Optimizing Tribological Performance of Layered Zirconium Phosphate Nanoplatelets in Oil Through Tailored Surface and Interlayer Functionalization
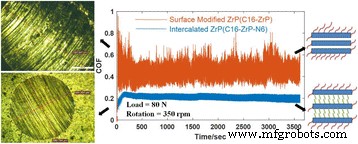
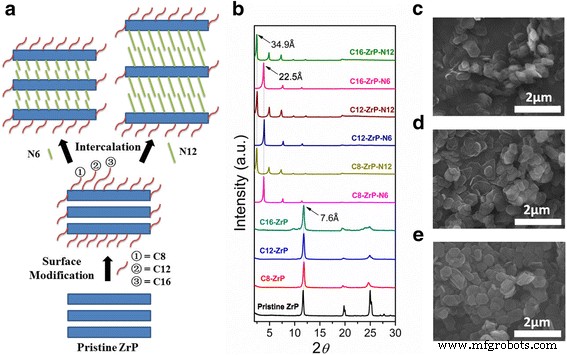
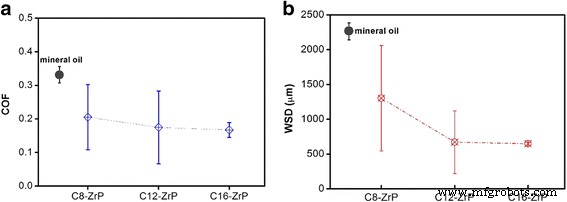
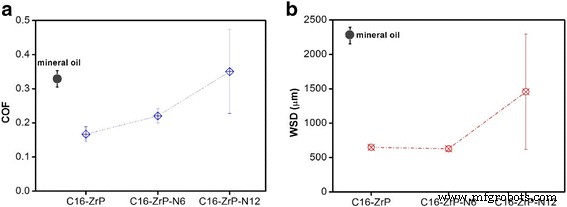
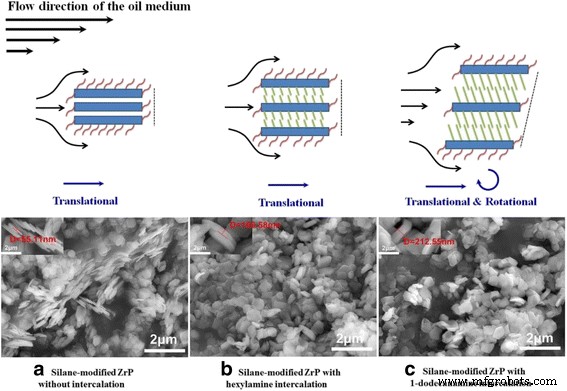
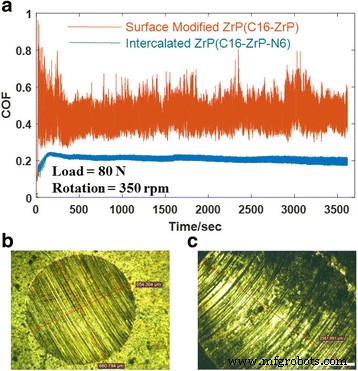
Two‑dimensional inorganic layered nanoplatelets are emerging as highly effective lubricants in both solid and oil‑dispersed systems. In this study we systematically examined how surface grafting and interlayer intercalation influence the friction‑reduction and anti‑wear properties of α‑zirconium phosphate (ZrP) nanoplatelets in mineral oil. Pristine ZrP was first functionalized with silane coupling agents bearing C8, C12, or C16 alkyl chains to improve oil solubility, and then the modified plates were intercalated with hexylamine (N6) or 1‑dodecanamine (N12) to expand the interlayer spacing. Tribological tests revealed that a longer alkyl chain on the outer surface, coupled with a modest increase in interlayer distance, yields the most favorable friction coefficients and wear scar diameters, particularly under higher load conditions. These findings demonstrate that judicious control of surface and interlayer chemistry can tune the performance of 2D nanoplatelets in lubricating oils, providing a pathway for designing next‑generation nanolubricants. Nanolubricating oils—conventional oils enhanced with inorganic nanoparticles—have attracted significant interest for their superior friction‑ and wear‑reducing capabilities compared with pure organic additives. Common nanofillers include zero‑dimensional oxides and fullerenes, one‑dimensional nanotubes, and two‑dimensional (2D) nanoplatelets such as graphene, MoS₂, and layered metal phosphates. The latter offer anisotropic geometry and interlayer sliding, making them particularly promising for lubrication. α‑Zirconium phosphate (ZrP) and its derivatives have recently emerged as a powerful 2D class, showing friction coefficients as low as 0.17–0.20 and wear scar diameters below 700 µm under heavy loads, outperforming MoS₂ and graphite in mineral oils. ZrP’s robust lattice and strong interlayer bonding contribute to its durability in oil media, while surface adsorption of lubricants can further enhance viscosity and film formation. Despite these advantages, practical application requires a deeper understanding of how particle size, surface chemistry, and interlayer spacing affect dispersion, rheology, and tribological performance. ZrP is inherently hydrophilic; therefore, oil‑soluble surfactants are essential for stable dispersions. Previous work intercalated alkyl amines into ZrP, but this approach simultaneously modifies the surface and the interlayer, obscuring their individual effects. Here we separate these two modifications to isolate their distinct contributions to lubrication. Pristine ZrP was prepared hydrothermally (Sun et al.). ZrOCl₂·8H₂O (4.0 g) was mixed with 40.0 mL of 6.0 M H₃PO₄, sealed in a Teflon‑lined vessel, and heated at 200 °C for 24 h. After cooling, the product was washed five times by centrifugation with deionized water to remove excess acid, dried at 80 °C for 24 h, and ground into fine powder. This material is referred to as pristine ZrP. Pristine ZrP (10 g) was