Eco‑Friendly Green‑Emitting Phosphor: Eu2+‑Doped Zeolite‑3A for High‑Purity Green LEDs
Abstract
Eu2+-activated phosphors are the cornerstone of modern lighting and display technologies, owing to their bright, stable, and eco‑friendly emission. Here we report a highly efficient green‑emitting phosphor based on zeolite‑3A doped with 1.3 wt % Eu. The material is synthesized by a high‑temperature solid‑state reaction that requires no reducing atmosphere or agents, making the process safe, cost‑effective, and environmentally benign. We systematically investigate the influence of sintering temperature on morphology, crystal structure, and luminescence, revealing that Eu3+ ions are progressively reduced to Eu2+ as the temperature rises. The optimized sample displays a broad excitation band from 310 to 450 nm and a prominent emission peak at 523 nm, with a photoluminescence quantum yield of 36.6 %. When incorporated onto a commercial UV‑emitting chip, the phosphor yields a pure green LED (CIE 1931 coordinates (0.295, 0.537)) with a brightness of 231.6 cd m–2 under 3 V. These results demonstrate a simple, green synthesis route and a promising green phosphor for next‑generation lighting and display applications.
Background
Luminescent materials underpin a wide array of devices from general lighting to high‑resolution displays. Rare‑earth (RE) europium‑activated phosphors have attracted sustained attention because they combine high brightness, chemical stability, and environmental friendliness. Europium exists in two oxidation states: Eu3+ typically emits in the red via 5d–4f transitions, while Eu2+ exhibits broad 4f65d→4f7 emission that spans the visible spectrum and can be tuned by the crystal field. Because Eu2+ is not found naturally, it must be generated by reducing Eu3+, traditionally in reducing atmospheres such as H2/N2 or CO, which raise safety concerns and cost.
Recent work has shown that Eu3+ can be reduced to Eu2+ in air at high temperature when hosted in rigid frameworks containing BO4, PO4, or AlO4/AlO6 groups. Zeolites, with their aluminosilicate framework and natural abundance, satisfy these requirements and provide a chemically robust host. Zeolite‑3A (Si/Al ≈ 1) has been widely used for down‑conversion phosphors. In this study we demonstrate that Eu2+-doped zeolite‑3A can be prepared via a simple high‑temperature treatment in air, yielding a bright green phosphor suitable for LED applications.
Methods
Aim of the Study
To develop a green, safe, and cost‑effective synthesis route for Eu2+-activated phosphors that deliver bright green emission for LED applications, without using a reducing atmosphere.
Materials
Zeolite‑3A (2/3 K2O·1/3 Na2O·Al2O3·2 SiO2·9/2 H2O, Si/Al ≈ 1) was purchased from Shanghai Tongxing Molecular Sieve Co., Ltd. Europium(III) oxide (Eu2O3) was obtained from Sinopharm Co., Ltd. Silicone resin and an InGaN blue chip (5 mm × 5 mm, λ = 375 nm) were supplied by Shenzhen Looking Long Technology Co., Ltd.
Synthesis of Samples
Eu2+-doped zeolite‑3A was prepared by a high‑temperature solid‑state reaction. Stoichiometric amounts of zeolite‑3A and Eu2O3 were mixed and ground in an agate mortar for 40 min, then sintered at various temperatures (600–1400 °C) in air for 3 h. After cooling, the resulting powders were collected.
Fabrication of Green LEDs
The phosphor powder was blended with silicone resin (mass ratio 1:5) and stirred until homogeneous. The mixture was coated onto an InGaN chip and cured at 60 °C for ~2 h, producing a ~1 mm thick composite layer.
Characterization
Morphology and crystal structure were examined by FESEM (FEI Sirion‑200) and XRD (Philips X’Pert, Cu Kα, λ = 0.15405 nm). Thermogravimetric analysis (TGA) was performed on an SDT Q600 from room temperature to 800 °C (10 °C min–1) in N2. Photoluminescence (PL) and photoluminescence excitation (PLE) spectra were recorded at room temperature using an Edinburgh Instruments FLS920 spectrometer with a 450‑W Xe lamp. Eu oxidation states were probed by XPS (ESCALAB 250). Electroluminescence (EL) spectra of the LEDs were obtained with an Ocean Optics FLAME‑S‑VIS‑NIR spectrometer and a fiber‑integrating sphere.
Results and Discussion
Figure 1a shows a pristine zeolite‑3A SEM image: irregular cubic particles ~1.5 µm in size. After sintering at 1400 °C for 3 h, the phosphor (Figure 1b) displays aggregated, irregular particles; XRD (Figure 1c) confirms retention of the pure zeolite‑3A phase with no secondary peaks, indicating successful Eu2+ incorporation without disturbing the lattice. TGA (Figure 1d) shows negligible mass loss for the doped sample, confirming thermal stability.
