Targeted Co‑Delivery of 5‑Fluorouracil and LY294002 via PEG‑Lipid Nanoparticles Enhances Antitumor Efficacy in Esophageal Squamous Cell Carcinoma
Abstract
We engineered a PEGylated nanoliposome co‑encapsulating the chemotherapeutic agent 5‑fluorouracil (5‑FU) and the autophagy inhibitor LY294002 (LY) to treat esophageal squamous cell carcinoma (ESCC). Comprehensive physicochemical and biological characterizations demonstrated successful dual‑drug loading and a controlled release profile, with LY released slightly faster than 5‑FU. The liposomes exhibited receptor‑mediated uptake, confirmed by confocal microscopy and FACS, and delivered higher intracellular concentrations of both agents. In vitro, the nanoliposome (FLNP) induced greater cytotoxicity and apoptosis (~48%) than either drug alone or their free combination, as evidenced by MTT, annexin V/PI, and Hoechst staining. Western blotting revealed upregulation of caspase‑3 and PARP and downregulation of Bcl‑2, confirming apoptosis induction. In a xenograft model, FLNP achieved superior tumor growth inhibition, reducing tumor volume to ~500 mm³ versus ~1,400 mm³ in controls (p < 0.0001). These findings support the promise of programmed co‑delivery of autophagy inhibition and chemotherapy via a single nanocarrier to overcome multidrug resistance in ESCC.
Background
Esophageal squamous cell carcinoma (ESCC) remains a major health burden in East Asia, with a 5‑year survival rate of only ~25 % (1,2). While surgery is the cornerstone of treatment, adjuvant chemotherapy can significantly improve outcomes (3,4). However, monotherapy often fails due to tumor heterogeneity and intrinsic resistance (5). Combination regimens that target distinct cellular pathways are therefore essential for synergistic efficacy (6,7).
Autophagy, a lysosomal degradation process, is frequently upregulated in cancer cells under stress and can protect them from chemotherapeutic agents (8). Inhibition of autophagy has been shown to sensitize tumor cells to cytotoxic drugs, thereby overcoming multidrug resistance (MDR) (9). LY294002 (LY) is a well‑studied PI3K inhibitor that effectively blocks autophagy (9). 5‑Fluorouracil (5‑FU), a fluorinated pyrimidine analog, is widely used in squamous cell cancers; its cytotoxicity arises from incorporation into RNA and inhibition of thymidylate synthase (10–12). Both 5‑FU and LY suffer from poor aqueous solubility and rapid systemic clearance, limiting their therapeutic window (6,7).
Nanocarriers, particularly PEGylated liposomes, have emerged as clinically validated vehicles for drug delivery, offering enhanced solubility, prolonged circulation, and passive tumor accumulation via the enhanced permeation and retention (EPR) effect (13–18). By encapsulating both 5‑FU and LY in a single PEG‑liposome, we hypothesize that early release of LY will disrupt autophagic survival pathways, rendering tumor cells more susceptible to 5‑FU and producing a synergistic antitumor response.
Methods
Materials
5‑FU (Sigma‑Aldrich, China) and LY294002 (Beijing Huafeng United Technology Corporation, China) were used as received. Lipids—egg phosphatidylcholine (EPC), DSPE‑PEG2000, and cholesterol—were sourced from Avanti Polar Lipids, China.
Preparation of Dual‑Drug‑Loaded Nanoliposomes
Phospholipids and cholesterol were dissolved in a methanol–chloroform mixture (10:4:1 molar ratio, 10 mg total). 5‑FU and LY (1.5 mg each) were added, and the solvent was evaporated at 60 °C under reduced pressure to form a thin film. Hydration with phosphate‑buffered saline (PBS) for 1 h followed by extrusion through a 200 nm polycarbonate membrane yielded the FLNP. Unencapsulated drug was quantified by HPLC. Drug loading (DL) and encapsulation efficiency (EE) were calculated as follows: DL (%) = (Weight of encapsulated drug / Weight of liposome) × 100; EE (%) = (Weight of encapsulated drug / Initial drug weight) × 100.
Particle Size Analysis
Dynamic light scattering (DLS) using a Malvern Zetasizer Nano ZS measured particle size and polydispersity index (PDI) at 25 °C.
