High‑Efficiency Tumor‑Targeted Photo‑Chemo Theranostic Nanoparticles: Artesunate‑Loaded, ICG‑Conjugated Human Serum Albumin
Abstract
We report a tumor‑specific theranostic platform that combines four clinically approved agents—artesunate (Arte), human serum albumin (HSA), folic acid (FA), and indocyanine green (ICG)—into a single nanoparticle (FA‑IHA NPs). These nanoparticles exhibit exceptional photo‑ and physicochemical stability. ICG imparts near‑infrared (NIR) fluorescence for imaging and confers photothermal (PTT) and photodynamic (PDT) therapeutic capabilities under a single 808 nm laser. Concurrently, 808 nm irradiation triggers controlled Arte release, amplifying chemotherapeutic efficacy. In vitro and in vivo studies demonstrate rapid cellular uptake and selective tumor accumulation, verified by confocal microscopy and NIR imaging in HepG2 xenograft mice. A single NIR irradiation session (808 nm, 1 W/cm²) combined with FA‑IHA NPs completely suppresses tumor growth without recurrence. These findings establish FA‑IHA NPs as a promising platform for clinical translation of image‑guided photo‑chemo therapy.
Background
Imaging‑guided photo‑chemo therapy (IGPC) has emerged as a compelling strategy for personalized oncology, marrying precise tumor localization with multimodal treatment while minimizing collateral damage (1–4). A successful IGPC platform must be: (i) multifunctional, integrating diagnostic and therapeutic modalities; (ii) biocompatible, stable, and tumor‑selective (5–8). Fluorescence imaging, owing to its high sensitivity and favorable temporal resolution, is commonly employed for real‑time guidance (9–16).
Photothermal therapy (PTT) and photodynamic therapy (PDT) can be unified under NIR irradiation, delivering both heat and reactive oxygen species (ROS) to ablate tumor cells (17–19). Nonetheless, monotherapy often fails to eradicate residual disease, leading to relapse (17–19). Conventional chemotherapy offers systemic cytotoxicity but suffers from non‑specificity and associated toxicities (20–22). Thus, IGPC, which combines localized phototherapy with targeted chemotherapy, holds promise for overcoming these limitations.
Nanomedicine has produced a variety of IGPC theranostics, including indocyanine green (ICG), metal‑based nanoparticles, carbon nanomaterials, and polymeric systems (23–27). ICG, an FDA‑approved NIR dye, is widely used clinically for cardiac output, liver function, and ophthalmic angiography (28–29). Its strong absorption at 800 nm facilitates efficient PTT‑PDT. However, free ICG is unstable, rapidly clears from circulation, self‑bleaches, and lacks tumor specificity (31–32). Encapsulation in carriers such as micelles, polymers, or protein nanostructures has improved its performance, yet the need for more biocompatible, targeted ICG‑based composites persists (33–34).
Here, we engineered a tumor‑targeted IGPC agent by covalently linking FA and ICG to HSA nanoparticles, simultaneously encapsulating Arte—a natural antimalarial compound with proven anticancer activity against liver, lung, and breast cancers (35). The resulting FA‑IHA NPs harness FA‑mediated receptor uptake, HSA’s excellent biocompatibility, and the dual therapeutic power of ICG and Arte. This multifunctional construct offers NIR imaging, PTT‑PDT, and controlled chemo‑delivery, positioning it as a versatile platform for clinical translation.
Methods
Materials
Reagents included EDC, NHS, and Arte (≥ 99%) from Sigma‑Aldrich; DAPI and CCK‑8 from Aladdin; NH₂‑PEG₂₀₀₀‑COOH and NH₂‑PEG₂₀₀₀‑FA from Xi’an Ruixi; DMEM and PBS from Gibco; ICG‑NHS from Dojindo. All chemicals were used as received.
