Rapid Capture of Escherichia coli at Ultralow Concentrations Using Positively Charged Magnetic Nanoparticles
Abstract
Rapid, culture‑free detection of bacteria is essential for timely outbreak control. Positively charged magnetic nanoparticles (NP+) exploit the natural negative surface charge of bacterial cells, enabling efficient capture of Escherichia coli at concentrations as low as 10 and 100 CFU/mL within one hour. Capture efficiency exceeded 90 % as determined by colony‑forming unit (CFU) counts. Transmission electron microscopy (TEM) and optical imaging confirmed the robust binding of NP+ to bacterial surfaces, whereas negatively charged particles (NP−) showed negligible affinity. These results demonstrate that electrostatic attraction can provide a ligand‑independent, broad‑spectrum bacterial capture platform.
Background
Infectious diseases remain a top global health priority, and waterborne bacterial contamination poses a significant risk. E. coli, a common gram‑negative pathogen, often appears at low levels in contaminated water and food, making detection challenging. Traditional culture‑based methods are time‑consuming, can miss viable‑but‑non‑culturable (VBNC) cells, and delay critical interventions. Rapid, magnetic‑based separation techniques offer quick, scalable, and automatable solutions for bacterial removal and detection. However, most magnetic systems rely on specific ligands, which can limit capture breadth and require extensive optimization.
The intrinsic negative surface charge of bacterial cells, first documented by Bechhold in 1904, underpins electrostatic interactions with positively charged nanomaterials. Recent work (Cohen et al., 2011) has shown that individual E. coli cells exhibit electrical spiking, reinforcing the role of membrane potential in cell‑surface interactions. Leveraging this property, we engineered polyethylenimine (PEI)–functionalized magnetic nanoparticles to capture bacteria across a wide concentration range without ligand dependency.
Materials and Methods
Nanomaterials
Reagents were sourced from Shanghai Reagent Company and Sigma‑Aldrich (USA). All solutions were prepared in Milli‑Q deionized water (18.2 MΩ cm). Branched PEI (Mw = 10,000, 99 %) was purchased from Alfa Aesar.
NP Syntheses
Fe3O4 cores were synthesized by solvothermal reaction at 200 °C for 12 h. A silica shell was formed via TEOS hydrolysis, followed by grafting of an APTES–TRITC complex to impart fluorescence. Negatively charged nanoparticles (NP−) were generated by retaining the APTES surface; positively charged particles (NP+) were produced by coating NP− with PEI, yielding abundant surface amines.
NP Characterization
TEM (300 kV) confirmed core–shell morphology and ~450 nm diameter. Dynamic light scattering (DLS) measured hydrodynamic diameters of 620 nm (NP+) and 700 nm (NP−). Zeta potential analyses revealed −26.6 mV for NP− and +28.1 mV for NP+ in neutral buffer, indicating stable dispersion suitable for bacterial capture.
Bacteria Preparation
Strain BL21 of E. coli was cultured in LB medium at 37 °C, 200 rpm for 16 h. Following serial dilution and plating, CFU counts were obtained. Cells were harvested by centrifugation, washed in PBS, and resuspended at ~1 × 103 CFU/mL. All handling complied with biosafety level 2 protocols.
Bacteria Capture Experiment
In a 1 mL PBS suspension, 40 µg of NP+ or NP− was mixed with bacteria and incubated for 10 min at room temperature. A permanent magnet attracted NP‑bound cells to the tube wall, after which unbound cells were washed away. Captured bacteria were released by removing the magnet and resuspended for subsequent analyses.
Bacterial Capture Efficiency of NPs at Different Concentrations
Capture efficiency was quantified by plating the total solution and counting CFUs. Experiments ranged from 5 to 100 µg/mL of NP+ or NP− with a bacterial load of 2 × 102 CFU/mL.
Capability of NPs to Capture Bacteria at Low Concentrations
To assess ultralow‑level performance, 40 µg of NP+ or NP− was incubated with 1 mL of PBS containing 10 or 100 CFU/mL of E. coli. Capture efficiencies were again determined by CFU enumeration.
Statistical Analysis
Data are expressed as mean ± SD. Two‑way ANOVA with post‑hoc testing (GraphPad Prism) determined significance at p < 0.05.