Morphology Analysis
Transmission electron microscopy (TEM, Hitachi H‑7600) and atomic force microscopy (AFM) characterized morphology.
In Vitro Release Study
Drug release from FLNP was evaluated in PBS (pH 7.4) at 37 °C using a dialysis membrane (MW ≈ 3,500). Samples were withdrawn at predetermined intervals and analyzed by HPLC (Hitachi L‑2200 autosampler, L‑2420 UV–Vis detector; Inertsil C18 column, 150 × 4.6 mm, 5 µm). Linear calibration ranged from 0.025–2 µg/mL (r² ≈ 0.9995).
Cell Culture
Human ESCC cell line EC 9706 was maintained in RPMI 1640 supplemented with 10 % FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin in a 5 % CO₂ humidified incubator.
Cellular Uptake Analysis
Fluorescent rhodamine B‑loaded FLNP were incubated with EC 9706 cells (10 µg/mL) for 2 h. Confocal laser scanning microscopy (CLSM, Nikon TE2000‑E) visualized intracellular distribution. Flow cytometry (BD FACSCALibur) quantified uptake at 1 h and 2 h time points.
Cytotoxicity Assay
MTT viability assays were performed on 1 × 10⁴ cells/well (96‑well plate). Cells were treated with free 5‑FU, LY, 5‑FU + LY, or FLNP for 24 h. Absorbance at 570 nm was measured using a Synergy HTX reader.
Apoptosis Assay
Annexin‑V/PI staining (BD Biosciences) followed by flow cytometry quantified early and late apoptosis after 24 h treatment (1 µg equivalent drug).
Hoechst 33342 Assay
Hoechst 33342 (1 µg/mL) staining of fixed cells visualized nuclear condensation under a fluorescence microscope.
Western Blotting
Protein extracts were separated by SDS‑PAGE and transferred to PVDF membranes. Blots were probed for cleaved caspase‑3, PARP, Bcl‑2, and p53 (AbClon). Detection used ECL and Kodak M35‑A processing.
In Vivo Tumor Growth Inhibition
Female athymic nude mice (6 weeks) received subcutaneous injections of 1 × 10⁶ OE‑19 cells (100 µL). When tumors reached ~80 mm³ (day 10), mice (n = 6 per group) received 5 mg/kg FLNP, free drug combinations, or controls via intraperitoneal injection thrice over 10 days. Tumor volume (V = ½ × length × width²) was measured with calipers for 20 days.
Statistical Analysis
Data were expressed as mean ± SD. Significance was assessed by t‑test or ANOVA (p < 0.05).
Results and Discussion
Co‑delivery of 5‑FU and LY within PEG‑liposomes achieved a synergistic antitumor effect that surpassed the efficacy of each agent alone or their free combination. This synergy is attributed to the sequential release of LY, which impairs autophagy and sensitizes cells to 5‑FU‑induced DNA damage.
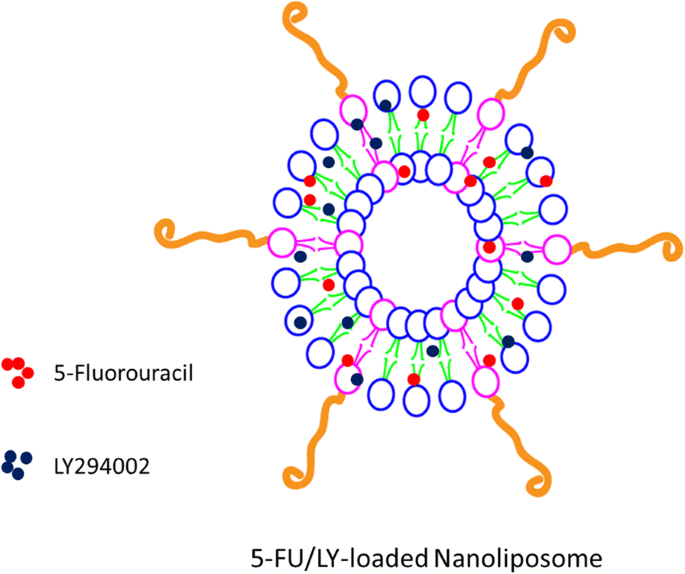
Schematic illustration of the preparation of 5‑fluorouracil and LY‑loaded PEGylated nanoliposomes.