Synthesis and Characterization of FA‑IHA NPs
Arte was dissolved in DMSO, then added to 15 mL water. HSA (10 mg) was introduced and stirred for 3 h at room temperature, followed by cross‑linking with 150 µL 0.5% glutaraldehyde. The mixture was dialyzed against distilled water (MWCO 8–12 kDa) for 24 h to yield Arte‑loaded HSA (Arte‑HSA). Activation of HSA’s carboxyl groups with EDC/NHS was followed by conjugation with NH₂‑PEG₂₀₀₀‑FA (3 h, 4 °C) and subsequent attachment of ICG‑NHS (30 min, RT). Final FA‑ICG‑HSA@Arte (FA‑IHA NPs) were purified by 24‑h dialysis in deionized water. Arte and ICG loading efficiencies were quantified by UV‑vis spectroscopy (loading % = W₁/W₂ × 100%).
Morphology was assessed by TEM (Hitachi). Hydrodynamic size and ζ‑potential were measured by DLS (Malvern Zetasizer). Absorbance spectra were recorded on a Shimadzu UV‑vis spectrophotometer. Photothermal responses were measured using an 808 nm CW laser (1 W/cm²) with temperature monitored by a Fluke thermocouple. Photostability and fluorescence longevity were evaluated over 5 cycles and 30 days, respectively.
Thermal and pH‑Triggered Arte Release
FA‑IHA NPs (50 µg/mL) were incubated at pH 6.5, pH 7.4, and pH 6.5 + NIR (808 nm, 1 W/cm², 1 min pulse) over 36 h. Arte release was quantified by UV‑vis at 287 nm in the supernatant.
Detection of Singlet Oxygen Production
DPBF (15 µL) was mixed with 1 mL of 10 µg/mL ICG or FA‑IHA NPs and irradiated (808 nm, 1 W/cm², 5 min). Absorbance decrease at 410 nm over time reflected singlet oxygen generation.
Cell Culture and Cellular Uptake
HepG2 cells (ATCC) were cultured in DMEM with 10% FBS, 1% penicillin‑streptomycin at 37 °C, 5% CO₂. For uptake studies, cells were incubated with free ICG, IHA NPs, or FA‑IHA NPs (0.05 mg/mL ICG) for 6 h, washed, fixed, DAPI‑stained, and imaged by confocal microscopy. Flow cytometry quantified internalization rates.
Generation of Intracellular ROS
HepG2 cells (2 × 10⁵ cells/mL) were treated with PBS, Arte, FA‑HA‑NPs, free ICG, IHA‑NPs, or FA‑IHA NPs for 12 h, then irradiated (808 nm, 1 W/cm², 5 min). DCFH‑DA (5 µg/mL) was added for 30 min, followed by flow cytometry and fluorescence microscopy to assess ROS levels.
In Vitro Tumor Combinational Photo‑Chemo Therapy
HepG2 cells (2 × 10⁴ cells/well) were treated with free ICG, Arte, IHA NPs, or FA‑IHA NPs (Arte 0–30 µg/mL) for 6 h, then irradiated (808 nm, 1 W/cm², 5 min) or left untreated. Cell viability was measured by CCK‑8 after 24 h. Live/dead staining (calcein‑AM/PI) confirmed cytotoxicity via confocal imaging.
Animal Model and In Vivo Fluorescence Imaging
Balb/c nude mice (n=5) were subcutaneously injected with 1 × 10⁶ HepG2 cells. IVIS Spectrum imaging (Caliper) tracked NIR fluorescence at 10 min, 6 h, 12 h, 24 h, and 48 h post‑tail‑vein injection of free ICG, IHA NPs, or FA‑IHA NPs.
In Vivo Tumor Combinational Photo‑Chemo Therapy
Mice were randomized into six groups (n=5): PBS, Arte, FA‑IHA NPs, ICG + NIR, IHA + NIR, FA‑IHA + NIR (equal Arte dose). NIR irradiation (808 nm, 1 W/cm², 5 min) was applied at 24 h and 48 h. Tumor volumes were recorded every 4 days and expressed relative to baseline. Post‑treatment, major organs were harvested for H&E histology.
Results and Discussion
Synthesis and Characterization of FA‑IHA NPs
Figure 1 schematically depicts FA‑IHA NPs’ design and therapeutic workflow. The nanoparticles were assembled via a simple, biocompatible method, integrating ICG for NIR imaging and phototherapy, and Arte for chemotherapeutic action.
