Alkali‑Metal‑Adsorbed Graphene‑Like GaN: Ultra‑Low Work Functions and Tunable Optoelectronic Properties
Abstract
We employed density functional theory to explore how alkali‑metal adsorption alters the electronic and optical behavior of graphene‑like gallium nitride (g‑GaN). All examined systems proved thermodynamically stable, with the hexagonal ring center (TM) emerging as the most favorable adsorption site. Charge transfer from the alkali atom to the g‑GaN layer induces clear n‑type doping, while chemisorption dominates the binding mechanism. Remarkably, the work function drops dramatically upon adsorption; cesium‑functionalized g‑GaN reaches 0.84 eV, highlighting its promise for field‑emission devices. Alkali‑metal coverage also increases the static dielectric constant and broadens the absorption spectrum, offering a versatile route to tailor g‑GaN optoelectronics.
Background
Traditional bulk GaN, a wide‑bandgap semiconductor, supports operation at extreme voltages, frequencies, and temperatures, and boasts high luminous efficiency, excellent thermal conductivity, chemical resistance, and radiation tolerance. In the realm of two‑dimensional (2D) materials, g‑GaN has recently been synthesized via a migration‑enhanced encapsulated growth method and exhibits a widened bandgap along with enhanced optoelectronic performance compared to its bulk counterpart.
Surface adsorption studies are central to surface physics and nanodevice engineering. Alkali metals readily donate electrons, transforming semiconductors into n‑type materials, lowering their work functions, and modifying optoelectronic properties. Prior work has demonstrated work‑function reduction in graphene and other 2D systems upon alkali‑metal adsorption. However, the full photoelectric response of alkali‑metal‑functionalized g‑GaN remains unexplored.
This work systematically examines the band structure, density of states, work function, and optical response of pristine and alkali‑metal‑adsorbed g‑GaN, providing insights relevant to field‑emission and optoelectronic device fabrication.
Methods
All calculations were performed with the Vienna Ab initio Simulation Package (VASP) using density functional theory. Exchange‑correlation interactions were treated with the generalized gradient approximation (GGA) in the Perdew–Burke–Ernzerhof (PBE) form, known for reliable surface studies. A plane‑wave cutoff of 500 eV, a 20 Å vacuum gap normal to the g‑GaN plane, and a 9 × 9 × 1 Γ‑centered k‑point mesh ensured convergence. Geometry optimizations continued until forces fell below 10−4 eV/Å and total energy changes were <10−4 eV.
The adsorption energy (Eads) was calculated as:
Eads = Eg‑GaN:X − Eg‑GaN − μX
where μX is the chemical potential of an isolated alkali atom; a negative Eads indicates a stable chemisorption.
Charge‑density differences were evaluated via:
Δρ = ρT − ρg − ρx
with ρT, ρg, and ρx representing the total, pristine, and isolated‑atom charge densities, respectively.
Results and discussions
Figure 1 illustrates four potential adsorption sites: TN (above N), TGa (above Ga), TB (mid‑bond), and TM (hexagon center). Table 1 shows that all adsorption energies are negative, confirming exothermic chemisorption; the TM site is energetically favored, so subsequent analyses focus on this configuration.
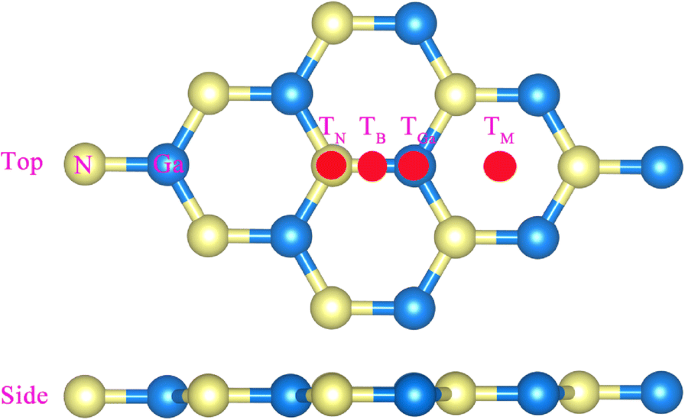
Model of g‑GaN with four adsorption sites.
Table 2 lists lattice constants, bond lengths, and adsorption heights. Pristine g‑GaN adopts a 3.254 Å lattice, in line with previous reports. Li/Na adsorption slightly contracts the lattice, whereas K/Rb/Cs expand it, reflecting the increasing alkali radius. Correspondingly, N–X and Ga–X bond lengths and adsorption heights rise with heavier alkali atoms.
