Helium: Properties, Production, and Global Supply
Background
Helium is the second lightest element in the periodic table and one of the most chemically inert gases. Its low density and non‑flammability make it indispensable for aerospace applications, from lifting blimps and balloons to providing a safe atmosphere for welding and crystal growth in semiconductor manufacturing. Liquid helium is also critical in research that requires temperatures close to absolute zero, such as superconductivity experiments.
Although the element is abundant in the universe, Earth’s atmosphere contains only trace amounts. Helium was first identified in 1868 when astronomers Pierre Janssen and Sir Joseph Lockyer observed an unidentified yellow spectral line during a solar eclipse. Lockyer coined the name “helium” from the Greek helios (sun).
In 1895, Sir William Ramsay extracted helium from the uranium mineral cleveite, confirming its terrestrial existence. A few years later, natural gas from a well near Dexter, Kansas, revealed concentrations as high as 2 %. Worldwide surveys showed varying helium levels, with the United States generally hosting the richest deposits.
Early 20th‑century lighter‑than‑air craft relied almost exclusively on hydrogen, despite its flammability. The U.S. recognized the strategic advantage of helium during World War I, designating it a critical war material and restricting exports. The 1925 Helium Conservation Act prohibited non‑government use. After the Hindenburg disaster in 1937, regulations eased, and helium replaced hydrogen for commercial airships.
During World II, helium again proved vital; it was used to inflate the tires of long‑range bombers, reducing aircraft weight and extending range. Post‑war demand spiked, prompting the 1960 Helium Act Amendments to establish a national storage program. By 1971, production had plateaued and the program was temporarily suspended, only to resume later. In 1993, government storage reached roughly 35 billion cubic feet (1 billion cubic meters).
Today, the bulk of helium‑bearing natural‑gas fields are located in the United States, with notable reserves also in Canada and Poland.
Raw Materials
Helium originates underground when alpha particles—products of radioactive decay of heavy elements like uranium and thorium—form helium nuclei. Some helium diffuses to the surface and escapes into space, while the remainder becomes trapped beneath impermeable rock layers and mixes with natural gas reservoirs. Helium concentrations vary from negligible to as high as 4 % by volume, but only deposits exceeding 0.4 % are commercially viable.
Helium can also be extracted by cryogenic air separation, though the process is cost‑intensive and yields very low recoveries, making it impractical for large‑scale production.
The Manufacturing Process
Helium is primarily recovered as a by‑product of natural‑gas processing. The gas contains methane and other hydrocarbons, which provide the energy content of natural gas. To meet market energy specifications, impurities—including nitrogen, water vapor, carbon dioxide, and helium—must be removed in an upgrading step.
When a gas stream contains more than ~0.4 % helium, cryogenic distillation is employed to isolate the helium. The resulting crude helium is further refined to achieve commercial purities above 99.99 %.
Pretreating
Because the downstream cryogenic column operates at extremely low temperatures, all components that could solidify—water vapor, CO₂, heavy hydrocarbons—must be removed beforehand to prevent pipe blockage.
- 1. The natural gas is pressurized to ~800 psi (5.5 MPa) and routed through a monoethanolamine scrubber to absorb CO₂.
- 2. A molecular‑sieve unit separates water vapor, allowing only smaller molecules to pass; the water is then back‑flushed and removed.
- 3. Activated‑carbon beds capture heavy hydrocarbons; the gas stream is now largely methane, nitrogen, and trace helium, hydrogen, and neon.
Separating
Fractional distillation, often called nitrogen rejection, isolates the primary components—methane and nitrogen—before the helium‑rich stream is sent to cryogenic processing. The procedure is illustrated in the diagram below.