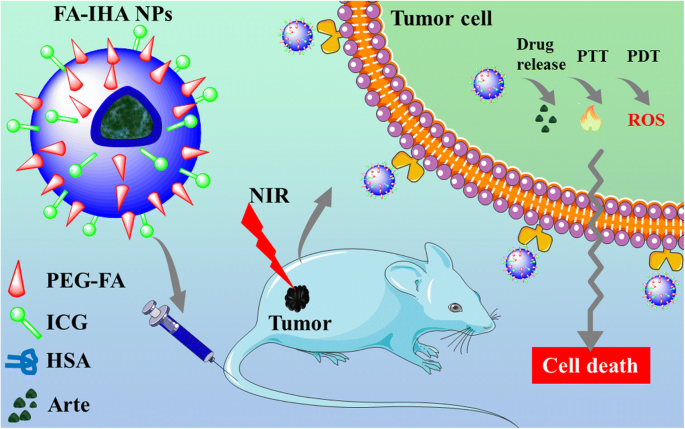
Schematic representation of FA‑IHA NPs use for imaging‑guided tumor‑targeted combinational photo‑chemo therapy in vitro and in vivo
Transmission electron microscopy revealed monodisperse, spherical particles (~131.2 nm). Dynamic light scattering confirmed a hydrodynamic diameter of 131 ± 2.3 nm across water, PBS, and cell medium, with a stable ζ‑potential of –29.2 ± 1.13 mV. Size remained unchanged over 7 days, indicating robust colloidal stability conferred by PEG and HSA coating. UV‑vis spectra displayed characteristic peaks of Arte and ICG, confirming successful loading (Arte 98.6 ± 3.1 %, ICG 56.9 ± 2.4 %). Fluorescence spectra matched free ICG, confirming preserved photophysical properties.
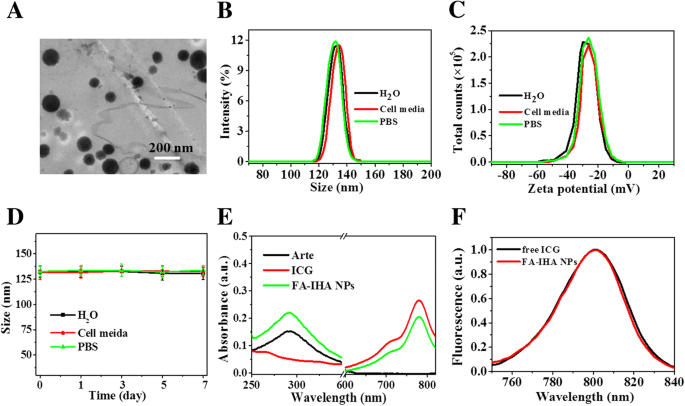
a TEM image of FA‑IHA NPs. b, c Size and ζ‑potential distribution of FA‑IHA NPs in water, cell medium, and PBS. d FA‑IHA NPs size change in water, cell medium, and PBS. e Absorbance spectra of free ICG, Arte, and FA‑IHA NPs. f Fluorescence spectra of free ICG and FA‑IHA NPs
Photothermal evaluation showed that FA‑IHA NPs (equal ICG content) raised the temperature by ~36 °C within 5 min under 808 nm (1 W/cm²) irradiation, while free ICG displayed a similar rise and water barely increased (<4 °C). Photostability tests revealed that FA‑IHA NPs maintained absorbance over five irradiation cycles, whereas free ICG lost intensity, underscoring the protective effect of covalent conjugation and HSA/PEG encapsulation. Fluorescence longevity over 30 days at 4 °C showed a 28 % reduction for FA‑IHA NPs versus an 88 % drop for free ICG, highlighting superior photostability.