Preparation and Characterization of 5‑FU and LY‑Loaded Nanoliposomes
Blank liposomes exhibited an average diameter of ~110 nm (PDI ≈ 0.15). Dual‑drug loading increased the size to ~150 nm (PDI ≈ 0.18), a dimension that favors EPR‑mediated tumor accumulation (17,18). The surface charge was +25 mV, conferring colloidal stability and minimizing nonspecific protein adsorption.

Physicochemical characterization of FLNP. a DLS size distribution; b TEM image; c AFM image.
Drug Loading and In Vitro Release
Encapsulation efficiencies were 92.5 % for 5‑FU and 86 % for LY, with a loading capacity of ~16 % w/w. Release profiles in PBS (pH 7.4) revealed a biphasic pattern: ~30 % 5‑FU and ~40 % LY released within 24 h, followed by a gradual release to ~60 % 5‑FU and ~75 % LY at 90 h (Fig. 3). LY’s faster release promotes early autophagy inhibition, priming cells for subsequent 5‑FU cytotoxicity.
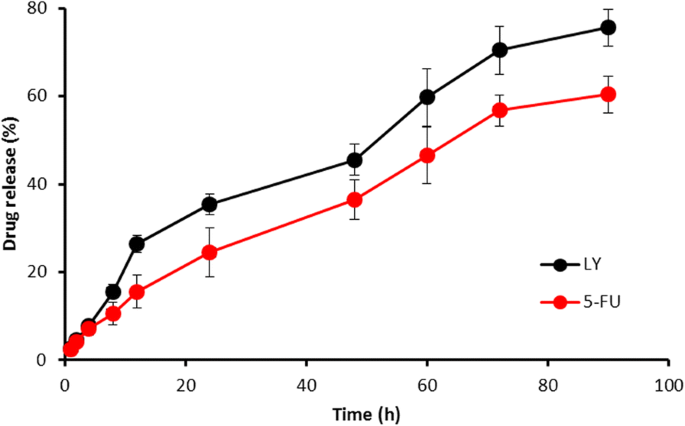
Release profiles of 5‑FU and LY from FLNP in PBS (pH 7.4). Drug amounts were quantified by HPLC.
Cellular Internalization Study
CLSM revealed intense red fluorescence in the perinuclear cytoplasm after 2 h, indicative of receptor‑mediated endocytosis. Flow cytometry confirmed time‑dependent uptake, with a marked increase between 1 h and 2 h (Fig. 4).
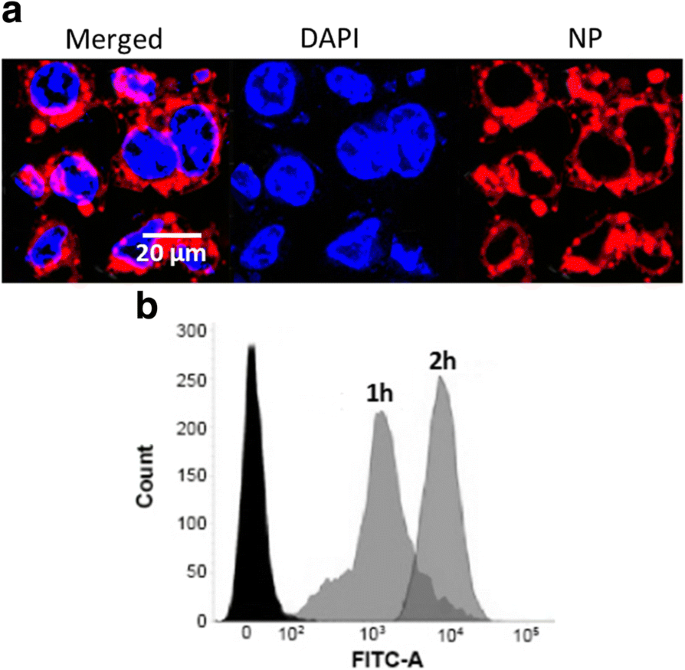
a CLSM image of EC 9706 cells after 2 h FLNP incubation. b Flow cytometry analysis at 1 h and 2 h.
