Decoding SARS‑CoV‑2 Genomes: DNA & Antibody Tests for COVID‑19
“Testing, testing, testing!” has become a rallying cry for COVID‑19 containment. Global health authorities—including the World Health Organization and national agencies—continue to emphasize that rapid, widespread testing is the linchpin of any effective pandemic response.
Extensive diagnostic coverage informs targeted public‑health measures—such as social distancing, stay‑at‑home orders, and, when necessary, curfews—by revealing who is infected and where the virus is spreading. These insights relieve pressure on health‑care systems and, ultimately, save lives.
Genome‑based analysis is the cornerstone of any SARS‑CoV‑2 test, whether it is a DNA‑based assay (RT‑PCR, isothermal amplification, or CRISPR) or a serologic test that uses viral antigens. A global, non‑profit consortium has catalogued every commercially available test; here we focus on the two primary modalities in use today: nucleic‑acid detection and antibody screening.
DNA‑Based Testing
Current clinical protocols rely on extracting viral RNA from nasopharyngeal or oropharyngeal swabs and amplifying a unique segment of the SARS‑CoV‑2 genome. The gold‑standard method is Reverse Transcription‑Polymerase Chain Reaction (RT‑PCR), while Abbott Laboratories’ isothermal platform offers a rapid alternative.
RT‑PCR proceeds in three stages: (1) RNA extraction; (2) reverse transcription to cDNA; and (3) temperature‑cycling PCR that denatures, anneals primers, and extends the product, yielding exponential amplification. Results are typically available within a few hours. The World Health Organization and the Centers for Disease Control and Prevention provide detailed protocols.
In contrast, Abbott’s isothermal assay eliminates the need for thermal cycling. It delivers a positive result in 5 minutes and a negative in 13 minutes1.
Target selection hinges on genome‑wide comparisons that pinpoint regions unique to SARS‑CoV‑2. The virus’s ~30,000‑base single‑stranded RNA encodes four structural proteins—envelope (E), membrane (M), nucleocapsid (N), and spike (S). Comparative analyses identify unique sequences within ORF1ab, N, RdRp, and S that serve as primer and probe templates. Current RT‑PCR kits target these regions2, and Abbott’s ID NOW™ COVID‑19 assay focuses on RdRp.
Emerging Technologies
CRISPR‑based diagnostics—leveraging the enzyme’s sequence‑specific cleavage—offer a scalable testing alternative. The SHERLOCK platform (Specific High‑Sensitivity Enzymatic Reporter UnLOCKing) combines CRISPR‑Cas13 with a lateral‑flow readout, producing results in ~1 hour. DETECTR, a CRISPR‑Cas12 assay, delivers outcomes in 30 minutes34.
Sequence‑directed testing provides definitive evidence of viral presence, a prerequisite for informed public‑health decisions. In the United States, testing has traditionally focused on symptomatic individuals, but asymptomatic transmission remains a significant risk, underscoring the need for broad diagnostic coverage.
Antibody‑Based Testing
Serologic assays detect antibodies generated in response to SARS‑CoV‑2, enabling identification of individuals who have been previously infected and may possess protective immunity. These tests also facilitate the selection of convalescent plasma donors for therapeutic use—a strategy currently under investigation in the U.S.
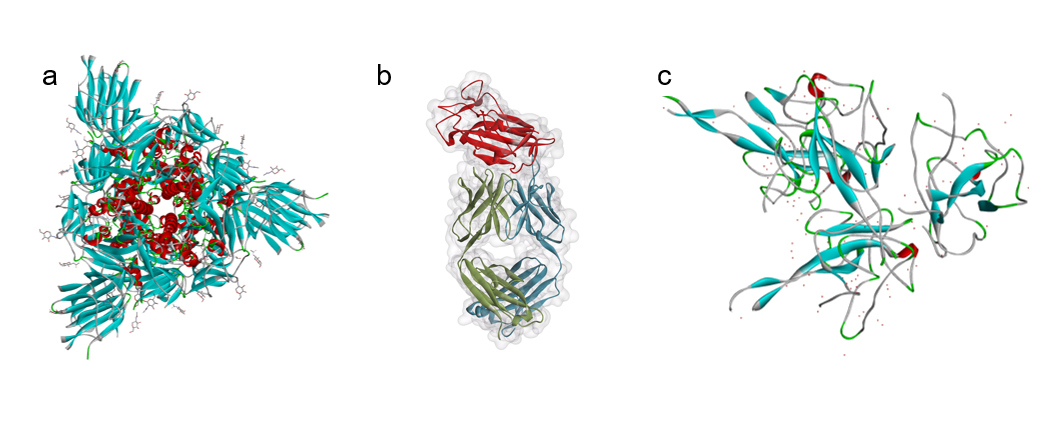
Successful antibody diagnostics hinge on selecting viral antigens that elicit a robust immune response. The spike (S) protein, particularly its receptor‑binding domain (RBD), is a primary target because it is highly immunogenic. Other candidates include the nucleocapsid (N) protein and full‑length spike constructs. Antigens are produced in recombinant systems, then incorporated into enzyme‑linked immunosorbent assays (ELISAs) or rapid lateral‑flow formats.
Developing reliable home‑testing kits remains a challenge, largely due to the need for proper protein folding and post‑translational modifications. The U.S. Food & Drug Administration has recently approved the first antibody test, providing a framework for estimating population immunity5.
In the current pandemic landscape, DNA‑based and antibody‑based diagnostics are pivotal for shaping public‑health interventions aimed at curbing SARS‑CoV‑2 transmission. Rapid, accurate testing remains essential as we continue to “flatten the curve.”
For further information, please contact us: https://www.3dsbiovia.com/about/contact/.
Biologics
- Unraveling the Origins of SARS‑CoV‑2: A Genomic Perspective
- COVID‑19’s Impact on Manufacturing: Insights from 10,000+ Global 3D Printers
- Software Testing for IoT Devices: Key Challenges & Solutions
- Navigating the COVID-19 Recession: Automation’s Role in Manufacturing Resilience
- How COVID-19 Reshaped Global Trade: Long-Term Effects and Lessons
- COVID-19 Detector: Responding to the Crisis with Innovative Technology
- Why Motor Insulation Testing Is Critical for Reliability and Cost Savings
- Unified COVID-19 Test: Detect, Track Variants & Co-Infections in One Procedure
- Manufacturing Resilience in the COVID-19 Era: Adapting to Pandemic Challenges
- How Automation Accelerated During COVID-19: Trends, Flexibility, and the Rise of Cobots



