High‑Performance Binder‑Free NiCo₂S₄@NiMoO₄ Core‑Shell Nanotube Array on Ni Foam for Supercapacitor Applications
Abstract
Core‑shell architectures combine the merits of both core and shell materials, delivering enhanced conductivity and enlarged surface area—key attributes for next‑generation energy storage. Here, we report a hierarchical NiCo₂S₄@NiMoO₄ core‑shell heterostructure grown as a nanotube array directly on Ni foam (NF). The novel architecture yields a specific capacitance of 2006 F g⁻¹ at 5 mA cm⁻², surpassing pristine NiCo₂S₄ nanotubes (≈1264 F g⁻¹). In an asymmetric supercapacitor (ASC) pairing NiCo₂S₄@NiMoO₄/NF with activated carbon (AC), we achieve 78 % capacitance retention after 2000 cycles, an energy density of 21.4 Wh kg⁻¹ and a power density of 58 W kg⁻¹ at 2 mA cm⁻². These results highlight the promise of NiCo₂S₄@NiMoO₄/NF as a binder‑free electrode for high‑performance supercapacitors.
Background
The rapid rise in global energy demand has spurred extensive research into clean, renewable storage technologies. Supercapacitors stand out for their high power density, fast charge–discharge capability, long cycle life, and environmental friendliness, making them attractive for grid stabilization and electric vehicles [7–9]. Traditional electrostatic supercapacitors rely on high‑surface‑area carbon materials (CNTs, graphene, activated carbon) that store charge via electric double‑layer formation. In contrast, pseudocapacitive materials—transition‑metal oxides, sulfides, and conducting polymers—provide higher specific capacitances through Faradaic redox reactions [10–12].
Among oxides, ternary compositions such as NiCo₂O₄, CuCo₂O₄, NiMoO₄, and CoMoO₄ offer improved conductivity and richer active sites compared with their binary counterparts, yet challenges remain: limited intrinsic conductivity, sluggish ion diffusion, and volume changes during cycling [16–18]. Transition‑metal sulfides, particularly NiCo₂S₄, have attracted attention for their superior electrical conductivity and abundant redox activity, yet many reports still report capacitances below 1500 F g⁻¹ [26].
Core‑shell heterostructures have emerged as a powerful strategy to address these limitations. By coating a conductive, porous core with a redox‑active shell, one can synergistically enhance surface area, conductivity, and electrochemical stability [27–33]. Direct growth on current collectors (e.g., Ni foam) further improves mechanical adhesion and electrical contact, boosting active‑material utilization [34].
Building on these principles, we synthesized a NiCo₂S₄@NiMoO₄ core‑shell nanotube array on Ni foam via a facile hydrothermal route followed by annealing. The resulting binder‑free electrode exhibits a specific capacitance of 2006 F g⁻¹ at 5 mA cm⁻² and retains 75 % of its initial capacitance after 2000 cycles at 50 mA cm⁻². When paired with AC in an ASC, the device delivers a 1.6 V operating window, 21.4 Wh kg⁻¹ energy density, and 78 % capacitance retention over 2000 cycles, underscoring its potential for practical energy‑storage applications.
Methods
Synthesis of NiCo₂S₄/NF
NiCo₂S₄ nanotube arrays were fabricated on cleaned Ni foam (1 × 4 cm) via a two‑step hydrothermal process. First, a Co(NO₃)₂·6H₂O/Ni(NO₃)₂·6H₂O/urea solution (2:1:5 molar ratio) was reacted at 120 °C for 12 h in a Teflon‑lined autoclave, yielding a Ni‑Co precursor. Subsequent ion‑exchange with 0.03 M Na₂S at 90 °C for 12 h transformed the precursor into NiCo₂S₄. The mass loading averaged 2 mg cm⁻².
Synthesis of NiCo₂S₄@NiMoO₄/NF
NiCo₂S₄/NF was immersed in a 70 mL solution containing 1 mmol Ni(NO₃)₂·6H₂O and 1 mmol Na₂MoO₄·2H₂O, followed by hydrothermal treatment at 100 °C for 4 h. The product was then annealed at 400 °C for 2 h under Ar to form the NiMoO₄ shell, yielding a core‑shell structure with a mass loading of ~3 mg cm⁻².
Material Characterization
X‑ray diffraction (XRD) confirmed the presence of NiCo₂S₄ and NiMoO₄ phases. Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) revealed a 3‑D grass‑like array of 70–100 nm‑diameter NiCo₂S₄ nanotubes, uniformly coated by 300 nm‑thick NiMoO₄ nanosheets. X‑ray photoelectron spectroscopy (XPS) verified the oxidation states of Ni²⁺/Ni³⁺, Co²⁺/Co³⁺, and Mo⁶⁺, confirming the intended core‑shell composition.
