Electroless HF/AgNO3 Etching of n‑Si(100) Wafers: High‑Density Silicon Nanowire Arrays with Low Reflectance and Ohmic Conductance
- Tuning Morphology, Optics, and Conductivity of Al₂O₃/ZnO Nanolaminates Through Bilayer Thickness Control
- Enhanced Near‑Infrared Performance of Sulfur‑Hyperdoped Silicon Photodiodes Fabricated by Femtosecond Laser‑Induced Pulsed Melting
- Engineering n-Type GeBi Thin Films by Molecular Beam Epitaxy: Crystalline Control and Infrared/THz Optical Performance
- Alkali‑Metal‑Adsorbed Graphene‑Like GaN: Ultra‑Low Work Functions and Tunable Optoelectronic Properties
- High‑Performance Pr2CuO4 Nanosheet Adsorbent: Controlled Synthesis, Superior Selectivity for Malachite Green, and Mechanistic Insights
- Tailoring Electronic and Optical Properties of WSSe Bilayer via Strain Engineering



