Optimizing Morphology and Etching Dynamics of Porous Silicon Nanowires by Metal‑Assisted Chemical Etching
Abstract
Porous silicon nanowires (SiNWs) fabricated by metal‑assisted chemical etching (MACE) are increasingly valuable for optoelectronics, thermoelectrics, and biomedical sensing. Precise control of their morphology—length, diameter, porosity, and crystallinity—is essential for device optimization, yet the influence of key etching parameters remains incompletely understood. In this study, we systematically investigated the impact of silver (Ag) catalyst deposition time, the HF–oxidant molar ratio (χ = [HF]/([HF]+[H₂O₂])), and water concentration on the morphology and etching kinetics of degenerately doped p‑type Si wafers. Our results show that the surface evolves from a microporous, cratered texture to a uniform SiNW array when χ is sufficiently high. Etch rates at the nanowire base and tip are governed by primary Ag‑mediated dissolution and secondary reactions involving metal ions and diffused holes, respectively. By tuning χ and water concentration, we achieved independent control of porosity and length, demonstrating a practical route to tailor SiNW properties for high‑performance applications.
Background
Silicon remains a premier substrate for nanoscale fabrication because of its abundance and the maturity of its integration technologies. One‑dimensional Si structures—nanowires, nanopillars—have attracted intense interest for photovoltaics [1, 2], thermoelectrics [3, 4], energy storage [5, 6, 7], flexible electronics [8], biochemical sensing [9], and biological interfacing [10]. The sustained appeal of these nanostructures is driven not only by their unique electrical, optical, thermal, and mechanical properties but also by the availability of simple, low‑cost, and highly controllable fabrication routes such as MACE [11, 12]. MACE enables wafer‑scale, defect‑free SiNWs with defined length, porosity, conductivity, doping, and crystal orientation simply by selecting the Si wafer, etchant composition, temperature, and time [13–23]. Additional control of diameter and array pitch is achievable via catalyst‑patterning techniques (nanosphere lithography [24], interference lithography [25], block copolymer lithography [26]), which contrasts with reactive ion etching or vapor‑liquid‑solid growth that require expensive equipment and often produce surface defects or uncontrolled crystallographic orientations [11].
Highly doped Si wafers are especially attractive for MACE because they yield porous SiNWs with high crystalline quality [14, 17, 19]. The resulting porous shell confers tunable photoluminescence [15], reduced thermal conductivity [27], and a high specific surface area [28], making them promising for optoelectronics [14, 16], thermoelectrics [3, 27], photocatalysis [28, 29], and energy storage [5]. Their biocompatibility and biodegradability further enable biolabeling [15] and drug‑delivery applications [30]. While porous SiNWs can be produced on low‑doped wafers with high oxidant concentrations [15], using highly doped substrates removes the need for post‑etch doping and preserves electrical conductivity, a critical advantage for thermoelectric devices where a high figure of merit is achieved by reducing thermal conductivity without compromising electrical conductivity [27]. However, the resistance of porous SiNWs can be significantly higher than that of solid SiNWs [14], highlighting the trade‑off between porosity and electrical conductivity.
Comprehensive understanding of how etching parameters influence the morphology of highly doped SiNWs is essential to unlock their full potential. Prior studies [13–23] have examined the effects of H₂O₂ concentration [14–17, 20, 21, 23], HF concentration [21], HF/H₂O₂ volume ratio [19], etch duration [14, 16, 17, 19–21, 23], and temperature [19–21, 23] on porosity [14, 16–19, 20], length [16, 17, 19–21, 23], etch rate [15], and overall morphology [15, 16, 21]. Yet, systematic studies of the HF–H₂O₂ molar ratio (χ) and water concentration—both pivotal to morphology and etch kinetics—remain sparse. Only a few works have explored wide χ ranges (0.4–0.98 [15], 0.7–0.99 [19]), and none have fully examined the role of water concentration. Furthermore, while the mechanisms of morphological evolution, porosification, and tapering in lightly and highly doped SiNWs have been addressed [15, 17, 32], the interplay of deposited Ag‑mediated etching, re‑nucleated Ag etching, hole diffusion, and reactant diffusion on SiNW length evolution has not been fully elucidated. This study fills that gap by employing two‑step MACE on degenerately doped p‑type Si wafers with electrolessly deposited Ag catalysts and a broad range of χ and water concentrations.
