Aluminum Types Explained: Classifications, Properties, and Practical Applications
How to choose proper types of aluminum for different casting applications? Here we specify aluminum classifications, their properties, and a guide to select aluminum for a range of casting uses.
Aluminum is one of the most abundant elements on the Earth and widely used in many different industrial and non-industrial applications because of its good properties and versatility. From shipbuilding, automotive, aviation, decorative parts, outdoor furniture to kitchen accessories, we all see aluminum applications.
Aluminum varies in many types. Depending on the specific uses, there will be suitable types of aluminum with their particular properties. With different properties and chemical composition, each type of aluminum will behave differently in the process.
Therefore, it is very important to classify aluminum to understand the properties of each type to help the manufacturer choose the proper material for casting that meets the desired quality and customer’s requirement.
Grab this demand of knowledge, we open an article to discuss how to classify basic and common different types of aluminum and aluminum alloys go along with their characteristic properties and main applications.
Stay with us to dig out the answer!
Aluminum
Aluminum is considered as one of three most common elements on Earth, just place behind Oxygen and Sillic.
The chemical symbol of Aluminum is Al; Mass atom is 27 with atomic number of 13.
Although aluminum is quite common in nature, it is rarely found in its pure form. Instead, aluminum is often found in substances such as clay, bauxite, etc. Statistically, aluminum is found in about 270 different types of minerals.
General Physical Properties of Aluminum
- High aesthetic: Aluminum has nice silver whiter and a good appearance when processed.
- It is soft (second only to gold); light in weight (its density of aluminum is only about one-third of iron or copper).
- Malleable (places in the sixth)
- Good electrical and thermal conductivity.
- Good corrosion resistance and durability due to the Al2O3 protective oxide layer
- It is also non-magnetic and does not burn when exposed to air under normal conditions.
- Low melting point (660° C) that can facilitate the melting process
- It does not have high cast-ability due to large shrinkage (up to 6%).

General Mechanical Properties of Aluminum
Aluminum has low mechanical properties (σb = 6Kg / mm2, HB = 25, δ = 40%) so it is easy to deform, low cutting machinability.
The yield strength of pure aluminum is about 7–11 MPa, while aluminum alloy has a yield strength ranging from 200 MPa to 600 MPa.
Aluminum alloy
Aluminum alloy definition
When fabricating aluminum, it is common for manufacturers to add alloys (such as Cu, Mn, Mg, Si, Sn, Zn) to improve the properties of aluminum.
Depending on each purpose of the application, they are combined in different to produce an aluminum alloy with the desired properties.
To sum up, aluminum alloy is the combination of the aluminum element with alloying elements to increase workability, corrosion resistance, electrical conductivity, density, strength, etc. comparing with the pure aluminum element.
Characteristics of Alloying Elements
The basic alloying elements used to improve the properties of aluminum alloys include: Cu, Si, Mn, Mg and Zn.
Here are the properties of each alloy element when adding to the aluminum:
| Alloy element | Properties |
| Copper (Cu) | Improves durability and formability |
| Silicon (Si) | Reduces melting temperature, increases flow dilution, and improves casting properties |
| Manganese (Mn) | Increases strength and toughness |
| Magnesium (Mg) | Improved durability, and resistance to corrosion |
| Mg / Si | Increases strength, formability and tensile properties |
| Zinc (Zn) | When combined with Mg and Cu, it improves durability |
Designation System of Wrought aluminum alloy and Cast aluminum alloy
Separately, based on the method of fabrication and usage, we divide aluminum alloy into two main groups is Wrought aluminum alloy and cast aluminum alloy.
Wrought aluminum alloy
As mention above, the alloy elements are added to aluminum in a certain proportion. With wrought aluminum alloys, the amount of alloy elements is less than 4% of the total mass.
Wrought aluminum alloy is made by melting aluminum ingots with alloy elements, then cast into large plates before rolling, forging, or pulling into different object shapes. Based on the fabrication, wrought aluminum alloy comes in a variety of shapes, including sheet, plate, foil, wire or rod, etc.

Depending on the alloying element composition, wrought aluminum alloy can be grouped into 7 main groups and 1 special group (group 8) as shown below.
In which, groups 2, 6, 7, 8 are heat-treatable groups to improve some mechanical properties, while groups 1, 3, 4, 5 belong to non-heat-treatable group.
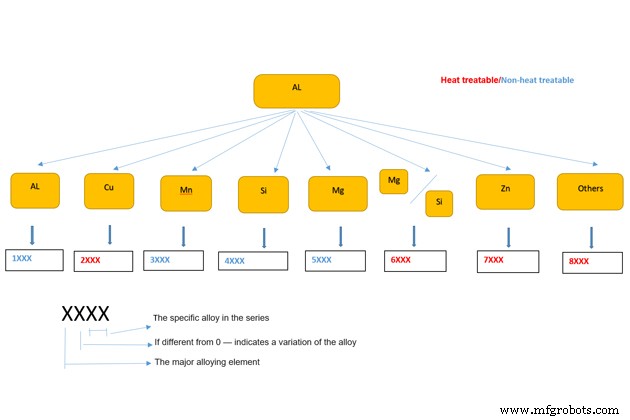
For Example:
Aluminum alloy 5083 means belonging to group 5, Aluminum-Magnesium (Al / Mg) alloy; the original version (0); and the 83 identifies it in the 5xxx series. Aluminum 5183 is the first variant version of aluminum 5083.
All wrought aluminum alloy series follow this explanation. There is only one exception is 1XXX series of aluminum alloys (pure aluminum). In which, the last third and fourth digits indicate the minimum aluminum percentage above 99%.
For example, aluminum alloy 1350 has 99.50% minimize pure aluminum.
Manufacturing process
- Tantalum: Key Properties and Diverse Industrial Applications
- Top 10 Properties of Aluminium and Their Industrial Applications
- Arduino Sensors: Types, Applications, and Real‑World Projects
- Refractory Metals: Key Properties, Types, and Industrial Applications
- Aluminum Bronze: Key Properties & Industrial Applications
- Understanding Molding Sand: Types, Properties, and Its Role in Sand Casting
- Discover 9 Essential Threading Tools and Their Key Applications
- Exploring Hot and Cold Chamber Die Casting: Types, Applications, and Advantages
- Types of Shaper Machines & Their Classifications – A Comprehensive Guide
- Exploring Metal Types: Key Properties and Applications