Electrochemical Measurements
Electrochemical performance was evaluated in a three‑electrode setup (working electrode: NiCo₂S₄ or NiCo₂S₄@NiMoO₄/NF; counter electrode: Pt; reference: SCE) using 2 M KOH. Cyclic voltammetry (CV), galvanostatic charge‑discharge (GCD), and electrochemical impedance spectroscopy (EIS) were performed. Specific capacitance was calculated from GCD curves via Cₛ = IΔt/(mΔV).
Fabrication of the Asymmetric Supercapacitor
The ASC was assembled in a two‑electrode configuration with NiCo₂S₄@NiMoO₄/NF as the positive electrode and AC as the negative electrode, separated by filter paper and immersed in 2 M KOH. The AC electrode was prepared by mixing AC (85 wt %), acetylene black (10 wt %) and PVDF (5 wt %) in NMP, coating onto Ni foam, and drying at 60 °C under vacuum. Mass balance (Q₊ = Q₋) ensured optimal loading, yielding ~24.84 mg cm⁻² for AC.
Results and Discussion
Figure 1 illustrates the fabrication sequence: (a) hydrothermal growth of NiCo₂S₄ nanotubes on Ni foam, (b) ion‑exchange conversion to sulfide, and (c) hydrothermal deposition followed by annealing to form the NiMoO₄ nanosheet shell.
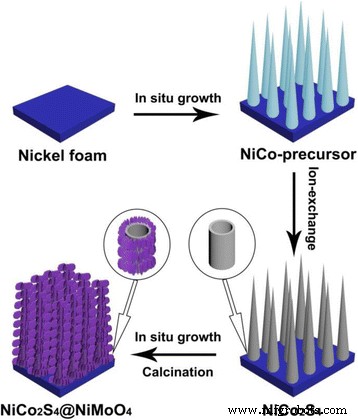
Schematic of the NiCo₂S₄@NiMoO₄/NF core‑shell synthesis
XRD patterns (Figure 2) confirm the coexistence of NiCo₂S₄ (PDF 43‑1477) and NiMoO₄ (PDF 86‑0362) phases. XPS spectra (Figure S2) show Ni²⁺/Ni³⁺, Co²⁺/Co³⁺, and Mo⁶⁺ states consistent with the intended heterostructure.
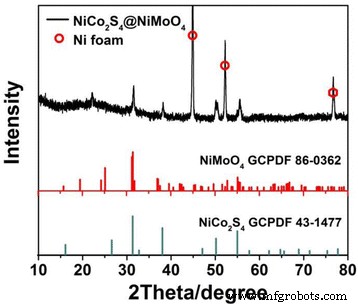
XRD pattern for NiCo₂S₄@NiMoO₄/NF
SEM and TEM images (Figure 3) reveal a uniform 3‑D array of NiCo₂S₄ nanotubes (70–100 nm diameter) coated by interconnected NiMoO₄ nanosheets (~300 nm thick), yielding a hierarchical core‑shell architecture with an overall diameter of ~700 nm. HRTEM confirms lattice spacings of 0.28 nm (NiCo₂S₄) and 0.243 nm (NiMoO₄), matching the (311) and (021) planes, respectively.

SEM and TEM images of NiCo₂S₄/NF and NiCo₂S₄@NiMoO₄/NF (a–f), plus HRTEM (g–i)
Electrochemical tests (Figure 4) show that the core‑shell electrode exhibits enlarged CV areas and extended GCD plateaus, reflecting enhanced redox activity from both NiCo₂S₄ core and NiMoO₄ shell. Specific capacitance values reach 2006 F g⁻¹ at 5 mA cm⁻², compared to 1264 F g⁻¹ for bare NiCo₂S₄. Even at 50 mA cm⁻², the core‑shell retains 65 % of its capacitance versus 45 % for the pristine material, demonstrating superior rate capability.
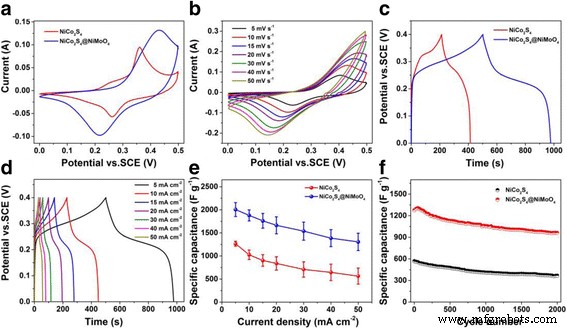
Electrochemical performance of NiCo₂S₄ and NiCo₂S₄@NiMoO₄ (a–f)
Cycling tests (Figure 4f) show 75 % capacitance retention after 2000 cycles at 50 mA cm⁻², outperforming the bare NiCo₂S₄ (65 %). EIS Nyquist plots (Figure S4) exhibit minimal change pre‑ and post‑cycling, confirming stable charge‑transfer pathways.