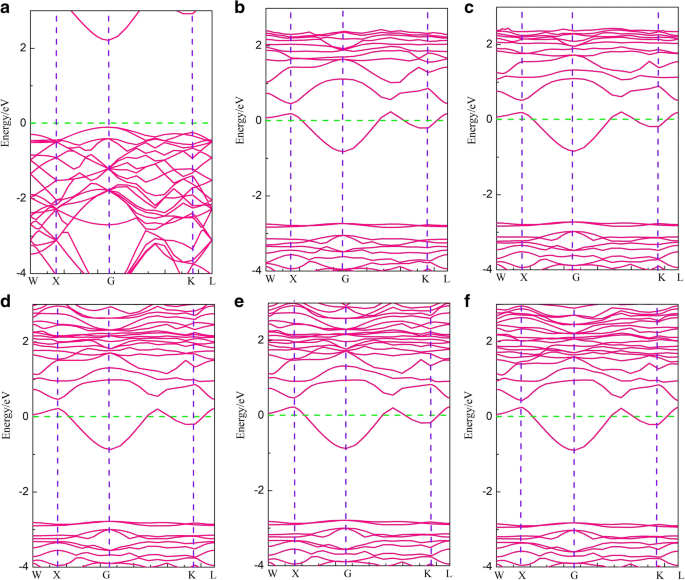
Band structures: a pristine g‑GaN; b–f Li, Na, K, Rb, Cs adsorbed. The pristine material is a semiconductor (2.1 eV gap). Alkali adsorption raises the Fermi level into the conduction band, imparting metallic character with a residual ~1.92 eV gap.
The density of states (Fig. 3) confirms semiconducting behavior for pristine g‑GaN, with valence‑band maxima dominated by N‑2p and Ga‑4p orbitals. In the adsorbed systems, states near the Fermi level arise mainly from Ga‑4s, N‑2p, and the alkali s orbitals.
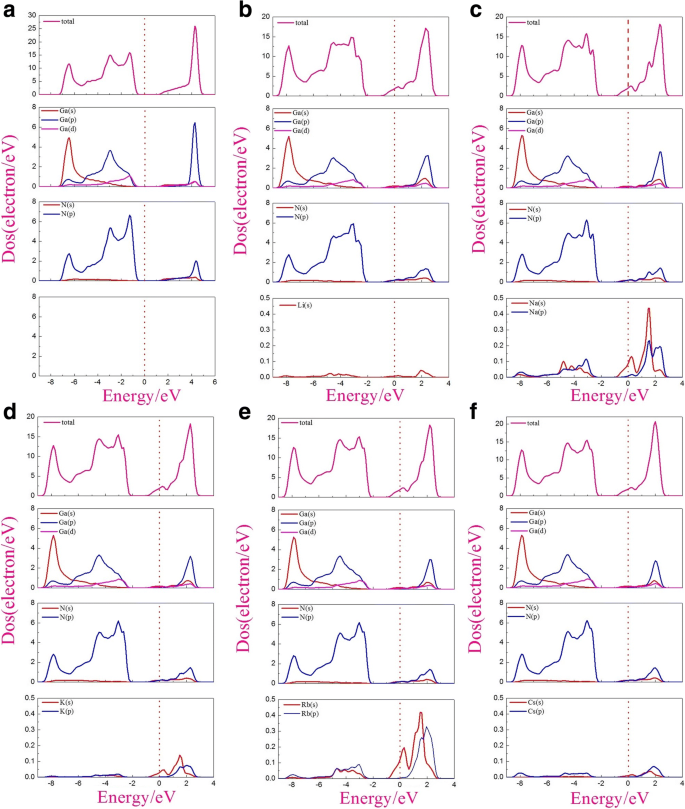
Density of states: a pristine, b–f Li, Na, K, Rb, Cs adsorbed.
Charge‑density difference maps (Fig. 4) reveal electron accumulation between the alkali atoms and the three neighboring N atoms, confirming chemisorption and ionic bonding. Bader analysis quantifies charge transfer: Li → 0.88 e, Na → 0.78 e, K → 0.80 e, Rb → 0.79 e, Cs → 0.79 e.
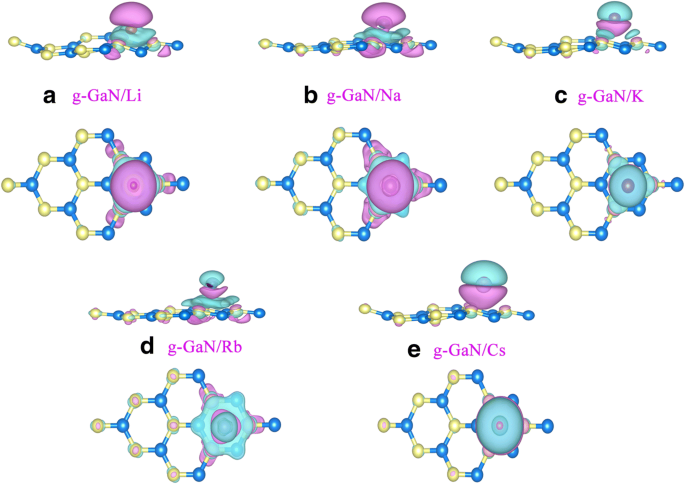
Charge‑density difference for g‑GaN/alkali. Magenta: electron gain; cyan: loss.