dispersed in toluene with 20 g of one of three alkyl silanes (C8, C12, or C16). The mixture was heated at 100 °C with stirring for 48 h, then the solvent was removed by centrifugation and the solid was washed three times with petroleum ether. The dried powders were obtained at 70 °C for 24 h and ground again. The resulting surface‑modified samples are denoted C8‑ZrP, C12‑ZrP, and C16‑ZrP. Two grams of each surface‑modified ZrP were mixed with either 5 g hexylamine (N6) or 10 g 1‑dodecanamine (N12) in 60 mL hexane. The mixtures were sonicated at 40 kHz for 3 h, then washed three times with petroleum ether and dried at 70 °C for 24 h. The intercalated products are labeled C8‑ZrP‑N6, C8‑ZrP‑N12, C12‑ZrP‑N6, C12‑ZrP‑N12, C16‑ZrP‑N6, and C16‑ZrP‑N12. A 1.0 wt% master batch of each ZrP sample was made by mixing the powder with mineral oil under mechanical stirring, followed by 20 min ultrasonication. The master batch was then diluted to 0.1 wt% with base mineral oil under ultrasonication, producing the test oils. Crystal structures were assessed by X‑ray diffraction (Rigaku DMAX‑2500). Morphology was examined by SEM (TESCAN Vega3) at 30 kV. Surface functional groups were identified by FTIR (PerkinElmer Spectrum Two). Tribological performance was measured with a Bruker UMT‑2 four‑ball tester (ASTM D4172) under 70 N load and 350 rpm for 1 h. Coefficients of friction (COFs) were recorded at 100 Hz, and wear scars were imaged optically. Surface roughness of the steel balls was 155.0 ± 14.8 nm. Schematic of the four‑ball tribology setup. The pristine ZrP layers are terminated with hydroxyl groups, enabling both hydrogen bonding between layers and surface functionalization. Conventional amine intercalation attaches to both the outer surface and the interlayer, so we first grafted silanes onto the surface to lock the exterior while leaving the interlayer free for subsequent amine insertion. XRD patterns confirmed that C8‑, C12‑, and C16‑ZrP maintained the original interlayer spacing (7.6 Å), indicating that the silanes did not intercalate. After amine intercalation, the spacing expanded to 22.5 Å with hexylamine and 34.9 Å with 1‑dodecanamine, as expected from the longer alkyl chains. Surface and interlayer modifications of ZrP nanoplatelets: (a) schematic; (b) XRD patterns; (c) SEM of pristine; (d) surface‑modified; (e) surface‑modified plus intercalated. All modified samples exhibited platelet morphology with diameters of 600–800 nm; the modifications did not alter the lateral dimensions. Photographs after 2 h dispersion (Fig. 3) demonstrated that only surface‑modified ZrP remained stably dispersed, whereas pristine ZrP sedimented rapidly. Dispersion of ZrP in mineral oil: (a) immediately after mixing, (b) after 2 h. Samples: C16‑ZrP, C16‑ZrP‑N6, and pristine ZrP. Concentration = 0.1 wt%. Tribological testing (70 N, 350 rpm, 1 h) revealed that longer alkyl chains on the surface reduced COF and wear scar diameter (WSD). C8‑ZrP showed a COF of 0.20, 40% lower than base oil (0.33). C12‑ZrP and C16‑ZrP achieved COFs of 0.18 and 0.17, respectively, while WSDs dropped to 700 µm and 600 µm, respectively—reduction of 70% and 74% relative to base oil. The best performance came from C16‑ZrP, which also exhibited the smallest data scatter, indicative of superior dispersion stability. Friction coefficients (a) and wear scar diameters (b) for