Transition‑Metal Doping of Kaolinite Nanoclay: Structural and Electronic Insights from DFT
Abstract
Using dispersion‑corrected density functional theory (DFT‑D2), we investigated kaolinite nanoclays doped with transition metals Cr, Mn, Fe, and Co. The study examined how these dopants modify the crystal geometry and electronic structure, evaluating ferromagnetic (FM), antiferromagnetic (AFM), and nonmagnetic (NM) configurations. Calculations of crystal volume, lattice constants, bond lengths, charge distribution, and spin moments revealed that Cr³⁺ and Fe³⁺ favor the AFM state, Mn³⁺ stabilizes both AFM and FM states, while Co³⁺ prefers the NM state. Metal substitution also expands the lattice and introduces localized states within the band gap, offering new avenues for functionalizing kaolinite in catalysis and energy storage.
Background
Kaolinite, a widely distributed nanoclay mineral, possesses a layered structure and abundant surface hydroxyl groups that confer unique physical properties. These characteristics have attracted researchers in materials chemistry, environmental science, and mineral physics [1–11]. Its versatility has led to applications in plastics, catalysis, and cement production, and recent studies have explored functionalizing kaolinite as a support for nanoparticles in solar energy phase‑change materials [4,5] or conductive oxide coatings [9,12]. Hybridization with functional nanoparticles has been shown to enhance photocatalytic activity of Pd–ZnO and luminescence of CdS via synergistic effects [6,7]. Surface modification through anchoring functional groups [13,14] or acid activation pretreatment [2] further improves kaolinite’s performance.
Experimental and theoretical investigations have elucidated the structure and energetics of kaolin‑group minerals [15–22]. Heavy‑metal adsorption studies on kaolinite surfaces, including Cd, Cu, Hg, and Ni(II) [23], as well as Pb(II) and uranyl species [24–28], have mapped adsorption hierarchies and energetic profiles. Doping with Mg, Ca, and Fe has been shown to influence H₂O penetration and interlayer interactions [29]. Electronic structure analyses of pristine and defected kaolinite using standard DFT and hybrid functionals [30] revealed the impact of intrinsic defects. Recent DFT models have also captured the evolution of kaolinite during dehydroxylation, dealumination, and silica condensation, highlighting how Al removal alters geometry and electronic properties [1,31,32].
Transition‑metal doping is a well‑established strategy for tuning the properties of oxides such as Al₂O₃ [33], TiO₂ [34], MOFs [35], and other solids [36]. Extending this approach to kaolinite offers the potential to tailor its structural and electronic characteristics for advanced applications. In this work, we systematically explored Cr, Mn, Fe, and Co substitution at the Al³⁺ sites of kaolinite, assessing geometric, electronic, and magnetic outcomes using DFT‑D2 calculations.
Methods
All calculations were performed with the CASTEP code [37] employing the Perdew–Burke–Ernzerhof (PBE) GGA functional [38] augmented by Grimme’s DFT‑D2 dispersion corrections [39]. A plane‑wave cutoff of 500 eV and a 2 × 2 × 3 Monkhorst–Pack k‑point mesh were used for geometry optimizations and electronic structure evaluations. Convergence criteria were set to 1.0 × 10⁻⁶ eV/atom for SCF tolerance, 0.03 eV/Å for atomic forces, 0.05 GPa for stress, and 0.001 Å for ionic displacement. Valence states included O(2s²2p⁴), Al(3s²3p¹), Cr(3s²3p⁶3d⁵4s¹), Mn(3d⁵4s²), Fe(3d⁶4s²), and Co(3d⁷4s²). Ultrasoft pseudopotentials (USPP) were employed for Mn, Fe, and Co, while norm‑conserving pseudopotentials were used for the remaining elements. Geometry optimizations were carried out using the Broyden–Fletcher–Goldfarb–Shanno (BFGS) algorithm, and initial magnetic moments were varied to explore FM, AFM, and NM states.
Results and Discussion
The starting kaolinite structure corresponds to our previous work [1]. Figure 1 displays the relaxed 2 × 2 × 1 supercell (four kaolinite units). Kaolinite’s framework, Al₂Si₂O₅(OH)₄, consists of an octahedral Al–O sheet and a tetrahedral Si–O sheet linked via apical oxygen (Oₐ). The Si–O tetrahedron features one Oₐ and three basal O_b atoms, while the Al–O octahedron incorporates two Oₐ atoms and four hydroxyl O atoms shared with neighboring octahedra. Hydroxyl groups divide into inter‑layer OH_inter (surface) and inner OH_inner (internal) species. Consequently, the crystal contains distinct Si–Oₐ, Si–O_b, Al–O_inter, Al–O_inner, and Al–O bonds, as illustrated in the figure.
