Antibacterial Soap: Composition, Manufacturing, and Regulatory Landscape
Antibacterial soap is a specially formulated cleanser designed to eliminate germs on skin and surfaces. Available in liquid or bar form, it combines detergent ingredients with antimicrobial agents to provide reliable hygiene.
Background
Initially marketed as deodorant soaps to control body odor caused by bacteria on sweat, antibacterial bars gained prominence in the 1950s with brands such as Dial and Lifebuoy. While many of these bar soaps remain on the market, liquid antibacterial hand washes have surged in popularity. Leading consumer brands include Lever’s Caress, Dove’s Liquid, and Procter & Gamble’s Oil of Olay. Professional healthcare products—surgical scrubs, wound disinfectants, and wound cleansers—also rely on antibacterial formulations.
In the United States, all antibacterial soaps that make germ‑killing claims are classified as over‑the‑counter (OTC) drugs. This status allows them to be sold without a prescription, provided they meet FDA guidelines outlined in a monograph. While the FDA has not issued a final monograph, a series of Tentative Final Monographs (TFMs) has been published since 1974. These documents define antibacterial cleansers and categorize them into seven groups, ranging from consumer antimicrobial soaps to healthcare hand washes and surgical scrubs.
Regulatory history reveals significant shifts: the 1994 TFM removed a separate regulatory path for consumer antibacterial soaps, aligning them with professional products in terms of active ingredients and allowed claims. Consequently, consumer soaps must use the same active compounds as healthcare products, which may not fully align with their typical usage patterns. Industry experts expect a final monograph to be issued in the near future, potentially reshaping the market.
Annual sales of antimicrobial products in the U.S. reached $600 million, driven largely by liquid hand cleansers. Traditional antibacterial deodorant bars have largely been supplanted by antiperspirants/deodorants, underscoring the shift toward liquid formulations.
Design
Formulating an antibacterial soap requires balancing antimicrobial efficacy with user comfort. Key considerations include target pathogens, contact time, foam characteristics, rinsability, and skin feel. Aesthetic attributes—appearance and fragrance—are also critical for consumer acceptance.
The chemist develops a comprehensive recipe, specifies manufacturing procedures, and establishes product specifications to ensure consistency and quality.
Raw Materials
Water, typically deionized or distilled, constitutes 40–80% of the formula and serves as the carrier for other ingredients. Hard‑water ions can interfere with detergents, making purified water essential.
Two antibacterial agents dominate current formulations:
- Trichlocarban (3,4,4′‑trichlorocarbanilide) – commonly used in bar soaps, effective at 0.5%.
- Triclosan (2‑hydroxy‑2′,4,4′‑trichlorodiphenyl ether) – widely used in liquid soaps, also effective at 0.5%.
Both agents denature microbial cell contents or disrupt metabolism, providing broad‑spectrum activity.
Surfactants, the primary cleansing agents, create foam and solubilize oils. Primary surfactants (e.g., alkyl sulfates, ether sulfates, olefin sulfonates, amphoteric surfactants) typically compose 20–40% of the formula. Secondary surfactants—amides, betaines, sultaines, and alkyl polyglucosides—fine‑tune foam quality and skin feel, usually at 1–10%.
Additional ingredients adjust viscosity, fragrance, color, and appearance:
- Thickeners (gums, starches, polymers) at 0.1–1% improve flow.
- Fragrances at 0.1–1% mask base odors and enhance appeal. Fragrances may comprise dozens of components and must be compatible with the detergent base.
- Colorants (FDA‑approved D&C or FD&C dyes) at <0.01% enhance visual appeal.
- Pearlizing agents (glycol stearate, titanium‑coated mica) at ≤1% give a pearled look.
- Preservatives at ≤1% protect liquid soaps from mold and bacterial growth, supplementing the primary antimicrobial agents.
- Botanical extracts, proteins, and natural oils may be added for added consumer appeal.
