Laundry Detergent: History, Chemistry, and Production Processes
Background
The first soaps were produced in antiquity by boiling fats and ashes. Archaeological digs in ancient Babylon uncovered evidence that soap was used as far back as 2800 B.C.. By the second century A.D., Romans were regularly making soap, likely having started earlier.
During the Middle Ages, soap use fell in Europe, but the fifteenth century saw a resurgence. Olive‑oil based soap from Castile, Spain, reached many parts of the world and remains a benchmark of quality today.
In the colonial era and the eighteenth century, Americans often made soap at home. This home production continued until the 1930s, when the industry shifted to large‑scale manufacturing. Germany first produced a synthetic detergent during World War I. The 1946 breakthrough combined a surfactant (a surface‑acting agent) and a builder (a chemical that enhances surfactant performance). Post‑war economic growth and the advent of inexpensive washing machines propelled detergent sales; by 1953, detergent sales surpassed soap sales in the United States.
Raw Materials
While commonly called “soap,” laundry detergent is a synthetic blend that functions similarly to soap but offers key advantages. A soap molecule features a hydrocarbon tail and a carboxylic acid head. The hydrophilic head attracts water, and the hydrophobic tail binds to oil and grease. This dual nature lifts dirt from fabric and carries it into wash water, aided by agitation and rinsing.
The problem with soap in hard water—rich in calcium, magnesium, iron, and manganese—is the formation of insoluble precipitates. These deposits are hard to rinse, leave visible stains, and stiffen fabric. Even softer water eventually produces precipitates over time.
Detergent surfactants are often derived from crude oil. Sulfuric acid reacts with the processed hydrocarbon to produce a fatty‑acid‑like molecule, and an alkali converts it into a surfactant that does not bind to hard‑water minerals, preventing precipitates.
Builders are a core component of modern detergents. They boost surfactant efficiency, sequester hard‑water minerals, emulsify oil and grease, and protect washing machines from corrosion. Common builders like sodium silicate also help maintain the chemical balance of wash water.
Other ingredients include antiredeposition agents (preventing soil from re‑settling), fluorescent whitening agents (convert UV light to visible blue light for brightness), oxygen bleaches such as sodium perborate (enhance detergency, especially in phosphate‑free formulas), and processing aids like sodium sulfate (prevent caking and standardize density).
Enzymes (proteins that break down stains) and fragrances are standard. Suds‑control agents ensure that excessive foam does not damage the machine.
The Manufacturing Process
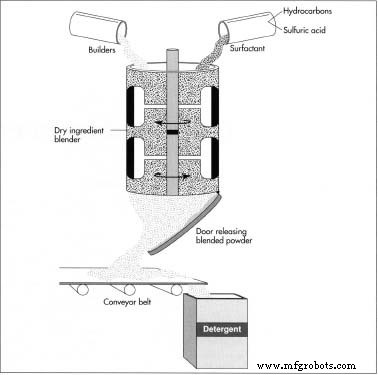
Three methods exist for dry detergent production, but two dominate the industry. The blender process—favored by smaller firms—mixes ingredients in large vats and then packages the blend. Blender capacities range from 500 to 10,000 pounds (227–4,540 kg), ideal for batch production.
The agglomeration process is continuous and preferred by large manufacturers, producing 15,000–50,000 pounds (6,800–22,700 kg) per hour. The third method, involving wet blending and hot‑air drying, is largely obsolete due to high fuel costs and engineering challenges.
The blender process
- Ingredients are loaded into a tumbling or ribbon blender. The tumbling blender rotates and shakes, while the ribbon blender uses internal blades to mix.
- After mixing, the mixture flows through a bottom outlet onto a conveyor belt, where it is transferred to packaging stations for boxes or cartons.
The agglomeration process
- Dry ingredients are fed into a Shuggi agglomerator, where high‑speed blades create a fine, homogeneous mixture.
- Liquid ingredients are sprayed onto the dry mix; the blend heats exothermically, forming a hot, viscous gelatin‑like liquid.
- The liquid exits onto a drying belt, where it cools, aerates, and breaks into friable granules.
- Granules are pulverized and screened to remove lumps, producing a uniform dry detergent.
The slurry method
- Ingredients are dissolved in water to form a slurry, which is pumped through nozzles in a cone‑shaped chamber. Simultaneous hot, dry air is introduced from below.
- As the slurry dries, bead‑like dry detergent falls to the bottom of the cone, ready for collection and packaging.
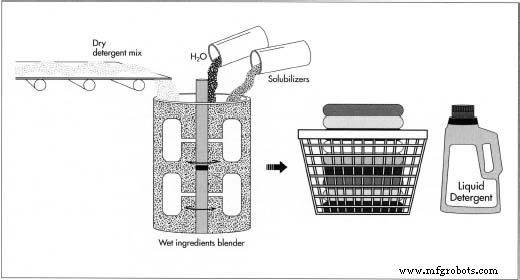
Liquid detergent
- To produce liquid detergent, the dry powder is re‑mixed with water and solubilizers—chemicals that promote even dispersion.
Quality Control
Manufacturers employ rigorous testing to ensure detergent performance. One method shines light onto a soiled fabric washed with the test detergent. The reflected light intensity, compared to that from a pristine fabric, measures cleanliness. A 98 % reflection rate indicates excellent cleaning.
Another test involves burning a small amount of laundered, soiled fabric in the lab. The weight of ash and combustion gases reveals residual dirt. Results close to a clean control sample confirm effective cleaning.
Byproducts and Environmental Impact
Two major environmental concerns have shaped detergent development. Phosphate builders once contributed large amounts of phosphorous to waterways, fueling algal blooms that deplete dissolved oxygen and threaten aquatic ecosystems. Legislative pressure in the 1960s spurred research into phosphate‑free builders, which are now standard in many states.
Foam pollution was another issue. In the 1950s, excessive foam from surfactants like ABS (alkyl benzene sulfonate) appeared in rivers. ABS’s complex structure slowed biodegradation, leading to foam buildup. Manufacturers replaced ABS with LAS (linear alkylate sulfonate), which biodegrades rapidly and has since become the primary foaming agent.
Manufacturing process
- How Lawn Sprinklers Work: Design, History, and Modern Manufacturing
- Dishwasher Technology: History, Design, and Future Innovations
- The Evolution of Pump‑Action Water Guns: From Invention to Global Market Leader
- The Evolution, Materials, and Manufacturing of Modern Toilets
- Antibacterial Soap: Composition, Manufacturing, and Regulatory Landscape
- Fire Hydrants: Design, Manufacturing, and Their Role in Urban Fire Safety
- Water: History, Types, and Modern Treatment Processes
- The Science Behind Shampoo: Ingredients, Manufacturing, and Future Trends
- Salt: Production, Uses, and Health Impact
- Soap: From Ancient Cleansing to Modern Manufacturing