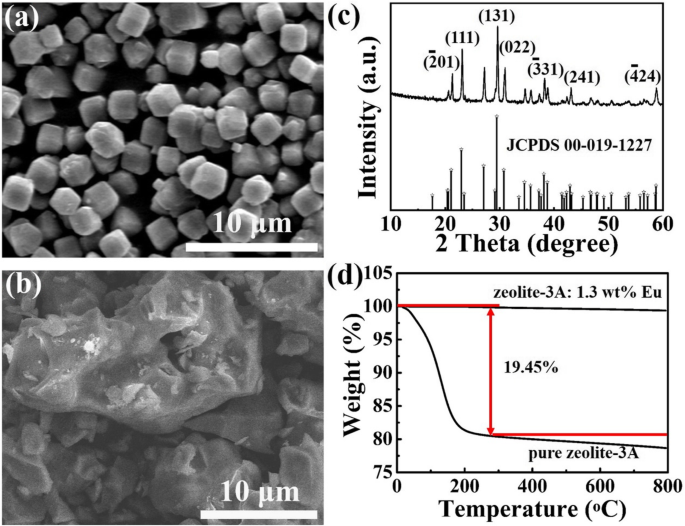
The SEM images of a pure zeolite‑3A and b zeolite‑3A: 1.3 wt % Eu sintered at 1400 °C for 3 h. c XRD pattern of the phosphor. d TG curves of pure zeolite‑3A and the phosphor.
Figure 2 explores temperature effects. SEM images (a–d) show that below 800 °C the morphology remains cubic, but at 1000 °C and above particles aggregate into larger clusters. XRD (Figure 2e) reveals that samples sintered below 800 °C contain Eu2O3 impurities (extra peaks at 12.5° and 16.3°), indicating incomplete Eu incorporation. Above 1000 °C, only the zeolite‑3A pattern appears, confirming successful Eu2+ doping.
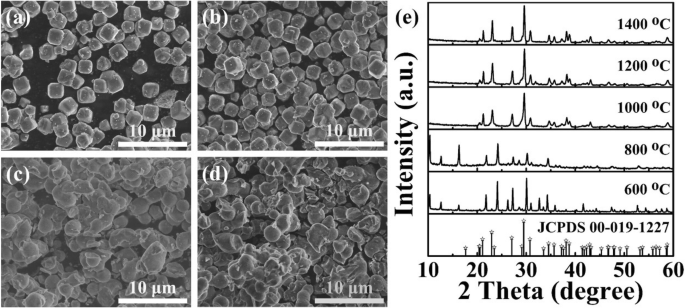
SEM images of zeolite‑3A: 1.3 wt % Eu sintered at 600 °C (a), 800 °C (b), 1000 °C (c), and 1200 °C (d). e XRD patterns at the corresponding temperatures.
PL spectra (Figure 3a) illustrate the evolution from red Eu3+ emission (617 nm) at low temperatures to dominant green Eu2+ emission (523 nm) at 1400 °C. The broad excitation band (310–450 nm) and the sharp green emission peak at 523 nm are captured in the PL/PLE spectra (Figure 3b), matching the UV chip’s 375 nm output. XPS (Figure 4) shows characteristic Eu3+ peaks (1165 eV, 1135 eV) and Eu2+ peaks (1155 eV, 1125 eV), confirming partial reduction during high‑temperature sintering. The proposed mechanism involves Eu3+ substitution for K+ in the framework, vacancy creation, and electron capture leading to Eu2+ formation.
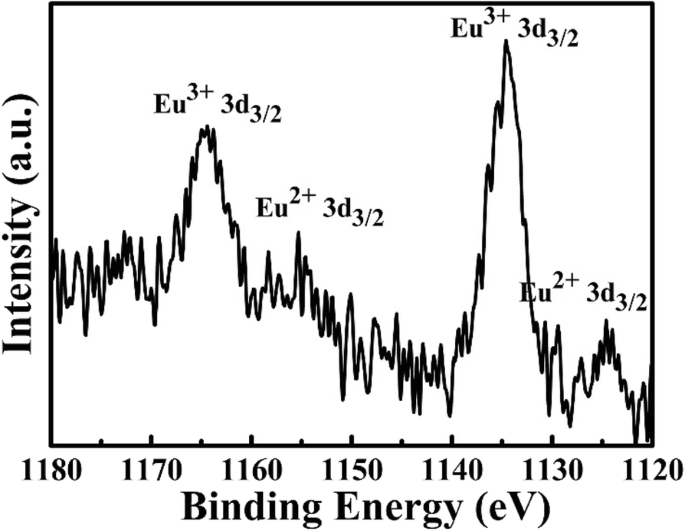
XPS spectrum for Eu in the phosphor sintered at 1400 °C.
