Enhancing Energetic Performance by Integrating Al/PTFE Nanolaminates into Copper Exploding Foils
Abstract
Micro‑scale energy devices, such as electric initiation systems, demand compact, high‑energy, and low‑ignition‑temperature materials. In this study, we developed a reactive Al/PTFE nanolaminate by alternating magnetron‑sputtered aluminum (Al) and polytetrafluoroethylene (PTFE) layers, creating a metastable fuel–oxidant–inert system. The resulting film demonstrates a remarkable onset temperature of 410 °C and releases 3,034 J g⁻¹ of heat. We integrated this nanolaminate with a copper (Cu) exploding foil to form a film bridge that exhibits a more violent explosion, greater mass ejection, and higher plasma temperature than a conventional Cu bridge alone. The flyer kinetic energy increased by ~30 % thanks to the additional chemical energy. These findings confirm that coupling Al/PTFE nanolaminates with Cu exploding foils significantly boosts electric‑initiator performance while maintaining a microscale footprint.
Background
Nanostructured energetic materials have attracted global attention over the past decade due to their low ignition temperatures, rapid energy release, high energy density, and tunable reactivity 1‑10. Their stored chemical energy can be triggered by electrical, optical, mechanical, or thermal stimuli, enabling diverse applications ranging from military initiators to automotive air‑bag propellants 11‑14. Fabrication techniques include nanopowder mixing, reactive milling, electrophoretic coating, and periodic nanolaminate deposition 15‑19. Nanolaminates, produced by sequentially depositing two or more films, offer precise control over layer thickness and count, allowing tailored energetic responses.
Exploding foil initiators (EFIs) convert electrical energy into a high‑pressure plasma that accelerates a flyer to detonate secondary explosives 20. As EFIs become smaller and more efficient, integrating nanoenergetic layers onto the metal bridge via MEMS technology offers a promising route to enhance performance while keeping device dimensions minimal. The additional chemical heat from energetic layers can amplify the Joule heating of the bridge, leading to higher plasma temperatures and improved flyer acceleration.
Al/PTFE nanolaminates are particularly attractive for EFI integration. Aluminum offers a high energy density when oxidized, while PTFE’s high fluorine content can react with Al to form AlF₃, releasing up to 5,571 J g⁻¹ theoretically 21. The decomposition of PTFE also generates gases that further pressurize the plasma, aiding flyer launch 22. This paper reports the design, fabrication, and testing of a Cu‑based film bridge incorporating Al/PTFE nanolaminates, and evaluates its electric‑initiation performance.
Methods
Deposition of the Al/PTFE Nanolaminates
Al/PTFE nanolaminates were deposited on alumina substrates by alternating DC magnetron sputtering of Al (purity > 99.999 %) and RF magnetron sputtering of PTFE (purity > 99.99 %). A rotating substrate table enabled multiple sequential layers. Deposition was performed under a base pressure below 5 × 10⁻⁴ Pa using argon as the sputtering gas. Optimized conditions were 1.1 Pa and 300 W for PTFE, and 0.45 Pa and 100 W for Al, yielding stable growth rates and high‑quality films.
Preparation of the (Al/PTFE)ₙ/Cu‑Integrated EFI
The integrated film bridge was fabricated on a 3‑inch alumina ceramic substrate. The process flow is illustrated in Fig. 1. Each unit consists of a Cu exploding bridge, a rectangular Al/PTFE nanolaminate on top, and Cu contact pads on either side.
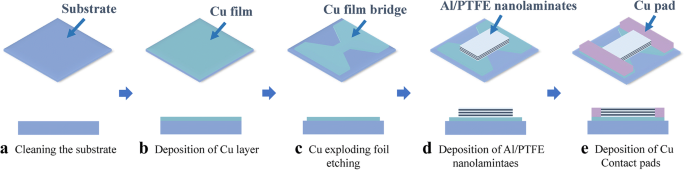
Schematic drawing and fabrication process flow of the (Al/PTFE)ₙ/Cu film bridge
Substrates were cleaned sequentially with acetone, isopropanol, and deionized water for 10 min, then dried in argon and baked at 120 °C for 1 h. A 2‑µm Cu layer was sputtered, patterned by photolithography, and wet‑etched using Cu etchant (CE‑100). The Cu bridge measured 600 µm × 600 µm. A ~2‑µm Al/PTFE nanolaminate (Al/PTFE/Al/PTFE/Al) was then deposited and lift‑off patterned using an image‑reversal process, leaving Al as the topmost layer. Finally, Cu contact pads were added to enable electrical connection. Individual units were diced from the wafer.