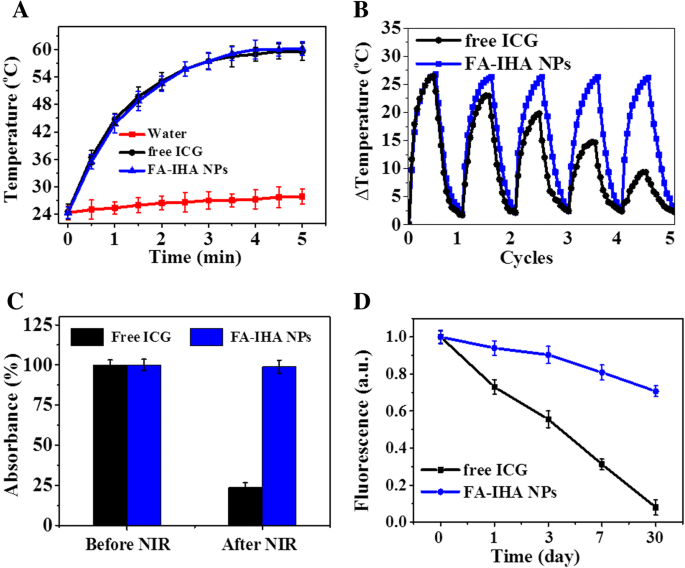
a Photothermal heating curves of water, ICG, and FA‑IHA NPs under 5 min 808 nm laser irradiation (1 W/cm²). b Temperature variations of ICG and FA‑IHA NPs after five continuous 5‑min irradiation cycles. c Absorbance change of FA‑IHA NPs at 780 nm before and after five irradiation cycles. d ICG and FA‑IHA NPs fluorescence change over 30 days
DPBF assays demonstrated that FA‑IHA NPs produced 58 % of the standard singlet oxygen yield within 5 min of NIR exposure, surpassing free ICG (35 %). This enhancement reflects the synergistic effect of dual phototherapy.
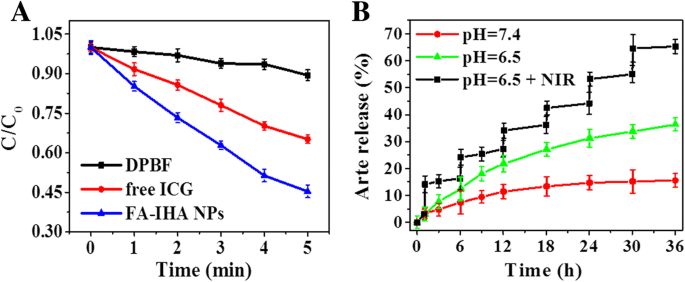
a Normalized DPBF absorbance in the presence of ICG, FA‑IHA NPs, and blank sample under 808 nm laser irradiation (1 W/cm²). b Release kinetics of Arte from FA‑IHA NPs under pH 7.4 and pH 6.5 with or without NIR laser irradiation, respectively
Under acidic conditions (pH 6.5) and NIR irradiation, Arte release from FA‑IHA NPs increased from 11.6 % (pH 7.4) to 68.4 % (pH 6.5 + NIR). The dual triggers—thermal expansion of HSA and proton‑mediated surface charge alteration—facilitate controlled drug release (37–38).
Cell Uptake and Detection of Intracellular ROS
Confocal imaging revealed pronounced red ICG fluorescence in HepG2 cells treated with FA‑IHA NPs, surpassing free ICG and IHA NPs. Flow cytometry quantified a 52.3 % uptake rate for FA‑IHA NPs, markedly higher than IHA NPs (25.2 %) and free ICG (3.9 %)—a clear indication of FA‑mediated receptor targeting (39–41).