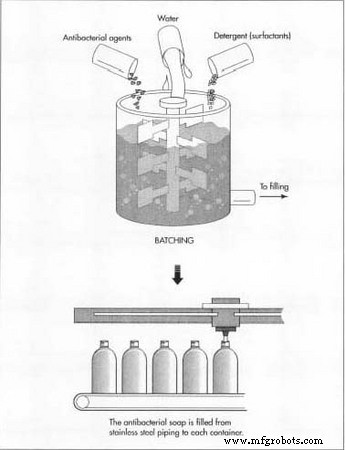
The first ingredient added to the tank is typically water because it is usually the most plentiful ingredient. The other ingredients are added to the tank as specified by the manufacturing procedure. Ingredients that are heat sensitive are added as the batch is cooled to room temperature.

The Manufacturing Process
Preparatory Steps
- Prior to manufacturing, each ingredient is tested for quality. Approved components are pre‑weighed and stored near the batching area. Equipment—tanks, plumbing, pumps, and transfer vessels—are cleaned and sanitized to prevent microbial contamination and chemical residue.
Batching
- Liquid antibacterial soaps are produced in stainless steel tanks, often up to 3,000 gal (24,000 lb). Mixing is performed with propeller or sweep‑style mixers to provide efficient agitation without excessive aeration. Heating or cooling is achieved via a jacketed tank with steam or chilled water. Automated systems control mixing speed and temperature for consistent quality.
- The first ingredient—water—is added, followed by other components as per the procedure. Heat‑sensitive ingredients are introduced once the mixture reaches room temperature. The batch is held at a controlled temperature for a specified duration to ensure homogeneity. A sample is then analyzed to confirm it meets specifications before release.
Filling and Packaging
- After approval, the product is pumped directly to a conveyor line equipped with filling equipment or transferred to a temporary storage tank for later filling. Nozzles dispense a measured volume into plastic bottles, which are then sealed with screw caps or dispensing pumps. Bottles receive lot numbers and expiration dates via ink‑jet printing, then are packed into cardboard cartons for shipment.
Quality Control
Quality assurance is integral to antibacterial soap manufacturing. Prior to market release, formulas undergo stability testing to verify that functional and aesthetic properties remain intact over a projected three‑year shelf life. During production, each ingredient is chemically tested before batching. Post‑batching, the product is evaluated for correct ingredient proportions, physical properties (viscosity, pH), and antibacterial activity. The glove‑juice test samples sweat from a volunteer’s hand after using the soap; the sample is cultured on a specialized medium to detect microbial growth. Minimal or no growth indicates effective antibacterial performance.
Byproducts and Waste
Incorrect batches can generate significant waste. However, many can be corrected: adjustments to ingredient levels or dilution can bring them within specifications. Contaminated batches must be disposed of in compliance with local, state, and federal regulations, as adulterated OTC drugs cannot be sold.
The Future
Future antibacterial soaps will evolve through both chemical innovation and regulatory change. New surfactants and additives promise better foaming, gentler cleansing, improved biodegradability, and cost savings. Regulatory clarity hinges on the FDA’s final monograph, which is not expected for the next few years. Once finalized, products may require reformulation to meet updated standards.
Manufacturing process
- Why Rapid Prototyping is Essential for Successful Product Development
- Soap: From Ancient Cleansing to Modern Manufacturing
- Laundry Detergent: History, Chemistry, and Production Processes
- Shaving Cream: History, Ingredients, and Modern Manufacturing
- Driving Innovation at Mar‑Bal: Turning Disruptive Change into Market‑Defining Success
- Industries We Serve: Expertise in Food, Defense, Automotive, Mining, Agriculture, and Energy
- Carbolic Soap Explained: History, Uses, and Disinfectant Power
- Copper-Infused Antibacterial Stainless Steel: Enhanced Safety & Corrosion Resistance
- Investment Casting: How Metal Products Are Made
- Modern Ceramic Production: From Raw Materials to Finished Goods