In Vitro Anticancer Effect
MTT assays demonstrated dose‑dependent cytotoxicity for all formulations. FLNP achieved an IC₅₀ of 0.58 µg/mL, markedly lower than free 5‑FU (2.68 µg/mL), LY (5.98 µg/mL), and the free combination (1.54 µg/mL). Enhanced efficacy is attributed to improved drug delivery and sequential release (Fig. 5).
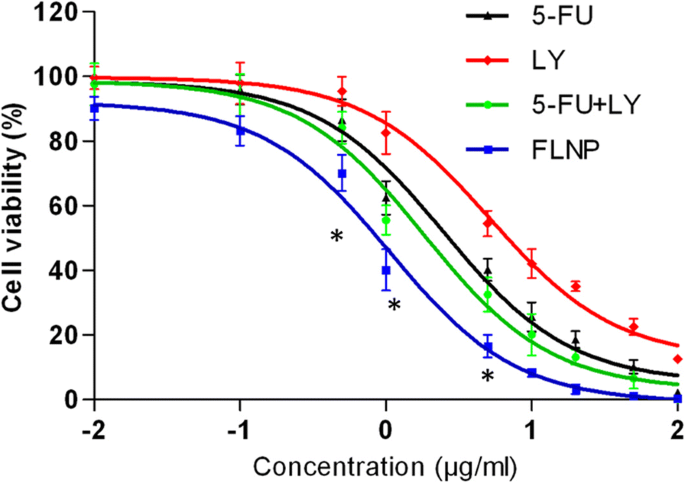
Cell viability of EC 9706 cells after 24 h exposure to free 5‑FU, LY, 5‑FU + LY, and FLNP (MTT assay).
Apoptosis Evaluation
Annexin‑V/PI staining revealed 48 % apoptosis in FLNP‑treated cells, compared to 20 % for 5‑FU alone and 30 % for the free combination (Fig. 6). Hoechst staining corroborated nuclear condensation in FLNP‑treated cells.
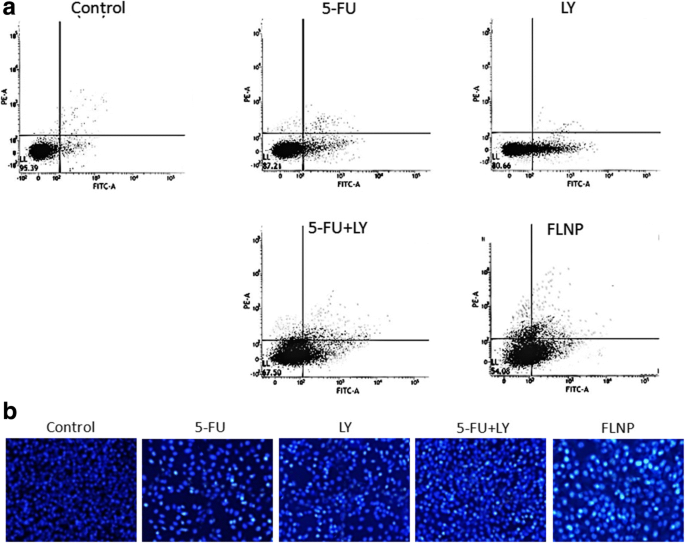
Flow cytometry of apoptosis in EC 9706 cells after 24 h FLNP treatment.
Western Blot Analysis
FLNP markedly increased cleaved caspase‑3 and PARP while reducing Bcl‑2 and p53 expression, confirming activation of the intrinsic apoptotic pathway (Fig. 7).