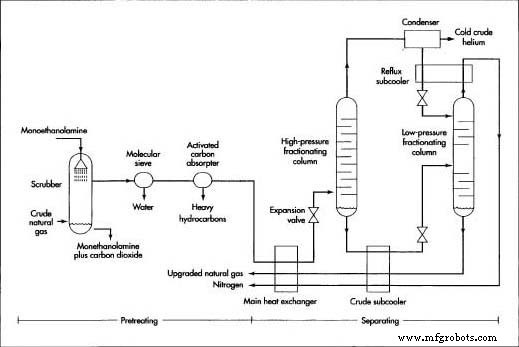
- 4. A plate‑fin heat exchanger cools the incoming stream while a counter‑flow of very cold methane and nitrogen warms the gases.
- 5. The gas expands through a valve, dropping pressure to ~145–360 psi (1.0–2.5 MPa). Rapid expansion cools the mixture and begins methane liquefaction.
- 6. The partially liquid gas enters the high‑pressure column; as it rises, methane continues to liquefy at the bottom while nitrogen rises to the top.
- 7. The liquid methane (crude methane) is extracted, subcooled, and passed through a second expansion valve to ~22 psi before entering the low‑pressure column. Here, most nitrogen is removed, yielding a methane‑rich liquid with ≤4 % nitrogen. The liquid is warmed, evaporated, and upgraded to natural gas, while nitrogen is vented or recycled.
- 8. Gases from the high‑pressure column are cooled in a condenser; nitrogen condenses as vapor, and the remaining gas—crude helium—contains 50–70 % helium, 1–3 % unliquefied methane, small amounts of hydrogen and neon, and the balance nitrogen.
Purifying
Crude helium undergoes a multi‑stage purification to reach commercial purity levels.
- 9. Cooling to –315 °F (–193 °C) condenses nitrogen and methane, leaving ~90 % helium.
- 10. Adding air introduces oxygen; the gas is preheated, then passed over a catalyst that reacts hydrogen with oxygen to form water vapor, which is condensed and removed.
- 11. A pressure‑swing adsorption (PSA) unit captures remaining contaminants. Thousands of micron‑pore particles adsorb nitrogen, methane, and residual water. Subsequent depressurization releases the adsorbed gases, leaving ~99.99 % pure helium.
Distributing
Helium is shipped either as a gas at ambient temperature or as a liquid at cryogenic temperatures. Gaseous helium is stored in forged steel or aluminum alloy cylinders at 900–6,000 psi (6–41 MPa). Bulk liquid helium is transported in insulated containers with capacities up to ~14,800 gallons (56,000 L).
- 12. For liquid or ultra‑pure helium, a cryogenic adsorber removes residual neon and trace impurities at –423 °F (–253 °C), achieving ≥99.999 % purity.
- 13. The gas then passes through heat exchangers and expanders in a liquefier, reaching –452 °F (–269 °C) to liquefy.
- 14. Large shipments of liquid helium use double‑shell, vacuum‑insulated trailers or containers. The inner shell contains helium; the outer shell provides insulation, and in overseas shipments a secondary shell with liquid nitrogen absorbs heat, evaporating as it warms.
Quality Control
The Compressed Gas Association grades helium by impurity content. Commercial grades begin with M (99.995 % pure) and progress to N, P, and G (99.9999 % pure). Regular sampling and analysis verify compliance with these standards.
The Future
In 1996, the U.S. government proposed terminating the state‑funded helium storage program, sparking concern among scientists. Helium is essentially a by‑product of natural‑gas processing; without storage, much of it would be vented into the atmosphere and lost. Some experts warned that global reserves could be depleted by 2015 if the program ceased.
Manufacturing process
- Gas Lanterns: Evolution, Design, and Modern Manufacturing
- Natural Gas: From Extraction to Distribution – A Comprehensive Overview
- The Evolution and Modern Technology of Gasoline Pumps
- Industrial Full‑Face Gas Mask Production & Quality Standards
- Advanced Arduino-Based Hazardous Gas Leak Detection System
- Optimizing Blast Furnace Gas Cleaning for Energy Efficiency
- ULCORED: Advanced Direct Reduction Iron Production with CO₂ Capture
- Gas Valves Explained: Types, Functions, and Safety Tips
- Gas Welding Explained: Techniques, Applications, Pros & Cons
- Biogas Explained: Renewable Energy from Organic Waste