Characterization of the Al/PTFE Nanolaminates
Cross‑sectional structure was examined by TEM. To probe the Al/ PTFE interface, a ~1‑nm Al overlayer was sputtered onto PTFE and analyzed by XPS. Energy release was quantified by differential scanning calorimetry (DSC) from 25 °C to 800 °C at 10 °C min⁻¹ under flowing argon; sample mass was ~10 mg.
Electric Explosion Test of the Film Bridge
Electric‑explosion characteristics were measured using a custom system based on the Cu/Al/CuO bridge methodology 23. Plasma temperature was inferred from double‑line copper emission spectroscopy 24,25. High‑speed imaging (20,000 fps) captured the explosion dynamics, while photonic Doppler velocimetry (PDV) measured flyer acceleration.
Results and Discussion
Characterization of the Al/PTFE Nanolaminates
Figure 2a shows a clear cross‑section with alternating dark (Al) and bright (PTFE) layers. Each Al and PTFE monolayer measures ~50 nm and ~75 nm, respectively. High‑resolution images (Fig. 2b,c) reveal a nano‑polycrystalline Al lattice and an amorphous PTFE matrix. Electron diffraction confirms these microstructural features, underscoring the engineered interfacial diffusion pathways that drive energetic release. The uniform thickness allows precise tuning of layer counts to optimize performance.

a Cross‑sectional bright‑field TEM image of the Al/PTFE nanolaminates.b High‑resolution image of the Al layer and its electron diffraction pattern.c High‑resolution image of the PTFE layer and its electron diffraction pattern.
XPS analysis (Fig. 3) confirms the formation of Al–F bonds at the interface. Al 2p peaks at 72.2 eV indicate metallic Al, while additional peaks at ~75 eV correspond to oxidized Al, slightly shifted due to interfacial reactions. The F 1s peak at 686.6 eV matches AlF₃, evidencing initial chemical interaction during deposition. These results support the metastable reaction system comprising fuel (Al), oxidant (PTFE), and inert Al‑F layers that restrains premature decomposition while preserving high energy density 26,27.
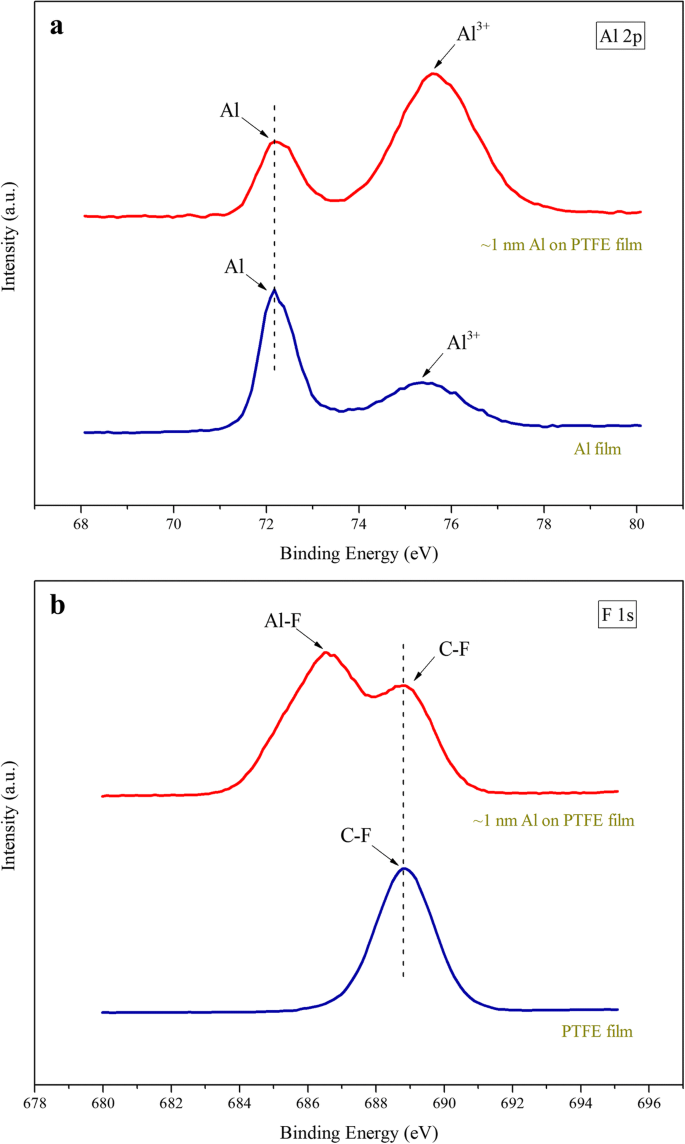
a Al 2p core‑level spectra of Al film and PTFE film with a ~1‑nm Al overlayer.b F 1s core‑level spectra of PTFE film before and after Al deposition.