The work function drops from 4.21 eV (pristine) to 2.47 eV (Li) down to 0.84 eV (Cs), the lowest among the examined adsorbates. This reduction surpasses that observed in alkali‑metal‑adsorbed GaN nanowires, likely due to the distinct monolayer geometry, and positions Cs‑g‑GaN as an attractive candidate for field‑emission applications.
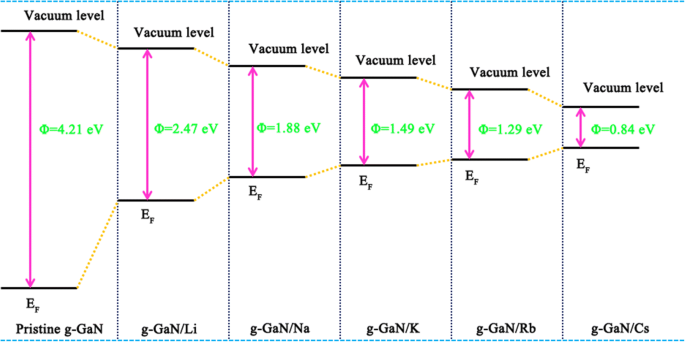
Work‑function schematic for pristine and alkali‑metal‑adsorbed g‑GaN.
Optical analysis shows that alkali adsorption markedly increases the static dielectric constant (ε1(0): 1.48 → 2.33–3.81) and shifts key absorption peaks to lower energies, extending the spectrum into the visible range. The absorption edge of pristine g‑GaN starts at 2.77 eV; after adsorption, a new low‑energy peak appears at 1.61 eV. Refractive indices also rise (n0: 1.22 → 1.53–1.99), and reflectivity peaks shift and diminish, indicating tunable light–matter interaction suitable for optoelectronic devices.
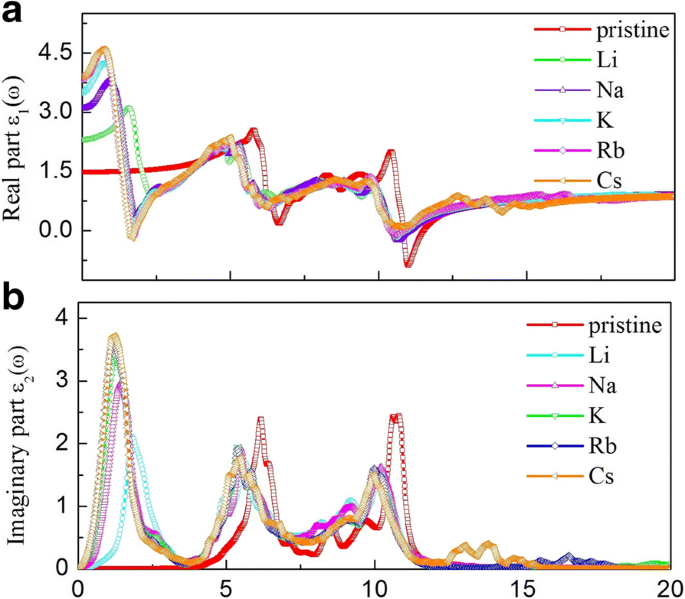
Real (a) and imaginary (b) dielectric functions for pristine and alkali‑adsorbed g‑GaN.
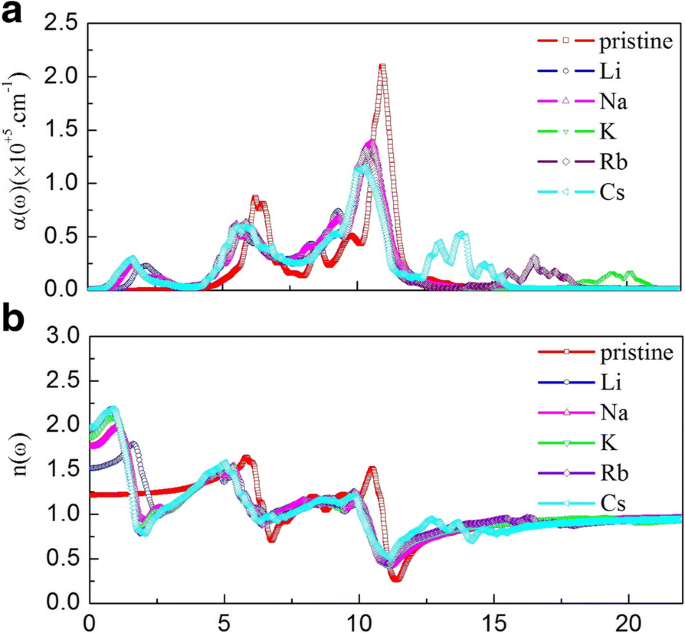
Absorption coefficient (a) and refractive index (b) for pristine and alkali‑adsorbed g‑GaN.