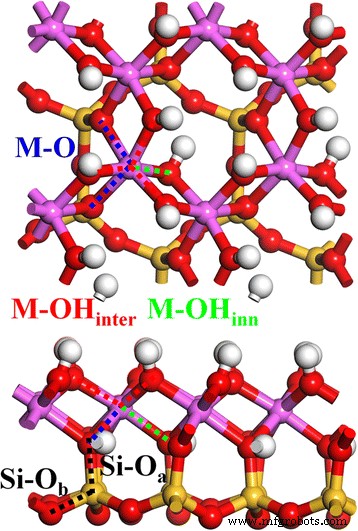
Top (up) and side (down) views of kaolinite. The Si–Oₐ (black), Si–O_b (black), M–OH_inter (red), M–OH_inner (green), and M–O (blue) bonds are indicated by dotted lines.
Dispersion forces dominate interlayer stabilization in clays. Among hybrid functionals, PBE‑D2 accurately reproduces experimental lattice parameters while remaining computationally efficient [21,44,45]. Comparison with experimental data (Table 1) shows PBE‑D2 reduces the relative error in unit‑cell volume to ~0.4 % (vs. ~3.4 % for plain PBE). Lattice vectors a and b exhibit ~0.4 % error, and the interlayer distance c contracts by 0.17 Å (~2 %) under dispersion corrections. Bond lengths of Si–Oₐ, Al–OH_inner, and Al–O are largely unchanged, whereas Al–OH_inter (critical for surface chemistry) shows substantial improvement. The two distinct Al–O bonds (~1.95 and 2.00 Å) arise from lattice mismatch between Si–O and Al–O sheets; both PBE and PBE‑D2 slightly overestimate these distances (average ~1.96 Å).
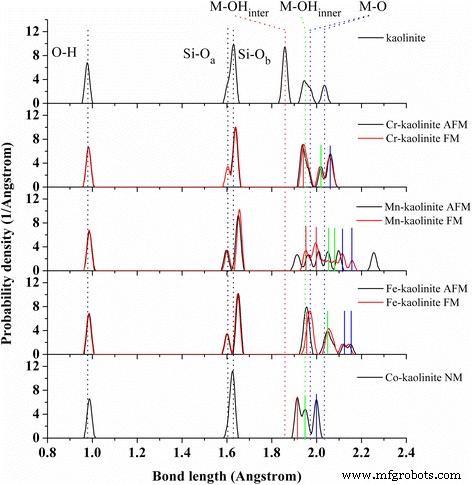
Bond distributions in Cr‑, Mn‑, Fe‑, and Co‑doped kaolinite. Different magnetic states are shown for each TM‑kaolinite. The averaged Si–O and M–O bond lengths are indicated by dotted lines; solid lines highlight M–OH_inter (red), M–OH_inner (green), and M–O (blue) bonds for the most stable configurations.
Transition‑metal substitution involved replacing Al³⁺ with TM³⁺ (Cr³⁺, Mn³⁺, Fe³⁺, Co³⁺). Structural responses are similar across PBE and PBE‑D2, but we focus on PBE‑D2 results. Table 1 summarizes lattice parameters, bond lengths, charges, spins, and magnetic states. Energy differences per TM atom between AFM and FM states are 0.022 eV (Cr), –0.006 eV (Mn), and 0.094 eV (Fe). Co‑doped kaolinite stabilizes only in the NM state.
Unit‑cell volumes expand in the order Mn‑ > Fe‑ > Cr‑ > pristine > Co‑, primarily due to longer M–O bonds relative to Al–O, enlarging a and b vectors. Si–O_b bonds also elongate, subtly distorting α and β angles. Mn‑kaolinite shows a 1.4 % volume increase in FM versus AFM, whereas Cr‑ and Fe‑kaolinite volumes are largely unaffected by magnetic ordering. Magnetic moments of the dopants mirror those in TM‑doped Al₂O₃ [33], while Mulliken charges are slightly higher, suggesting enhanced reactivity.
Figure 2 highlights the evolution of bond lengths upon doping. M–O and Si–O_b distances increase, and the split Al–O bonds vanish after Cr and Co substitution. Magnetic ordering significantly affects Mn‑bond distributions, but Cr and Fe bonds are relatively insensitive to spin state.