surface‑modified ZrP samples. Intercalation effects were probed with the C16‑ZrP series. Adding hexylamine (C16‑ZrP‑N6) slightly increased COF to 0.21 but maintained a low WSD of 550 µm. In contrast, 1‑dodecanamine (C16‑ZrP‑N12) raised COF to 0.35—exceeding base oil—and WSD increased to 1,400 µm. These results indicate that a modest interlayer expansion (22.5 Å) does not degrade performance, whereas a large expansion (34.9 Å) compromises lubrication, likely due to reduced interlayer cohesion and altered aspect ratio. Friction coefficients (a) and wear scar diameters (b) for C16‑ZrP, C16‑ZrP‑N6, and C16‑ZrP‑N12. Aspect ratio calculations (diameter/ thickness) showed a decrease from ~10 for pristine ZrP to ~5–6 after hexylamine and ~3 after 1‑dodecanamine intercalation. Higher aspect ratios facilitate alignment and shear‑induced lubrication, whereas lower ratios lead to increased rotation and less effective load support. Proposed rheology of surface‑modified ZrP with (a) no intercalation, (b) hexylamine, (c) 1‑dodecanamine. Under an increased load of 80 N, C16‑ZrP‑N6 maintained a stable COF of ~0.20 and a WSD of 650 µm, whereas C16‑ZrP’s COF surged to ~0.45 with a 2,600 µm wear scar, confirming the advantage of modest interlayer expansion under heavy load. SEM of wear scars (Fig. 9) corroborated the smoother surface for C16‑ZrP‑N6 compared to C16‑ZrP. Friction coefficients at 80 N (a) and wear scar images for C16‑ZrP‑N6 (b) and C16‑ZrP (c). SEM images of wear scars for C16‑ZrP‑N6 (a) and C16‑ZrP (b) under 80 N. We have delineated how surface and interlayer chemistry independently affect the tribological performance of layered ZrP nanoplatelets in mineral oil. Surface grafting with longer alkyl silanes improves dispersion and reduces both COF and WSD, with C16‑ZrP delivering the best baseline performance. Subsequent intercalation with hexylamine preserves these benefits and even enhances performance under heavier loads, whereas 1‑dodecanamine intercalation, which dramatically expands the interlayer spacing, degrades lubrication. These results underscore the importance of finely tuning both surface hydrophobicity and interlayer spacing to engineer high‑performance nanolubricants.Abstract
Background
Methods
Synthesis of Pristine ZrP Nanoplatelets
Surface Modification of Pristine ZrP Nanoplatelets
Interlayer Modification of ZrP Nanoplatelets
Preparation of Nanolubricating Oils
Characterization
Results and Discussion


Conclusions
Nanomaterials
- Scratch & Sniff: The Science and Craft of Micro-Encapsulated Scent Printing
- Surface Properties Govern Oil Transport in Nanochannels: Molecular Dynamics Insights
- Enhancing Cell Adhesion on Polyetheretherketone (PEEK) through Gold Coating and Argon Plasma Treatment
- Optimizing ZnO Film Morphology and Properties via Interfacial Layer Design
- Hollow‑Structured LiNb3O8 Photocatalysts: Synthesis, Characterization, and Superior Methylene Blue Degradation
- How Elastic Stiffness and Surface Adhesion Govern Nanoparticle Bouncing Behavior
- How Surface Charge Alters Oil Contact Angles in Water‑Decane–Silicon Dioxide Systems
- Unveiling Surface-Related Excitons and Lasing in CdS Nanostructures
- High‑Surface‑Area Yolk‑Shell Calcium Phosphate Microspheres from Organic Phosphorous and Calcium Sources for Enhanced HEL Adsorption
- Leveraging IoT to Boost Performance in the Oil & Gas Industry