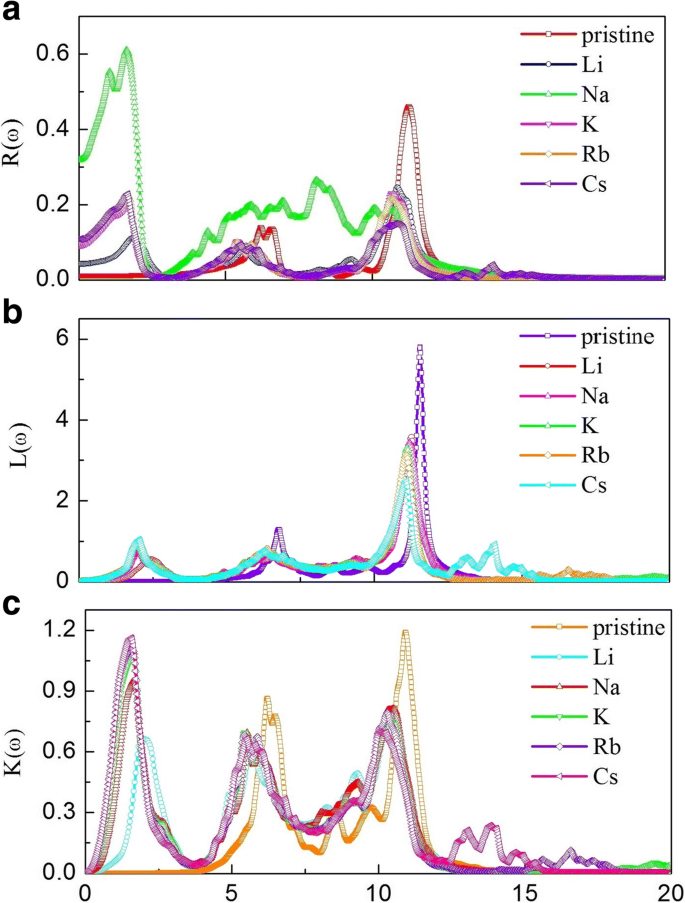
Reflectivity (a), loss‑energy (b), and extinction coefficient (c) for pristine and alkali‑adsorbed g‑GaN.
Conclusions
First‑principles calculations reveal that alkali‑metal adsorption on g‑GaN yields stable, chemisorbed structures with pronounced n‑type doping and dramatic work‑function reduction. Cesium‑functionalized g‑GaN attains a record low work function of 0.84 eV, making it highly suitable for field‑emission devices. Simultaneously, alkali adsorption enhances the static dielectric constant and broadens the absorption spectrum, offering a versatile strategy to engineer g‑GaN’s optoelectronic response for photodetectors, LEDs, and related technologies.
Abbreviations
- 2D
Two‑dimensional
- GGA
Generalized gradient approximation
- g‑GaN
Graphene‑like gallium nitride
- PBE
Perdew–Burke–Ernzerhof
- PDOS
Partial density of states
- TDOS
Total density of states
Nanomaterials
- Tuning Electronic and Optical Anisotropy in Monolayer GaS via Vertical Electric Fields
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Electroless HF/AgNO3 Etching of n‑Si(100) Wafers: High‑Density Silicon Nanowire Arrays with Low Reflectance and Ohmic Conductance
- Tuning Morphology, Optics, and Conductivity of Al₂O₃/ZnO Nanolaminates Through Bilayer Thickness Control
- Enhanced Near‑Infrared Performance of Sulfur‑Hyperdoped Silicon Photodiodes Fabricated by Femtosecond Laser‑Induced Pulsed Melting
- Engineering n-Type GeBi Thin Films by Molecular Beam Epitaxy: Crystalline Control and Infrared/THz Optical Performance
- Defect‑Driven Magnetism and Strain Engineering in Monolayer WSe2: A DFT Study
- Optical Properties and Growth Mechanism of Macroporous La₃Ga₅.₅Nb₀.₅O₁₄ Ceramic Synthesized via Pechini Process
- Tailoring Electronic and Optical Properties of WSSe Bilayer via Strain Engineering
- Raman Analysis of G and D′ Phonon Shifts in Vacancy-Engineered Monolayer to Few-Layer Graphene Across 78–318 K



