Drain Cleaners: Types, Production, Safety, and Environmental Impact
Background
Drain cleaners—also known as drain uncloggers—are specialized solutions used to dissolve organic blockages such as hair, waste, and food particles in kitchen sinks, tubs, and shower drains. In the United States, chemical cleaners dominate the market because of their superior effectiveness, while bio‑degradable, enzyme‑based alternatives are gaining traction for their lower environmental footprint.
Types of Drain Cleaners
- Acid‑Based Cleaners – Typically formulated with sulfuric acid (H₂SO₄) and proprietary additives. These products offer rapid, powerful removal of organic material but pose significant safety risks due to their corrosive nature.
- Alkaline Cleaners – Made primarily from sodium hypochlorite (NaClO) and surfactants. They are less aggressive than acid cleaners, cheaper to produce, and generally safer for household use, though still hazardous if mishandled.
- Biodegradable Cleaners – Enzyme or bacteria‑based formulations that soften and break down sludge. While safer for plumbing systems and the environment, they typically provide slower, less dramatic unclogging performance.
Safety Considerations
Manufacturers universally recommend wearing gloves and eye protection when handling chemical drain cleaners. These substances can cause severe skin burns, eye irritation, and respiratory distress if splashed or inhaled. Mixing drain cleaners with other household chemicals may produce toxic fumes, so products should be stored separately and kept out of reach of children.
Raw Materials
Acidic cleaners rely on sulfuric acid, a byproduct of iron ore processing that is predominantly sourced from Canada. The acid is typically blended with proprietary stabilizers to moderate its strength while maintaining effectiveness. Alkaline cleaners use sodium hypochlorite—produced by combining chlorine, sodium hydroxide, and water—augmented with surfactants that reduce surface tension and improve foaming control.
Manufacturing Process
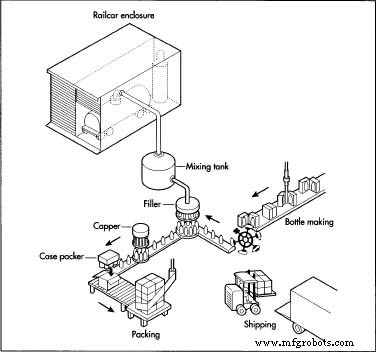
- Large bulk tanks receive sulfuric acid via rail or tanker trucks. The tanks are enclosed to contain fumes and protect operators from direct contact.
- Acid is transferred to a dedicated mixing vessel where it is diluted with controlled amounts of additives. The precise formulation is proprietary, varying by manufacturer.
- The mixture is homogenized to ensure uniform distribution of active ingredients.
- The finished solution is routed to a filler line that dispenses predetermined volumes into thick‑polyethylene bottles. Each bottle is sealed and capped automatically to prevent leakage.
- Alkaline cleaners follow a similar workflow, with additional steps to incorporate surfactants and ensure the final product meets safety and performance specifications.
Quality Control
Manufacturers rely on certified suppliers to provide raw materials that meet exacting grade and strength requirements. Batch testing—conducted in controlled environments—confirms that each product batch matches its label claims. Maintenance of hoses, fillers, and storage tanks is critical to prevent accidental leaks of corrosive chemicals.
Byproducts and Waste Management
Drain cleaner production is highly efficient, with negligible waste. However, consumer disposal remains a concern. Unused products should be taken to hazardous waste facilities; if unavailable, they must be diluted with copious water before being poured down a drain, never into a septic system. Empty containers, contaminated with acidic or alkaline residues, should be disposed of in regular trash rather than recycled.
The Future of Drain Cleaning
Environmental advocacy is driving a shift away from sulfuric acid and sodium hypochlorite, especially in regions where wastewater discharges into natural water bodies. Enzyme‑based cleaners that break down grease, food particles, and hair without harming ecosystems are increasingly promoted. Although these alternatives are safer for plumbing and the environment, their performance as aggressive uncloggers is limited, so many consumers still prefer traditional chemical solutions for quick relief.
Where to Learn More
Books
Ierley, Merritt. Open House: A Guided Tour of the American Home. New York: Henry Holt and Company, 1999.
Ierley, Merritt. The Comforts of Home. New York: Clarkson Potter, 1999.
Rybczynski, Witold. Home: A Short History of an Idea. New York: Penguin Books, 1986.
Other
Clorox Web Page. December 2001. https://www.clorox.com
Oral interview with Dennis West, Plant Manager of Rooto Corporation. Howell, MI. October 2001.
“Surfactants.” Encyclopedia Britannica CD Edition. Encyclopedia Britannica Inc., 1994-1998.
Nancy EV Bryk
Manufacturing process
- Junction Field‑Effect Transistors (JFET): Design, Operation, and Advanced Variants
- The Evolution of Vacuum Cleaners: From 19th-Century Sweepers to Modern Allergy‑Focused Models
- Parasitic Battery Drain: Symptoms, Causes, and How to Fix It
- Extending Oil Drain Intervals Safely: The Role of Base Number vs. Acid Number Analysis
- Choosing the Right Cleaner: A Practical Guide to Safety and Effectiveness
- Understanding Drain Valves: Function, Types, and Applications
- Enhancing Workplace Safety: Oil Mist Air Cleaners for a Cleaner, Healthier Cutting Environment
- Oil Mist Air Cleaners: Safeguarding Workplace Air Quality & Worker Health
- Essential Guide to Chip Vacuum Cleaners for Industrial Safety
- What Is a Case Drain in a Hydraulic Pump? Essential Function & Maintenance