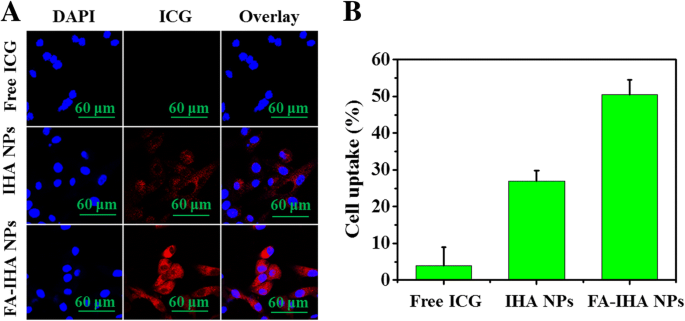
a Confocal fluorescent images of HepG2 cells after incubation with free ICG and IHA NPs, and FA‑IHA NPs. Red and blue colors represent ICG fluorescence and DAPI‑stained nuclei, respectively. b Flow cytometry measurement of ICG fluorescence intensities in HepG2 cells after incubation with free ICG, IHA NPs, and FA‑IHA NPs
ROS production, assessed with DCFH‑DA, was significantly higher in FA‑IHA NPs‑treated cells following 5 min NIR irradiation compared to all other groups, confirming enhanced photodynamic activity (Figure 6a,b).
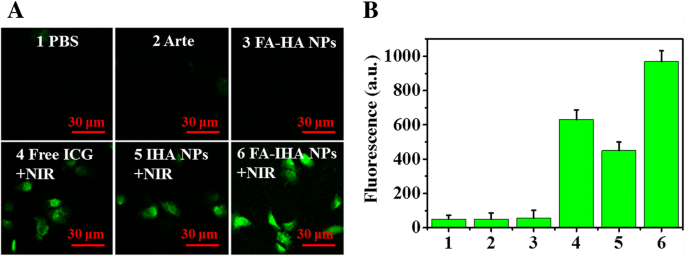
a Fluorescence images of ROS production in cancer cells treated with various drugs, and b corresponding fluorescent intensity: (1) PBS, (2) Arte, (3) FA‑HA NPs, (4) free ICG + NIR, (5) IHA NPs + NIR, and (6) FA‑IHA NPs + NIR
In Vitro Tumor Combinational Photo‑Chemo Therapy
FA‑IHA NPs achieved the highest temperature rise (~31 °C) among all treatments after 5 min NIR exposure, correlating with superior photothermal performance (Figure 7a). Without irradiation, Arte, IHA NPs, and FA‑IHA NPs displayed concentration‑dependent cytotoxicity, whereas free ICG remained inert. Post‑irradiation, cell viability dropped dramatically in the FA‑IHA NPs group, attributable to synergistic chemo‑photothermal effects. Calcein‑AM/PI staining confirmed near‑complete cell death in FA‑IHA NPs + NIR, whereas other groups retained viable cells (Figure 7d).
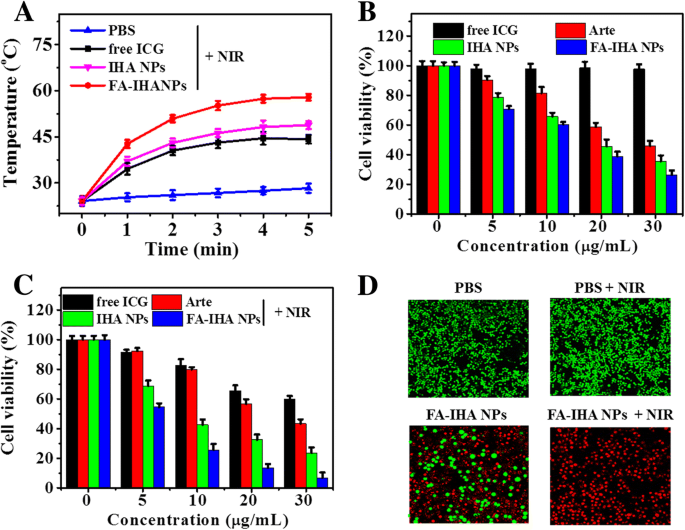
a Temperature change curves of PBS, free ICG, IHA NPs, and FA‑IHA NPs‑treated cells in 96‑well plates after 5 min NIR irradiation. b, c Cell viability of cells treated with free ICG, Arte, IHA NPs, and FA‑IHA NPs without or with 808 nm laser irradiation (5 min, 1 W/cm²), respectively. d Calcein‑AM/PI dual‑staining images of cells after treatment with PBS (control), PBS + NIR, FA‑IHA NPs, and FA‑IHA NPs + NIR, respectively
In Vivo Fluorescence Imaging
IVIS imaging revealed strong whole‑body fluorescence within 10 min post‑injection of all formulations. Tumor fluorescence peaked at 24 h, with FA‑IHA NPs exhibiting the highest signal at every time point, confirming FA‑driven tumor accumulation (Figure 8b). Ex vivo organ analysis at 24 h showed predominant liver uptake across all groups, consistent with hepatic clearance pathways (Figure 8c).
