Condoms: History, Production, and Health Impact
Background
Condoms—thin, flexible sheaths worn by men during intercourse—serve as a barrier method that prevents pregnancy and the transmission of sexually transmitted infections (STIs). A 1995 National Survey of Family Growth by the National Center for Health Statistics found that male condoms are the third most widely used birth‑control method in the United States, with a 17.7% prevalence, trailing only female sterilization (29.5%) and oral contraceptive pills (28.5%). When used correctly, condoms exhibit a failure rate of 2–3%, making them one of the most reliable contraceptive options available. While most condoms are manufactured from natural latex rubber, alternative materials such as lamb cecum and polyurethane are also employed.
In addition to contraception, condom use has been proven to significantly reduce the spread of STIs. The U.S. Surgeon General endorsed condoms in 1986 as the only effective barrier against AIDS transmission. They also effectively curb infections like chlamydia and gonorrhea. With widespread public‑health advocacy and visible retail placement, condoms that once resided behind prescription counters are now stocked on most store shelves, and approximately 450 million units are sold annually in the U.S.
Although manufacturers offer a range of styles—straight‑sided, contoured, ribbed, sensitive, or smooth—most latex condoms share similar performance characteristics. Variations such as lubricant or spermicide treatments, blunt ends, or reservoir tips are marketed primarily to build brand loyalty rather than to differentiate quality.
Lamb‑cecum condoms, derived from the blind pouch where the ileum meets the small intestine, are available but carry a higher price point and do not provide STI protection. In 1994 the FDA approved a polyurethane condom for sale in the U.S.; its effectiveness in preventing pregnancy and disease transmission has yet to be extensively validated.
History
The earliest recorded use of condoms dates back to ancient Egypt around 1350 BCE. In 1564, Italian anatomist Gabriele Falloppio described linen condoms designed to guard against venereal disease. The word “condom” itself stems from Dr. John Conton, an 18th‑century British physician who supplied condoms to King Charles II of France. The legendary lover Giovanina Casanova (1725–1798) is credited with using sheep‑intestine sheaths for protection. The first U.S. condom manufacturer was Schmid Laboratories, founded by Julius Schmid in 1883. Schmid, a former sausage‑skin maker, leveraged his expertise in animal membranes to produce the first lamb‑cecum condoms.
Concurrently, advances in rubber technology—such as vulcanization (developed in 1839 for automobile tires) and the discovery of latex in the 1930s—enabled the production of thinner, more pliable, and cost‑effective condoms. The mechanized dipping process further accelerated mass production and reduced manufacturing costs.
Raw Materials
Early condoms were crafted from lamb cecum. By 1990, lamb‑cecum condoms represented 5.5% of the market share but accounted for 20% of retail sales due to their premium pricing. The manufacturing process remains largely unchanged: cecums are washed, defatted, salted, and shipped to finishing plants. New Zealand, a major sheep‑raising nation, supplies most of the raw skins.
Latex condoms dominate today’s market. Because natural latex varies in strength and elasticity, manufacturers stabilize the material with chemical additives. Many brands also incorporate talc, lubricants, or spermicides during packaging.
The Manufacturing Process
Collecting the Raw Materials
- 1. Rubber latex is harvested from tropical plants. It is an emulsion of tiny rubber particles suspended in water; additives must bind to these particles during compounding.
Compounding
- 2. Chemical additives are blended into a paste and then mixed with the liquid latex to form a uniform compound.
Storage
- 3. The latex‑compound mixture is stored in drums for about seven days. During this period, vulcanization strengthens rubber bonds and allows trapped air to escape.
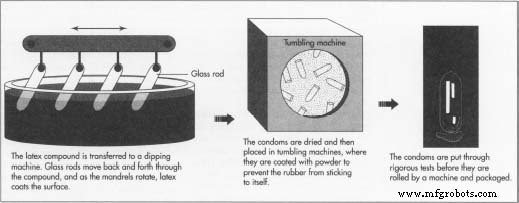
Dipping
- 4. The compound is fed into a dipping machine—a 30.5‑meter long apparatus featuring glass mandrels that rotate and dip into the latex. Multiple coats are applied, with hot‑air drying between dips to build the desired thickness.
- 5. Once fully dried, condoms detach automatically and a secondary machine trims the latex ring at the base.
Tumbling
- 6. Condoms enter a tumbling chamber where they are lightly coated with talc or a similar powder to prevent sticking.
Testing
- 7. After curing, each batch undergoes leak and strength testing. The inflation test requires the condom to stretch beyond 1.5 cubic feet before bursting, ensuring sufficient elasticity.
- 8. The water‑leakage test fills the condom with 300 ml of water and checks for pin‑hole leaks by rolling it over blotter paper.
- 9. An electronic test mounts the condom on a charged stainless‑steel mandrel and passes a conductive brush over it; any pin holes complete a circuit, causing automatic rejection.
Packaging
- 10. Condoms that pass all tests are rolled, lubricated, and optionally spermicide‑coated before being sealed in foil wrappers. The final roll facilitates easy use and storage.
Quality Control
Condoms are classified as Class II medical devices. Under the 1976 FDA Medical Device Amendments, each manufacturing plant undergoes inspection at least biennially. Equipment—especially continuous‑operation dipping machines—must be meticulously maintained to prevent clogs, rust, or contamination during downtime.
All U.S. condoms must meet specifications voluntarily developed by manufacturers and adopted by the FDA. Typical dimensions range from 150–200 mm in length, 47–54 mm in width, 0.03–0.09 mm in thickness (most between 0.05–0.06 mm), and a weight not exceeding 2 grams. Physical requirements include a minimum tensile strength of 15,000 psi and an elongation before breakage of 625 %.
The FDA conducts spot checks on every lot for defects such as cracking, improper drying, or latex sticking. Import batches are also tested. A lot fails if it shows more than 4 % dimensional deviation, 2.5 % tensile/elongation failure, or 0.4 % leakage.
The Future
Female Health Co., based in Chicago, introduced the Reality condom for women in 1994. Available through family‑planning clinics and in 12 European countries, this polyurethane sheath features an open ring and a closed ring, anchoring between the cervix and vagina. The company claims it is 40 times stronger than latex and costs roughly $3 per unit versus about $0.64 for male latex condoms.
In 1994, a new polyurethane male condom entered the market. Developed from research initiated in 1988, it offers comparable strength to latex but is one‑tenth as thick, making it an excellent alternative for latex‑allergic users.
Manufacturing process
- Light‑Emitting Diodes: From Inception to 100‑Year Lifespans
- Liquid Crystal Display (LCD): Technology, Manufacturing, and Future Outlook
- What Makes a Reliability Professional? A Practical Guide to Hiring & Leadership
- Micro Med Machining: Precision Beyond Swiss Lathe Standards
- Millwrights: Key Roles, Responsibilities, and Their Value in Canada
- The Role of a Rigger: Key Responsibilities & Skills
- Mastering Maintenance Scheduling: Boost Efficiency & Cut Costs
- Top 10 Manufacturing Failures: What You Need to Know
- Eco‑Friendly Robots: Revolutionizing Sustainability in Industry
- Unlocking the Power of Robotics: Key Benefits for Modern Industries