Projected density of states (PDOS) for Cr³⁺(d³), Mn³⁺(d⁴), Fe³⁺(d⁵), and Co³⁺(d⁶) (Fig. 3) and corresponding charge densities (Fig. 4) illustrate d‑state splitting into t₂g and e_g levels under octahedral symmetry. Jahn–Teller distortions are evident for Mn³⁺, where occupation of the d_z² orbital repels ligands along the z‑axis, elongating M–O bonds. Cr³⁺, Fe³⁺, and Co³⁺ display balanced t₂g/e_g occupations, yielding minimal Jahn–Teller effects. These electronic modifications introduce localized 3d states within the band gap, potentially enhancing kaolinite’s catalytic, CO‑capture, drug‑loading, and energy‑storage capabilities [50–57]. Similar strategies could extend to montmorillonite, perlite, and talc.
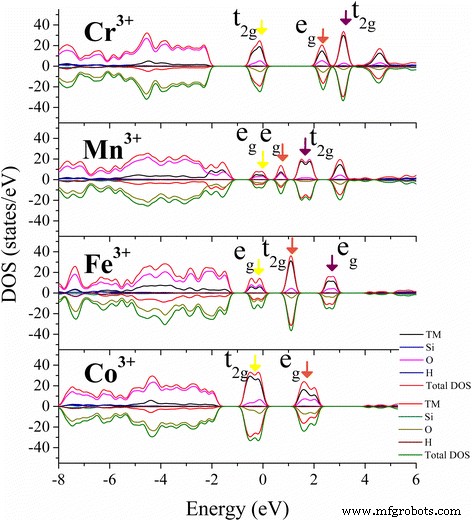
PDOS of the most stable TM‑doped kaolinite states. Yellow highlights the highest occupied 3d orbitals; brown and purple arrows point to the first and second lowest unoccupied 3d orbitals near the Fermi level.
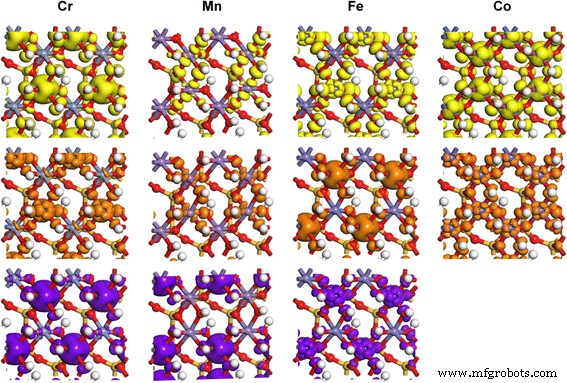
Partial charge density of TM 3d orbitals in TM‑doped kaolinite, corresponding to the PDOS states. Isosurface at 0.02 e/ų.
Conclusions
DFT‑D2 calculations reveal that Cr³⁺ and Fe³⁺ dopants preferentially stabilize the AFM state, Mn³⁺ favors the FM state, and Co³⁺ remains nonmagnetic. Transition‑metal substitution expands the lattice and reorganizes M–O bond distributions, while introducing 3d states that split more strongly within the kaolinite band gap. These insights provide a solid foundation for tailoring kaolinite’s structure and electronic properties for catalytic, adsorption, and energy‑storage applications.
Nanomaterials
- Comprehensive Atomic & Electronic Analysis of Stacked-Cup Carbon Nanofibers
- Tuning Electronic and Optical Anisotropy in Monolayer GaS via Vertical Electric Fields
- How Water Alters the Structure and Dielectric Behavior of Microcrystalline Cellulose
- Structural, Electronic, and Magnetic Characterization of Ag<sub>n</sub>V (n = 1–12) Clusters via DFT and CALYPSO Search
- Electronic Structure and Transport in InSe Nanoribbons: Edge Effects and Hydrogen Passivation
- Engineering Spin & Valley Physics in Silicene: Periodic Potentials Unlock Tunable Bandgaps and Dirac Points
- V, Cr, Mn Edge‑Functionalized Armchair Phosphorene Nanoribbons: Half‑Semiconductors and Spin‑p‑n Diodes
- Band‑gap Engineering in InGaNBi Quaternary Alloys: Composition‑Dependent Structural, Electronic, and Strain Properties
- Tailoring Electronic and Optical Properties of WSSe Bilayer via Strain Engineering
- Chromium: Essential Trace Element, Properties, and Practical Applications