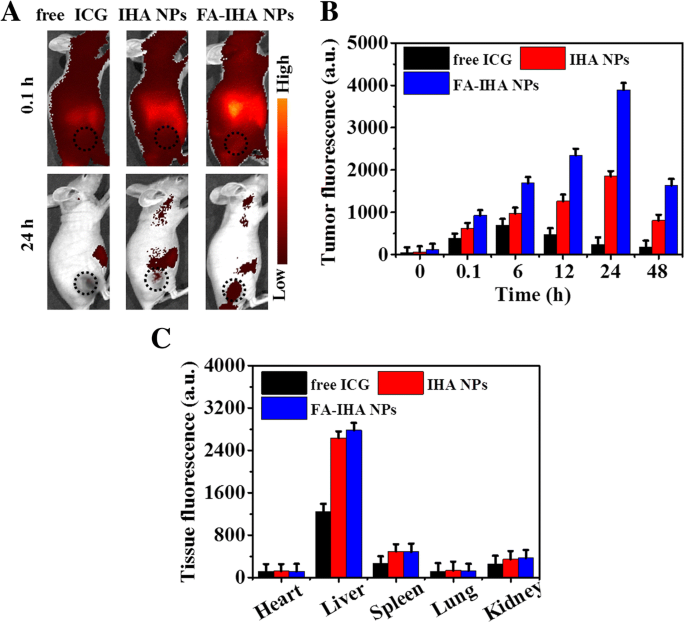
a Representative fluorescence images of tumor‑bearing mice after tail‑vein injection with free ICG, IHA NPs, and FA‑IHA NPs. The black dashed circles indicate the tumor region. b Quantitative in vivo analysis of the fluorescence signal in the tumor regions of mice treated with free ICG, IHA NPs, and FA‑IHA NPs as a function of injection time. c The fluorescence signal of major organs including heart, liver, spleen, lungs, and kidney
In Vivo Tumor Combinational Photo‑Chemo Therapy
Thermal imaging at 24 h post‑injection under NIR irradiation showed the greatest temperature increase (~22.1 °C) for FA‑IHA NPs, confirming potent photothermal conversion (Figure 9a,b). Two irradiation sessions (day 0 and day 2) led to complete tumor regression in the FA‑IHA + NIR group, with no relapse over 90 days and a 100 % survival rate, whereas all other groups failed to achieve sustained suppression (Figure 9c,d). Histological examination revealed no significant organ pathology in the FA‑IHA + NIR group, underscoring negligible systemic toxicity (Figure 10).