Results
Characterization of Magnetic NPs
The schematic in Fig. 1 illustrates the stepwise fabrication of NP− and NP+. TEM images confirm uniform SiO2 coating (~60 nm) around a ~450 nm Fe3O4 core. DLS shows narrow size distributions; zeta potential data confirm successful surface functionalization (Fig. 2).
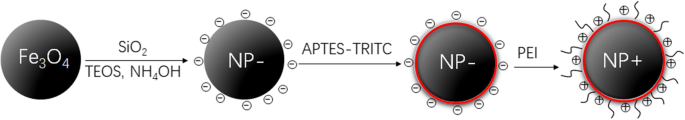
Design of the nanoparticles. Schematic diagram showing the design of surface‑charged, fluorescent, superparamagnetic composite nanoparticles (NPs).
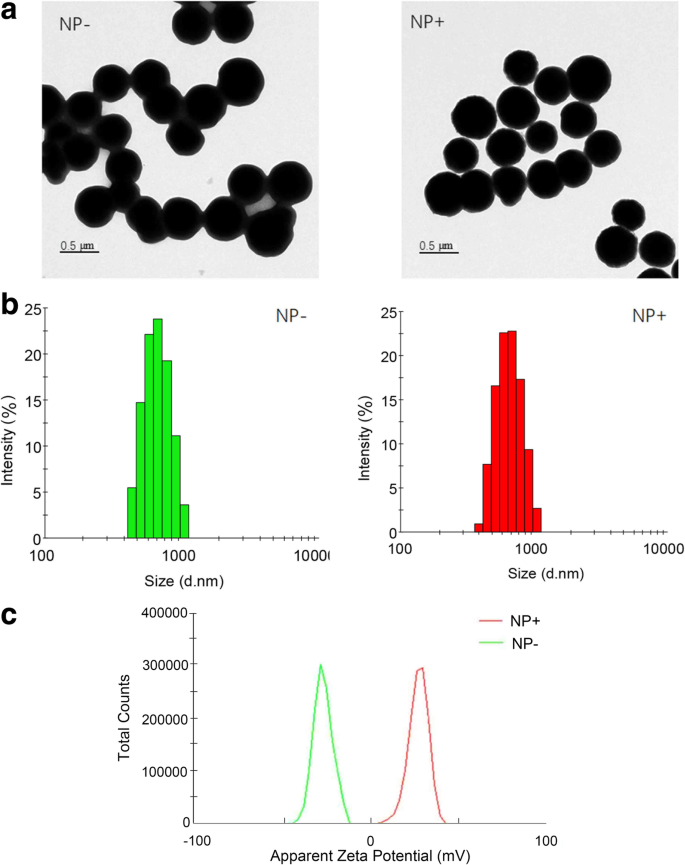
Characterization of the nanoparticles. a TEM image of NP+ and NP−. b DLS size distribution. c Zeta potential distributions.
Ability of Magnetic NPs to Capture E. coli
Figure 3 outlines the capture workflow: NP–bacteria incubation, magnetic attraction, washing, and release. NP+ rapidly enriched E. coli, whereas NP− showed negligible binding (Fig. 3).
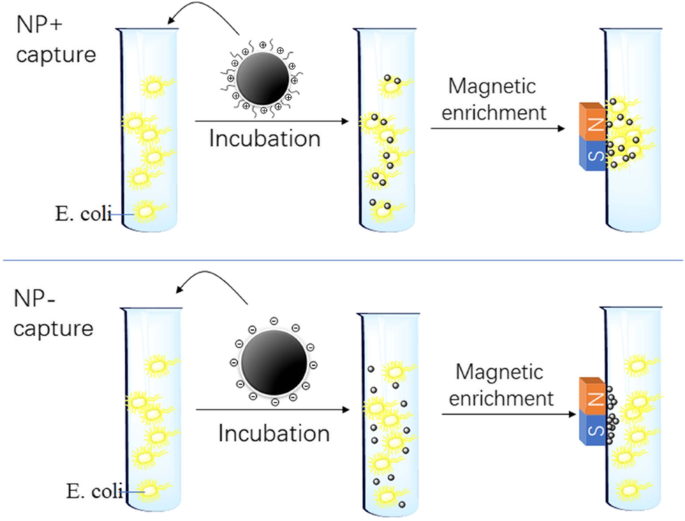
Illustration of the procedures of bacteria capture. E. coli in suspension are respectively mixed with NP+ and NP−, followed by 10 min incubation. The “magnetized” bacteria are attracted onto the vial wall by a magnet, washed, and counted.