DSC curves (Fig. 4) display a sharp exothermic peak at 507 °C, with an onset at 410 °C. Integrating the heat flow yields 3,034 J g⁻¹, a substantial fraction of the theoretical 5,571 J g⁻¹, indicating efficient but incomplete reaction likely due to the Al‑F barrier. Nonetheless, the low ignition temperature and high heat release make these nanolaminates suitable for EFI integration.
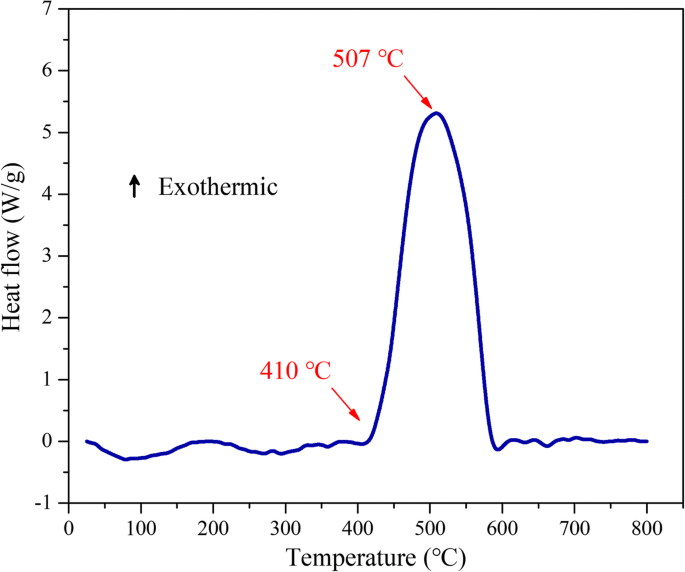
DSC curves of the Al/PTFE nanolaminates as a function of temperature in argon environment.
Electric Initiation Performances of the (Al/PTFE)ₙ/Cu Film Bridges
High‑speed footage (Fig. 5) reveals that the Cu bridge alone undergoes a rapid 250 µs explosion, while the integrated (Al/PTFE)ₙ/Cu bridge exhibits a prolonged 500 µs event with more vigorous plasma and larger ejected mass. This indicates that the nanolaminate’s chemical energy enhances the bridge’s ionization and plasma expansion.

High‑speed camera observation of the electric explosion processes for the Cu film bridge (a) and the (Al/PTFE)ₙ/Cu film bridge (b) at a 2,500‑V discharge voltage.
Plasma temperature, derived from copper emission ratios, peaks at 6,819 K for the Cu bridge and 8,289 K for the integrated bridge (Fig. 6). The ~1,470 K temperature increase reflects the significant heat released by the Al/PTFE reaction, facilitating faster ionization and a more energetic plasma.