Optimizing Eu concentration (Figure 5) reveals a maximum PL intensity at 1.3 wt % Eu; higher doping leads to concentration quenching due to increased Eu–Eu energy transfer. The relative PL intensity curve (Figure 5b) demonstrates reproducibility.
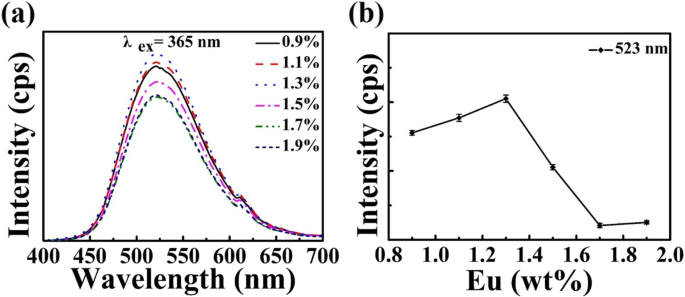
a PL spectra for varying Eu content (x = 0.9–1.9 wt %) sintered at 1400 °C. b Relative PL intensity versus Eu concentration.
The fabricated green LED (Figure 6a) shows EL peaks at 375 nm (chip) and 523 nm (phosphor). The device emits bright green light at 3 V, with CIE coordinates (0.295, 0.537) and a brightness of 231.6 cd m–2, indicating high color purity.
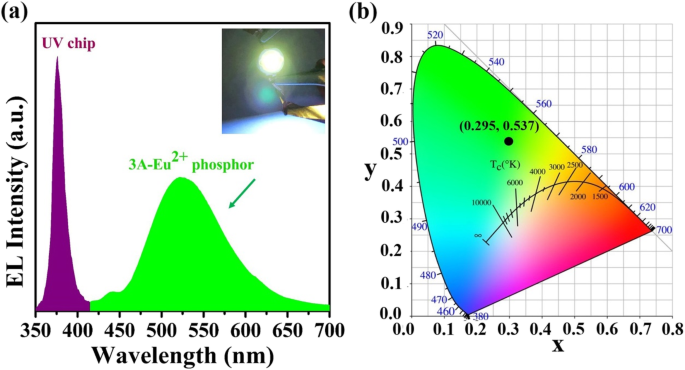
a EL spectrum at 3 V. Inset: photograph of the working green LED. b CIE color coordinates.
Conclusions
We have demonstrated a green, air‑based high‑temperature synthesis of a Eu2+-activated zeolite‑3A phosphor with a 36.6 % quantum yield and a 523 nm emission peak. The phosphor’s broad excitation (310–450 nm) matches commercial UV chips, enabling the construction of a high‑purity green LED (CIE (0.295, 0.537), 231.6 cd m–2). The process requires no reducing gases, making it safe, scalable, and environmentally friendly. These findings position Eu-doped zeolite‑3A as a promising material for future lighting and display technologies.
Availability of Data and Materials
The datasets supporting the conclusions of this article are available in the article.
Abbreviations
- CIE:
Commission Internationale de L’Eclairage
- EL:
Electroluminescence
- Eu:
Europium
- FESEM:
Field emission scanning electron microscope
- LED:
Light-emitting diode
- PL:
Photoluminescence
- PLE:
Photoluminescence excitation
- RE:
Rare-earth
- TG:
Thermogravimetric analysis
- XPS:
X-ray photoelectron spectroscopy
- XRD:
X-ray diffraction
Nanomaterials
- Mobile Maintenance Software: Driving Efficient Workflows
- Green One‑Pot Synthesis of Ag‑Decorated SnO₂ Microspheres: A Highly Reusable Catalyst for 4‑Nitrophenol Reduction
- Eco-Friendly Synthesis of InP/ZnS Core/Shell Quantum Dots for High-Performance Heavy-Metal-Free LEDs
- Palladium(II)-Imprinted Polymeric Nanospheres for Efficient Removal of Pd(II) from Water
- Green Synthesis of Gold and Silver Nanoparticles with Platycodon Saponins from Platycodi Radix
- High‑Efficiency Trilayer Phosphorescent OLEDs Without Electrode Modification Layers: Design, Mechanism, and Performance
- Enhanced Red Upconversion in Single β‑NaYF4:Yb/Er Microcrystals Doped with Mn²⁺: Tunable Multicolor Emission and Energy‑Transfer Dynamics
- Simple Fabrication and Performance of Polyaniline/CeO₂‑Co‑Decorated TiO₂ Nanotube Arrays for Efficient Photoelectrocatalytic Degradation of TBBPA
- Phase Engineering Boosts Efficiency of Quasi‑2D All‑Inorganic Perovskite LEDs via Cs Cation Ratio Control
- Enhancing Deep Ultraviolet LED Performance via Locally Modulated NPN‑AlGaN Current‑Spreading Layers