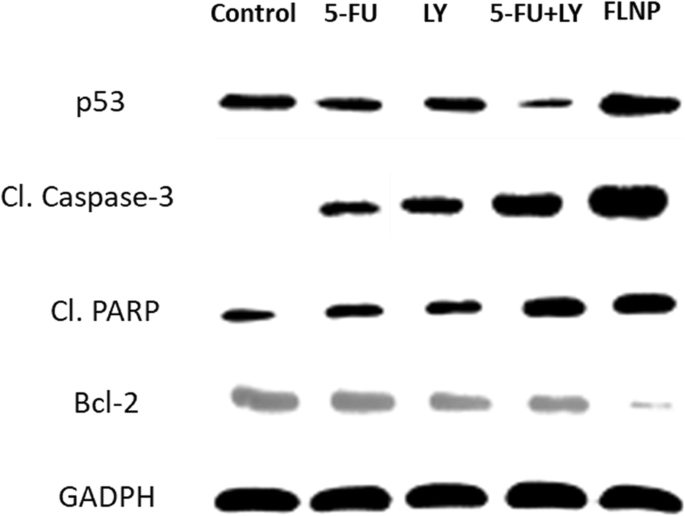
Western blot of apoptosis‑related proteins after FLNP treatment.
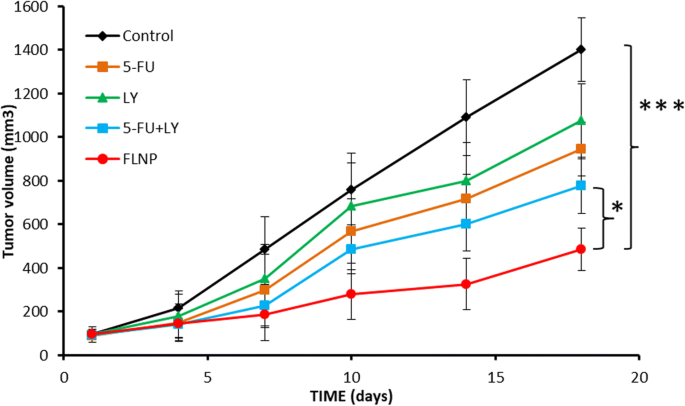
In vivo antitumor efficacy: FLNP reduced tumor volume to ~500 mm³ versus ~1,400 mm³ in controls (p < 0.0001).
In Vivo Tumor Growth Inhibition Analysis
FLNP consistently suppressed tumor progression over 20 days, achieving a >60 % reduction in final tumor volume relative to free drug groups. The enhanced therapeutic outcome is attributed to tumor‑selective accumulation (EPR) and sustained drug release.
Conclusions
Our PEG‑liposomal platform successfully co‑delivers 5‑FU and LY, achieving controlled, sequential release that amplifies cytotoxicity and apoptosis in ESCC. FLNP outperformed free drug combinations in vitro and in vivo, underscoring the potential of autophagy inhibition to overcome chemoresistance. Future studies will expand this strategy to other squamous cell malignancies and evaluate orthotopic and patient‑derived xenograft models.
Abbreviations
- 5‑FU:
5‑Fluorouracil
- EPR:
Enhanced permeation and retention effect
- ESCC:
Esophageal squamous cell carcinoma
- FLNP:
5‑FU and LY‑loaded lipid nanoparticles
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- gH625 Peptide vs. Folic Acid: Enhancing Fe3O4 Magnetic Nanoparticle Uptake Across the Blood–Brain Barrier
- Magnesium‑Doped Zinc Oxide Nanoparticles: Optimizing Photocatalytic Degradation and Antibacterial Efficacy
- Targeted Silica Nanoparticles Co‑Deliver miR‑495 and Doxorubicin to Overcome Multidrug Resistance in Lung Cancer
- Mesoporous Silica Nanoparticles Co-Deliver NQO1 Inhibitor and 5‑Fluorouracil, Enhancing Antitumor Activity in Head and Neck Squamous Cell Carcinoma
- Enhanced Antitumor Effect of Dacarbazine and All-Trans Retinoic Acid Co-Delivered via Lipid Nanoparticles for Malignant Melanoma
- Targeting miR‑301 Suppresses Angiogenesis and Tumor Growth in Esophageal Squamous Cell Carcinoma via PTEN Activation
- Chondroitin Sulfate-Enhanced Gelatin/Polycaprolactone Nanofibers: Antithrombogenic Scaffold with Superior Endothelial Affinity for Blood Vessel Tissue Engineering
- Quantitative Study of PLGA Nanoparticle Uptake in Laryngeal Cancer and Immune Cells to Improve Drug Delivery