Optical imaging (Fig. 4) shows NP− as monodisperse, whereas NP+ aggregates, reflecting distinct interaction modes. Fluorescence microscopy confirms co‑localization of TRITC‑labeled NP+ with DAPI‑stained E. coli (Fig. 5a). TEM reveals extensive NP+ clustering on bacterial cell walls (Fig. 5b), underscoring strong electrostatic affinity.
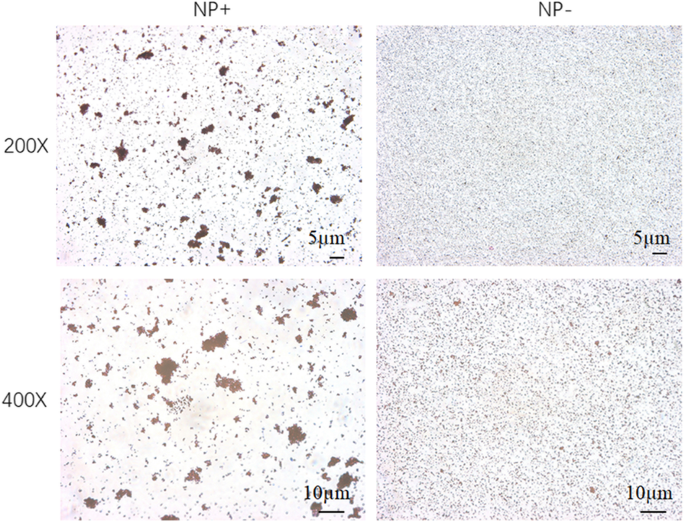
Comparison of E. coli binding capacity of NP+ and NP−. Left: phase‑contrast of bacteria bound to NP+. Right: NP‑only control.
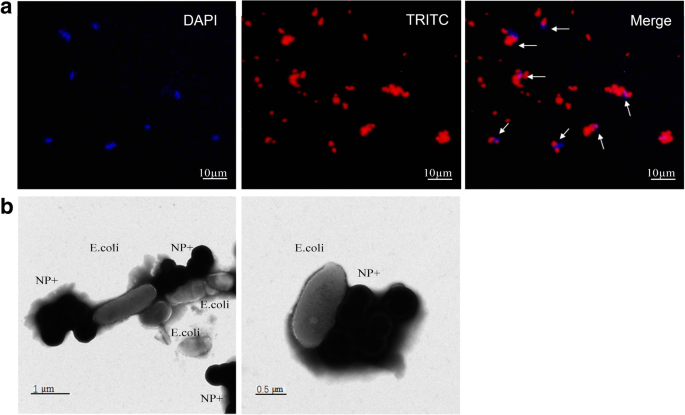
Fluorescent and TEM images of E. coli binding with NP+. a Fluorescence: DAPI (blue) + TRITC (red). b TEM: NP+ aggregates on bacterial surface.
Detection of Low Concentrations of E. coli
Increasing NP+ dosage from 5 to 100 µg/mL yielded a dose‑dependent capture efficiency (Fig. 6). At 40 µg/mL, NP+ achieved 81 % efficiency (p < 0.001), whereas NP− captured only 12 % even at 100 µg/mL.
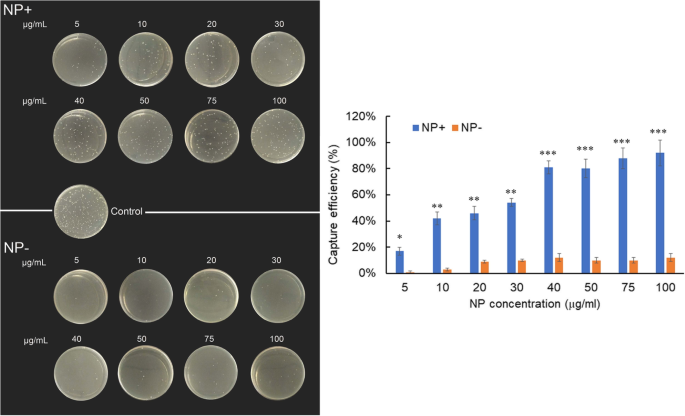
Capture efficiencies of E. coli by NP+ or NP− at various concentrations indicated. Left: LB‑agar plates. Right: efficiency plot.