Electroless Ag deposition yields randomly distributed SiNWs with varied diameters [13] and offers the simplest, most cost‑effective route to Si nanostructures, albeit with a lower achievable aspect ratio compared to Au catalysts [11]. By varying χ and water concentration across a wide range, we characterized how etchant composition controls nanostructure morphology and etch rates, and we explored the mechanisms of hole injection, Si dissolution, metal‑ion re‑nucleation, and reactant diffusion that shape the final SiNW arrays. Additionally, we demonstrate that porosity can be tuned independently of length by selecting appropriate χ and water concentrations, providing a powerful tool for device engineers.
Methods
We used single‑side polished, boron‑doped p‑type Si (100) wafers (resistivity 0.001–0.005 Ω·cm) with a 50‑nm thermal oxide layer on the polished side. The wafers were cleaned with deionized (DI) water, acetone, and ethanol, followed by dilute HF to strip the oxide and produce H‑terminated surfaces. The backside was coated with AZ5214 photoresist to confine etching to the polished surface. Si samples were cleaved into 1 × 1 cm² pieces from the same wafer for each experiment set.
Two‑step MACE was employed: (1) electroless Ag deposition in 4.8 M HF/0.005 M AgNO₃ for varying durations (Fig. 1a), followed by (2) etching in HF–H₂O₂–H₂O solutions with different χ and water concentrations (Table S1) for set times (Fig. 1b). After etching, samples were rinsed with DI water, then immersed in 1:1 HNO₃ for 10 min to dissolve residual AgNPs, rinsed again, and finally treated with 5 % HF for 3 min to remove any re‑oxidized layer before thorough DI water rinses. All processing was conducted at room temperature (22–23 °C) in separate plastic beakers containing 15 mL of the required solution, and performed in the dark to prevent photo‑induced reactions.
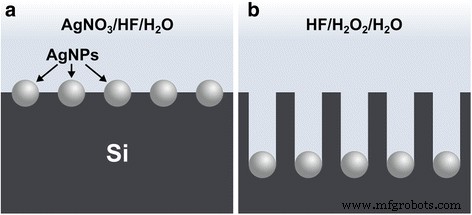
Schematic cross‑sectional view of the two‑step MACE process. a Electroless deposition of AgNPs on the Si surface in an AgNO₃/HF solution. b AgNP‑catalyzed etching of Si in HF–H₂O₂–H₂O to form Si nanostructures.
Etched surfaces were characterized by scanning electron microscopy (SEM, Phenom Pro or FEI/Philips XL‑30) and transmission electron microscopy (TEM, Philips CM20). For SEM, the backside photoresist was removed with acetone, samples were rinsed with ethanol, and dried on a hotplate to minimize SiNW agglomeration from water evaporation [19].
Results and Discussion
Silver Catalyst Deposition and Formation of Porous SiNWs
We first examined the effect of Ag deposition time (10 s–15 min) in 0.005 M AgNO₃/4.8 M HF on SiNW morphology. After etching in 4.8 M H₂O with χ = 0.95 for 30 min, short deposition times (≤2 min) produced a dense array of vertical pores with occasional lateral pits (Fig. 2a). At 1 min, pores were too sparse to form distinct nanowires. A 4‑min deposition yielded defect‑free SiNWs (Fig. 2b), while longer times eliminated pits. Interestingly, the SiNW etch rate varied non‑monotonically with deposition time (Fig. 2c), rising from 10 s to 4 min, dipping between 4–6 min, and then rising again at 15 min with lower overall rates.