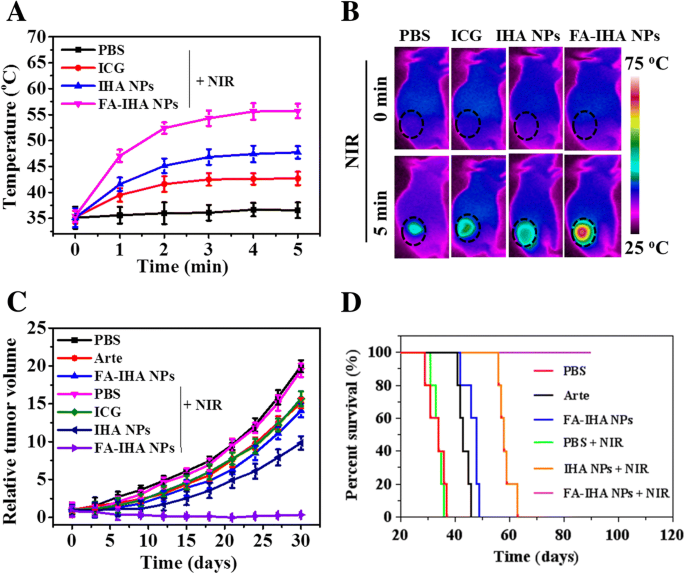
a Tumor region temperatures in tumor‑bearing mice after tail‑vein injection of PBS, ICG, IHA NPs, and FA‑IHA NPs at 24 h under 5 min of NIR irradiation (808 nm, 1 W/cm²). b Thermal images of tumor‑bearing mice after tail‑vein injection of PBS, ICG, IHA NPs, and FA‑IHA NPs at 24 h under 5 min of NIR irradiation (808 nm, 1 W/cm²). c HepG2 xenograft tumor growth profile after intravenous injection of PBS, Arte, ICG, IHA NPs, and FA‑IHA NPs with or without 5 min of NIR irradiation (808 nm, 1 W/cm²). d Survival rate of tumor‑bearing mice after tail‑vein injection with PBS, free Arte, ICG, IHA NPs, and FA‑IHA NPs with or without 5 min of NIR irradiation (808 nm, 1 W/cm²)
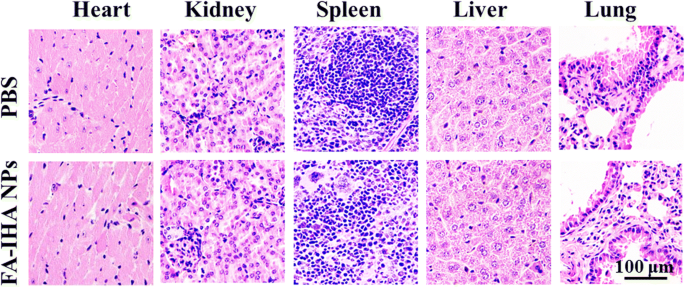
H&E‑stained tissue sections of major organs, including heart, liver, spleen, lungs, and kidney from mice treated with PBS and FA‑IHA NPs
Conclusions
We have engineered a multifunctional theranostic nanoparticle that covalently conjugates FA and ICG while encapsulating Arte within HSA. The FA‑IHA NPs demonstrate robust colloidal and photothermal stability, preserve NIR fluorescence, and release Arte upon NIR irradiation. FA‑mediated receptor targeting yields high cellular uptake and tumor accumulation, enabling a synergistic photothermal‑photodynamic‑chemotherapeutic assault that eradicates tumors in vitro and in vivo without recurrence. These results position FA‑IHA NPs as a highly promising platform for clinical translation of image‑guided, tumor‑targeted photo‑chemo therapy.
Abbreviations
- Arte:
Artesunate
- FA:
Folic acid
- HSA:
Human serum albumin
- ICG:
Indocyanine green
- NIR:
Near infrared
- PDT:
Photodynamic therapy
- PEG:
Polyethylene glycol
- PTT:
Photothermal therapy
Nanomaterials
- Resveratrol-Loaded Human Serum Albumin Nanoparticles with PEG–RGD Targeting: Extended Circulation, Enhanced Biocompatibility, and Potent Pancreatic Tumor Suppression
- PEG‑Coated Gold Nanostars: A Safe, High‑Contrast Agent for CT Imaging with Rapid Renal Clearance
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Her2‑Functionalized Gold‑Nanoshelled Magnetic Hybrid Nanoparticles: Dual‑Modal US/MR Imaging and Targeted Photothermal Therapy for Breast Cancer
- Targeted Ultrasound Imaging & Therapy of Ovarian Tumors Using pH‑Responsive Perfluoropentane Protein Nanoparticles
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy
- Targeted Curcumin Nanoparticles Activated by Low‑Intensity Focused Ultrasound for pH‑Responsive Theranostics in Ovarian Cancer
- Rapid Synthesis of BiF3@Ln (Gd, Yb, Er) PVP-Coated Nanoparticles for Advanced, Low‑Toxicity CT Imaging
- Nanoparticles: Transforming Cancer Diagnosis and Treatment with Precision