At ultralow loads (10 and 100 CFU/mL), NP+ consistently recovered >90 % of cells (Fig. 7), whereas NP− recovered <4 %. These results validate the superior electrostatic capture capability of NP+.
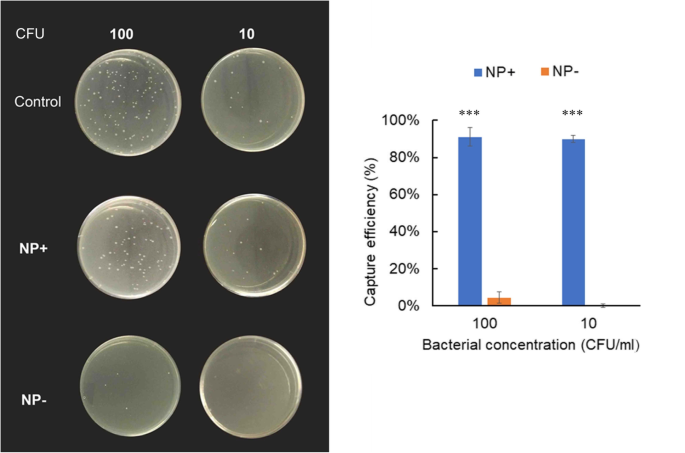
Capture efficiencies of E. coli by NP+ or NP− at ultralow concentrations. Left: agar plates. Right: efficiency plot.
Discussion
Gram‑negative and gram‑positive bacteria display strong electrostatic attraction to positively charged surfaces. The PEI‑functionalized NP+ offers abundant amine groups, yielding a +28.1 mV surface charge that promotes rapid binding even at <102 CFU/mL. This study demonstrates a simple, one‑hour protocol that captures E. coli at 10 CFU/mL—well below the detection limits of conventional enrichment methods.
Charge‑dependent capture efficiencies were evident: NP+ achieved up to 81 % efficiency at 40 µg/mL, while NP− remained ineffective. TEM images confirmed large NP+ aggregates on bacterial surfaces, suggesting that size and surface charge synergistically enhance capture.
While gram‑positive and gram‑negative cells differ structurally, both maintain a net negative surface charge at physiological pH due to lipopolysaccharides or teichoic acids. The larger NP+ (>600 nm) provide a “sponge” effect—greater surface area facilitates more extensive electrostatic interactions, whereas smaller particles might be limited by reduced surface coverage. Thus, our design prioritizes capture efficiency over antimicrobial activity.
Conclusions
PEI‑coated magnetic nanoparticles offer a rapid, ligand‑independent method to capture and analyze E. coli at ultralow concentrations. The approach yields high recovery (>90 %) within one hour, and the captured bacteria can be readily visualized by optical and TEM imaging. This platform is adaptable to a broad range of bacterial pathogens, enabling swift diagnostics and decontamination in clinical and environmental settings.
Nanomaterials
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Percolation-Induced Ferromagnetism in Potassium Tantalate and Niobate Nanoparticles
- Optimizing Heavy Metal Removal with Iron‑Modified Magnetic Biochar Nanocomposites
- Impact of Magneto‑Dipole Interactions on the Specific Absorption Rate of Iron Oxide Nanoparticle Assemblies
- How Synthesis Route Shapes the Structure, Magnetism, and Hyperthermia Performance of La₁₋ₓSrₓMnO₃ Nanoparticles
- Rapid One‑Pot Synthesis of Monodisperse CoFe₂O₄@Ag Core–Shell Nanoparticles with Exceptional Plasmonic and Magnetic Properties
- Optimizing Magnetofection of MG‑63 Osteoblasts with Uniform Magnetic Field and PEI‑SPIO Nanoparticles
- Bibliometric Trends in Drug Delivery and Magnetic Nanoparticles (1980‑2017)
- Optimizing Heat Generation in Triple‑Vortex Cylindrical Magnetic Nanoparticles for Cancer Hyperthermia
- Boosting Performance of All‑Inkjet‑Printed ZnO UV Photodetectors via Ag Nanoparticle Modification