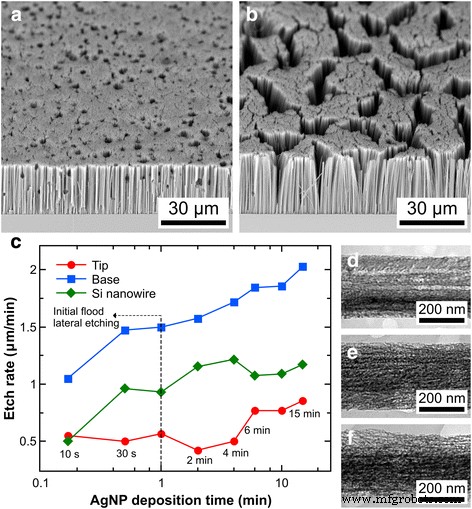
SEM images of SiNWs for Ag deposition times of a 30 s and b 4 min. c Etch rate versus deposition time. TEM images of the middle section for d 4, e 10, and f 15 min show increasing porosity. Etch duration: 30 min.
To rationalize the etch‑rate trend, we measured tip and base etch rates from cross‑sectional SEM images (Fig. 2c). Base rates increased monotonically with AgNP coverage, while tip rates displayed three regimes: high (≤1 min), low (1–6 min), and high (≥6 min). This behavior reflects the balance between primary Ag‑mediated etching at the base and secondary etching (metal‑ion re‑nucleation, hole diffusion) at the tip. Sparse Ag networks (≤1 min) cause lateral flooding and higher tip rates; intermediate coverage (1–6 min) suppresses tip etching; prolonged deposition (≥6 min) enhances tip rates again due to increased re‑nucleation.
Porosity increased with Ag deposition time (Fig. 2d–f). Higher Ag⁺ concentrations promote re‑nucleated metal and ion‑induced etching, while increased hole injection yields more diffused holes that drive pore formation. Porous shells formed around most nanowires, similar to prior reports [14, 17]. Longer deposition (15 min) produced rougher sidewalls and larger dark spots (~20 nm), likely from larger re‑nucleated Ag particles. Porosity varied along the nanowire axis (base to tip), consistent with prolonged exposure of upper sections to the etchant [16, 19]. Some nanowires exhibited biconic or hourglass profiles, indicating Ag⁺ redeposition on both the base and neighboring AgNPs.
Etchant Composition and Morphological Evolution of Si Nanostructures
Using a fixed Ag deposition time (4 min), we varied χ (0.7–0.99) and water concentration (46, 48, 50 M) while etching for 30 min. For 46 and 48 M, the surface evolved from microporous with craters (χ = 0.7, 0.75) to macroporous with deep pores (χ = 0.75, 0.80), and finally to SiNWs (χ ≥ 0.85). At 50 M, the progression differed: polished Si (χ = 0.7) → macroporous (χ = 0.75) → microporous with craters (χ = 0.8–0.85) → SiNWs (χ ≥ 0.9). Notably, SiNWs appeared over a broader χ range (≥0.85) than in prior work (χ > 0.95) [15], likely due to differences in AgNP loading, etch time, and post‑etch treatments.

SEM images of Si surfaces etched in solutions with water concentrations of a 46 M, b 48 M, and c 50 M and HF–H₂O₂ molar ratios (i) 0.7, (ii) 0.75, (iii) 0.8, (iv) 0.85, (v) 0.9, (vi) 0.95, and (vii) 0.99. d–g High‑magnification images of samples in (i), (ii), (iii), and (vii). Samples etched for 30 min.
Morphological trends can be rationalized by current density at the Ag–Si interface and Ag⁺‑induced etching [15, 17, 31, 38]. High χ (≥0.85–0.9) promotes microporous Si at low current density, followed by dissolution to yield SiNWs. Lower χ values favor macroporous Si with deep pores due to higher current densities that generate oxide and drive hole diffusion [31]. The emergence of microporous Si with craters at χ = 0.7–0.85 reflects low current density and concomitant Ag⁺‑induced porosification [31, 38]. For the most dilute etchant (50 M), higher χ is required because slow reactant diffusion limits etching at the nanowire base.
Non‑uniform etching (χ ≤ 0.8) arises from spatial variations in dissolved catalyst ion concentration and bubble‑mediated diffusion, leading to localized morphologies that cluster near the sample center. Uniform SiNW arrays form only within the optimal χ window. At χ = 0.99, a dense array of fine SiNWs appears, but many exhibit slanted sidewalls and lateral pits—hallmarks of Ag network disintegration and horizontal etching as H₂O₂ depletes.