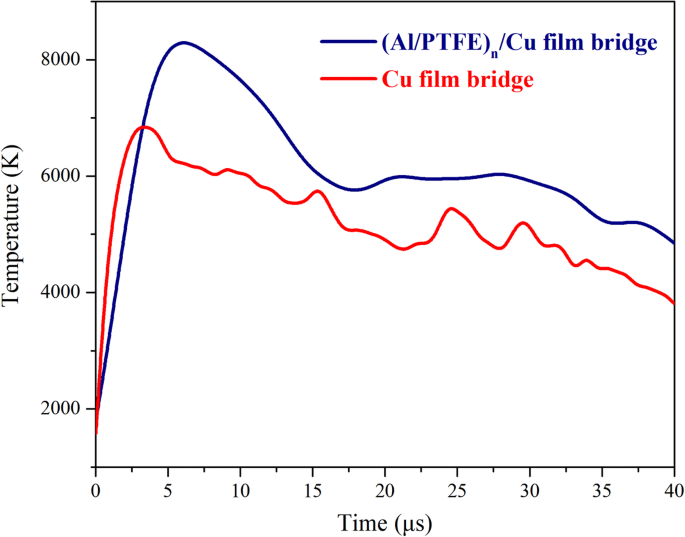
The temperature variation curves after data processing during the electric explosion process for the Cu film bridge and the (Al/PTFE)ₙ/Cu film bridge at a 2,500‑V discharge voltage.
Flyer velocity profiles (Fig. 7) show a peak of 2,792 m s⁻¹ for the Cu bridge and 3,180 m s⁻¹ for the integrated bridge, translating to a 29.9 % increase in kinetic energy. The launch timing is comparable, confirming that the Al/PTFE chemistry synchronizes well with the Cu bridge’s electric explosion.
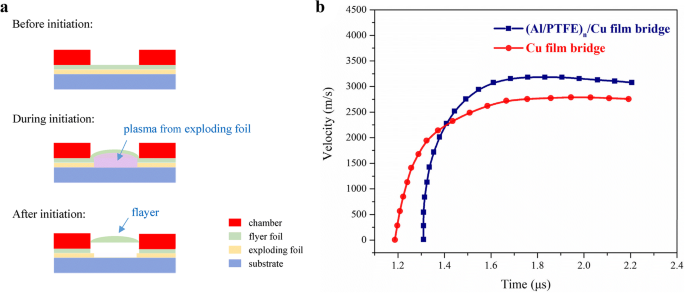
a Schematic illustration side view of EFI operation in electric initiation process.b The velocity variation curves reconstructed from PDV signal during the electric initiation processes for the Cu film bridge and the (Al/PTFE)ₙ/Cu film bridge at a 2,500‑V discharge voltage.
Conclusions
We successfully fabricated Al/PTFE nanolaminates via magnetron sputtering, achieving a 3,034 J g⁻¹ heat release and a 410 °C ignition temperature. By integrating these nanolaminates with a Cu exploding foil through MEMS fabrication, we created a compact film bridge that combines the rapid electric initiation of Cu with the additional chemical energy of Al/PTFE. The resulting device exhibited higher plasma temperatures, more vigorous explosions, and ~30 % increased flyer kinetic energy compared to a bare Cu bridge. These results demonstrate that Al/PTFE nanolaminates can substantially enhance EFI performance while retaining a microscale form factor.
Abbreviations
- Al:
Aluminum
- Cu:
Cuprum
- DSC:
Differential scanning calorimetry
- EFI:
Exploding foil initiator
- MEMS:
Microelectronic and mechanical systems
- NOC:
Nanoenergetic-on-a-chip
- PTFE:
Polytetrafluoroethylene
- TEM:
Transmission electron microscopy
- XPS:
X‑ray photoelectron spectroscopy
Nanomaterials
- The Emerging Role of IoT in the Energy Sector: Key Use Cases and Benefits
- Integrating Sensor Data into a Raspberry Pi: A Hands‑On Guide
- Synergistic Electrical Enhancement in Epoxy Composites with Carbon Nanotubes and Graphite Nanoplatelets
- Significantly Enhanced Photocurrent in High‑Conductance Topological Insulator Nanosheets
- Energetic Al/Ni Superlattice Enables High‑Speed Micro‑Plasma Generation with Enhanced Flyer Velocities
- Significant Boost of Light-Induced Transverse Thermoelectric Effect in Tilted BiCuSeO Films Using Ultra‑Thin Gold Nanoparticle Layers
- Monoclinic V1−x−yTixRuyO₂ Thin Films: Superior Thermal Sensitivity for Microbolometers
- High‑Efficiency 22%+ Perovskite Solar Cells Using Ultra‑Compact FA0.1MA0.9PbI3 Films
- 6 Powerful Advantages of IoT Transforming the Energy Sector
- Seamless Integration of Renewable Energy into the Power Grid