In the ASC configuration, the device achieves a 1.6 V operating window. CV curves remain rectangular up to 1.6 V (Figure 5a), and GCD curves show distinct plateaus (Figure 5c). Specific capacitance values of 60.05 F g⁻¹ (2 mA cm⁻²) and 35.45 F g⁻¹ (40 mA cm⁻²) are achieved. Cycling at 40 mA cm⁻² yields 78 % retention after 2000 cycles (Figure 5e). Energy and power densities reach 21.4 Wh kg⁻¹ and 58 W kg⁻¹, respectively, surpassing many reported Ni‑based ASC systems (Figure 5f).
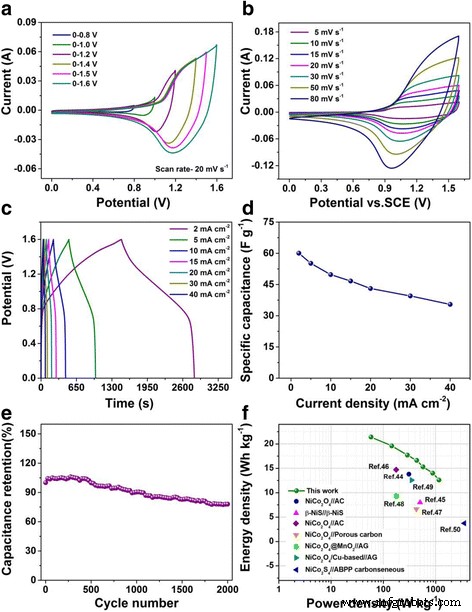
Performance of the NiCo₂S₄@NiMoO₄//AC asymmetric supercapacitor (a–f)
Conclusions
We have successfully fabricated a binder‑free NiCo₂S₄@NiMoO₄ core‑shell nanotube array on Ni foam. The electrode delivers an impressive 2006 F g⁻¹ at 5 mA cm⁻² and retains 75 % of its capacitance after 2000 cycles. When assembled into an asymmetric supercapacitor with AC, the device operates at 1.6 V, delivering 21.4 Wh kg⁻¹ energy density and 78 % capacitance retention over 2000 cycles. These findings demonstrate the potential of the core‑shell architecture for high‑performance, binder‑free supercapacitor electrodes.
Abbreviations
- ABPP:
Activated balsam pear pulp
- AC:
Active carbon
- AG:
Activated graphenes
- ASC:
Asymmetric supercapacitor
- CV:
Cyclic voltammetry
- DI:
Deionized
- EIS:
Electrochemical impedance spectroscopy
- GCD:
Galvanostatic charge‑discharge
- HRTEM:
High‑resolution transmission electron microscopy
- NF:
Ni foam
- PVDF:
Polyvinylidene fluoride
- SCE:
Standard calomel electrode
- SEM:
Scanning electron microscope
- TEM:
Transmission electron microscope
- XPS:
X‑ray photo‑electron spectroscopy
- XRD:
X‑ray diffraction
Nanomaterials
- High‑Performance Na4Mn9O18/Carbon Nanotube Cathodes for Aqueous Sodium‑Ion Batteries
- Highly Compressible Graphene/Polyaniline Aerogel: Superelasticity Meets 713 F g⁻¹ Capacitance for All‑Solid‑State Supercapacitors
- Electrochemical Energy Storage: Enhancing Capacitance with Reduced Graphene Oxide/Carbon Nanotube Composites
- Graphene‑Coated Porous Silicon Electrodes Deliver Exceptional Supercapacitor Performance
- Enhanced Organic–Nanostructured Silicon Hybrid Solar Cells via Surface Modification for Superior Efficiency
- Enhanced Deep‑UV Light Extraction in AlGaN Nanorod Arrays via Top‑Down Fabrication
- High‑Performance NiO Flakes@CoMoO₄ Nanosheets Core‑Shell Electrode on Ni Foam for Advanced Supercapacitors
- High‑Performance Flexible Photodetectors Based on Solution‑Processed CsPbI3‑xBrx Inorganic Perovskites
- Hierarchically Porous MoS₂/C Nanospheres from Nanosheets: Exceptional Electrochemical Energy Storage
- Ag-Doped Ga₂Te₃ Selector: Ultra-High Endurance Threshold Switching for 3D Cross-Point Memory