Etching Kinetics of Si Nanostructures During MACE
Figure 4 shows the apparent etch rate versus χ for various water concentrations. Etch rates increase with χ, peak at χ = 0.95, and then drop sharply at χ = 0.99, regardless of water concentration. Higher water concentrations reduce etch rates by diluting reactants and slowing diffusion. Tip etch rates decline beyond χ = 0.85 (46/48 M) or χ = 0.9 (50 M), while base rates rise at χ = 0.85–0.9 (46 M) but only modestly for higher water concentrations. Base rates remain high at χ = 0.95 and fall at χ = 0.99.
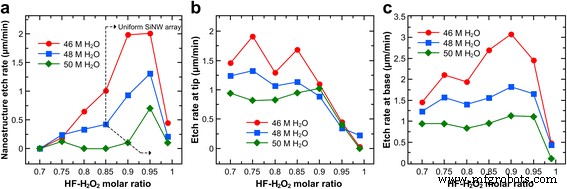
Apparent etch rates after 30 min of etching in solutions with different HF–H₂O₂ molar ratios and water concentrations. a Overall etch rate based on nanostructure length. b, c Etch rate at the tip and base of the Si nanostructures, respectively.
The evolution of SiNW length with time is governed by competing tip and base etching. At χ = 0.95, the longest SiNWs result from maximal base etching (optimal hole injection and HF dissolution) while tip etching remains low. At lower χ, increased Ag⁺ re‑nucleation and hole diffusion elevate tip rates, limiting length growth. Higher water concentrations dampen both tip and base rates due to reduced reactant concentrations and diffusion, whereas lower concentrations accelerate etching but can induce lateral pitting (Fig. 6).
Evolution of SiNW Length with Time for Different Etchant Compositions
We monitored SiNW length versus time for χ = 0.9, 0.92, 0.95, 0.98 and water concentrations of 46, 48, 50 M. Longer Ag deposition (10 min) minimized lateral pitting. Length increased with time but plateaued as tip etch rates rose or base rates declined (Fig. 5). At χ = 0.92, tip etch rates increased sharply, leading to erratic length evolution. Lower χ values produced defect‑free SiNWs, whereas higher χ (0.98) caused lateral pits. The overall trend mirrored that in Fig. 4, confirming the key role of χ in balancing tip and base etching.
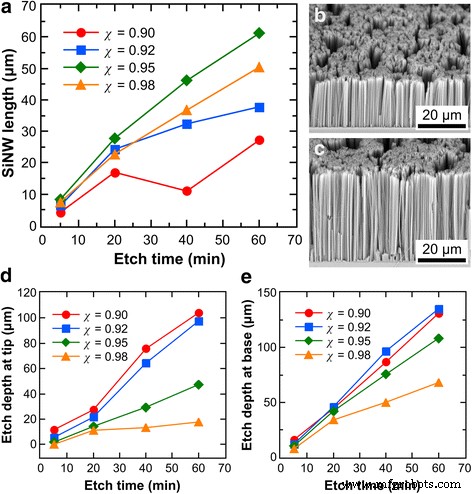
Evolution of SiNW length with time for different HF–H₂O₂ molar ratios at 48 M H₂O. a Effect of etch time on SiNW length. b, c SEM images after 1 h in solutions with χ = 0.9 and 0.98. d, e Bulk Si thickness relative to tip and base over time.
Fabrication of Highly Doped SiNWs with Controlled Porosity
Porosity is a critical parameter for SiNW‑based devices. It is influenced by H₂O₂ concentration [14–17, 20], etch time [14, 16–18, 20], temperature [20], HF/H₂O₂ ratio [19], and wafer resistivity [17]. In our Ag‑based MACE, porosity also scales with AgNP loading. Adjusting χ and water concentration independently of length allows precise porosity tuning. We compared porosity for χ = 0.92, 0.95, 0.98 (water fixed at 48 M) and water = 46, 48, 50 M (χ fixed at 0.95) while keeping SiNW length at 20 µm (etch times adjusted accordingly). Porosity inversely correlates with χ and directly with water concentration (Fig. 7).

TEM images of 20‑µm SiNWs showing porosity variation with (a)–(c) HF–H₂O₂ molar ratios (0.92, 0.95, 0.98) at 48 M H₂O, and (d)–(f) H₂O concentrations (46, 48, 50 M) at χ = 0.95. Low χ yields higher porosity; high water concentration also increases porosity, but very dilute etchants risk shell dissolution.
High porosity at χ = 0.95 reflects enhanced Ag re‑nucleation, ion‑induced etching, and hole diffusion compared to χ = 0.98. The unexpectedly lower porosity at χ = 0.92 likely arises from rapid shell collapse due to aggressive HF/H₂O₂ etching of highly porous structures, exposing the less porous core. Higher water concentrations prolong the etch duration, allowing more time for porosification while limiting aggressive dissolution inside pores, thereby preserving the shell (Fig. 7d–f).
Surface roughness along the nanowire axis consistently increases from base to tip, in line with longer exposure to the etchant [16, 19]. Porous, tapered SiNWs exhibit more pronounced tapering, while biconic or hourglass profiles correlate with higher porosity. To obtain highly porous SiNWs with small diameters, a more inert catalyst such as Au may be preferable [32].
In summary, porosity control in highly doped SiNWs of a specified length is achievable by selecting appropriate χ and water concentrations. Low porosity is obtained with high χ and low water concentration; high porosity requires moderate χ (~0.95) and higher water concentration, avoiding the shell collapse seen at very low χ. These guidelines, combined with Ag deposition control, offer a versatile toolbox for tailoring SiNW properties for diverse applications.
Conclusions
We successfully fabricated porous SiNWs from degenerately doped p‑type Si wafers using electroless Ag‑catalyzed MACE in HF–H₂O₂. Systematic variation of Ag deposition time, HF–H₂O₂ molar ratio (χ), and water concentration revealed the following key insights:
- There exists an optimal Ag deposition amount that yields defect‑free SiNWs; excess or insufficient deposition adversely affects morphology and etch rate.
- Etch rate varies non‑monotonically with Ag loading, providing an additional lever to tune porosity.
- The χ window (0.85–0.98 for 46–48 M water; >0.9 for 50 M) defines the conditions for uniform SiNW arrays.
- Etching kinetics at the tip and base are governed by primary Ag‑mediated dissolution, re‑nucleated Ag etching, hole diffusion, and reactant diffusion, leading to a gradual reduction in etch rate over time.
- By manipulating χ and water concentration, we independently control porosity and length, enabling the design of SiNWs with tailored properties for optoelectronic, thermoelectric, or biomedical devices.
These findings are broadly applicable to MACE with Au catalysts, where metal‑ion‑induced etching is suppressed and hole diffusion dominates porosification.
Abbreviations
- AgNP:
Ag nanoparticle
- DI:
Deionized
- MACE:
Metal‑assisted chemical etching
- SEM:
Scanning electron microscopy
- SiNW:
Silicon nanowire
- TEM:
Transmission electron microscopy
- χ:
HF–oxidant molar ratio
Nanomaterials
- Selective Laser Sintering (SLS): Cutting‑Edge Technologies, Advanced Materials, and Expanding Applications
- Fabrication of Ordered Au‑Capped GaAs Nanopillar Arrays via Metal‑Assisted Chemical Etching
- Electroless HF/AgNO3 Etching of n‑Si(100) Wafers: High‑Density Silicon Nanowire Arrays with Low Reflectance and Ohmic Conductance
- Tuning Morphology, Optics, and Conductivity of Al₂O₃/ZnO Nanolaminates Through Bilayer Thickness Control
- Large-Scale Silicon Nanowire Arrays on 6‑inch Mono‑ and Multi‑Crystalline Solar Cells via Enhanced Metal‑Assisted Chemical Etching
- Advanced c‑Si/c‑SiGe Wheel Test Structures for Precise Evaluation of Wet Etchant Anisotropy and Selectivity
- Accelerated, Directionally Controlled Silicon Nanowire Arrays via Electric Field‑Assisted Metal‑Assisted Chemical Etching
- Enhancing Thermoelectric Performance: Silicon Substrates with Highly Doped Porous Si via Metal-Assisted Chemical Etching
- Step Stencil Milling: Superior to Laser Cutting & Chemical Etching for PCB Production
- From Inception to Innovation: Tracing the Evolution of CNC Machining



