Mercury: Uses, Production, and Environmental Impact
Background
Mercury (Hg) is a dense, silvery liquid metal that has been used since antiquity for its unique properties. Historically valued for alloying with gold and silver, its role expanded during the 19th‑century California Gold Rush, when mercury ore deposits were extensively mined. Today, mercury’s applications are largely confined to chlorine and caustic soda production, as well as select electrical devices such as fluorescent lamps and mercury‑vapor lamps.
Archaeological evidence shows mercury was present in Egyptian tombs around 1500 B.C., likely for cosmetic or medicinal use. The Greek philosopher Aristotle described cinnabar processing in 350 B.C., and the Romans coined the term “hydrargyrum” (liquid silver), the origin of the chemical symbol Hg.
In the 16th century, mercury was employed to extract silver from ore, and in 1643 Torricelli introduced the mercury barometer. Subsequent inventions included the mercury thermometer (Fahrenheit, 1714) and, in 1828, the first dental amalgam fillings—though concerns over toxicity limited adoption until 1895 when G. V. Black demonstrated safety.
Throughout the 20th century, mercury appeared in batteries, paints, explosives, light bulbs, pharmaceuticals, fungicides, pesticides, and various manufacturing processes (paper, felt, glass, plastics). By the 1980s, growing awareness of mercury’s health and environmental hazards prompted a steep decline in use. In the United States, battery use fell to <5 % of 1988 levels by 1992, and electrical device usage dropped by 50 % during the same period. Paints, fungicides, and pesticides have been banned nationwide, while mercury use in paper, felt, and glass production has been voluntarily discontinued.
Global production is now limited to a handful of countries with less stringent environmental regulations. Spain ceased mining in 1989; the U.S. has also stopped primary mining, though small amounts are recovered during gold refining to mitigate contamination. In 1992, China, Russia (formerly USSR), Mexico, and Algeria were the leading producers.
Raw Materials
Mercury is rarely found in its elemental form; it is almost always extracted from ores. The most common ore is cinnabar (HgS), but other minerals include corderoite (Hg₃S₂Cl₂), livingstonite (HgSb₄S₈), montroydite (HgO), and calomel (HgCl). These ores form underground when mineral‑laden solutions ascend toward the surface, often in faulted and fractured rock at depths between 1 m and 1000 m.
Historical mining sites and tailings from older, less efficient operations also contribute secondary sources of mercury.
The Manufacturing Process
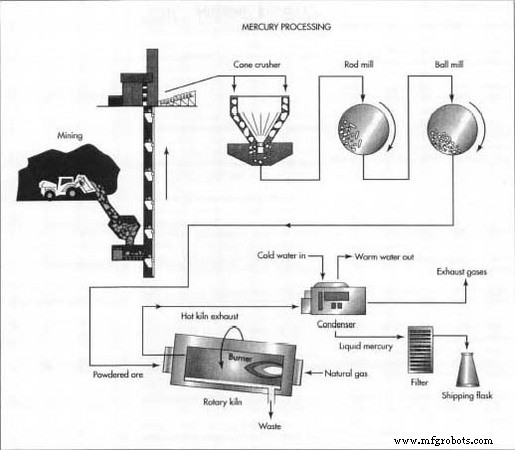
Extraction has remained essentially unchanged since Aristotle’s time. Cinnabar ore is crushed, heated, and the released mercury vapor is cooled and condensed. Nearly 95 % of the mercury can be recovered using this method.
Mining
Cinnabar deposits are typically near the surface; about 90 % require underground mining via tunnels, while 10 % can be extracted by open‑pit methods.
- 1. Ore is dislodged through drilling and blasting or by power equipment, then transported via conveyor belts, trucks, or trains.
Roasting
Due to high concentration, cinnabar can be processed directly. The ore undergoes crushing and grinding to a fine powder, then heated in a furnace or rotary kiln. Combustion of natural gas or other fuels supplies heat, and the ore reacts with atmospheric oxygen to release sulfur dioxide, while mercury vapor rises.
- 2. Crushing: A cone crusher reduces ore size.
- 3. Milling: Further size reduction using steel‑rod or ball mills.
- 4. Heating: Furnace or rotary kiln applies heat; mercury vapor is generated.
Condensing
- 5. The hot exhaust, containing mercury vapor, sulfur dioxide, water vapor, and fine dust, passes through a water‑cooled condenser.
- 6. Mercury condenses first (boiling point 675 °F/357 °C), separating from gases that are vented or treated.
- 7. Liquid mercury is collected; its high density causes impurities to float as a dark scum, which is filtered away. The resulting mercury is ~99.9 % pure.
Refining
Commercial‑grade mercury is typically 99.9 % pure and ready for use. For applications requiring higher purity, further refining—mechanical filtration, chemical oxidation, or electrolytic purification—is employed. The most common method is triple distillation, which progressively removes residual impurities and yields ultrapure mercury.
Shipping
- 9. Commercial mercury is sealed in wrought‑iron or steel flasks (76 lb/34.5 kg each). Ultrapure mercury is packaged in smaller glass or plastic containers for shipment.
Quality Control
Prime virgin‑grade mercury (99.9 % purity) is verified through spot checks for foreign metals such as gold, silver, and base metals. Ultrapure mercury undergoes triple‑distillation, followed by evaporation or spectrographic analysis to confirm absence of contaminants.
Health and Environmental Effects
Mercury is a potent toxin. Exposure routes include inhalation, ingestion, and dermal absorption, with inhalation of vapor being the most hazardous. Short‑term vapor exposure can cause weakness, chills, nausea, vomiting, and diarrhea; long‑term exposure leads to tremors, irritability, insomnia, confusion, and excessive salivation.
In typical diets, mercury primarily enters the body through fish consumption, where bioaccumulation occurs. Although gastrointestinal absorption is limited, chronic ingestion can result in cumulative effects.
Industrial exposure—especially in mining, ore processing, chlorine and caustic soda production, and dental amalgam placement—poses significant risks to workers.
In 1988, global human activities released approximately 24 million lb (11 million kg) of mercury into air, land, and water. The U.S. Environmental Protection Agency (EPA) has prohibited many mercury applications and set goals to reduce municipal refuse mercury from 1.4 million lb (0.64 million kg) in 1989 to 0.35 million lb (0.16 million kg) by 2000 through product reduction and recycling.
The Future
While mercury remains integral to certain products and processes, its use is expected to decline further. Advances in handling, recycling, and regulatory enforcement aim to minimize environmental releases and associated health risks.
Manufacturing process
- Gyroscopes: From Classic Toy to Modern Navigation Masterpiece
- The Craftsmanship and Standards Behind Every Official MLB Baseball
- Micro Craft: Decades of Excellence in Aerospace Testing Solutions
- Preventing Tool Deflection: Causes, Consequences, and Practical Remedies
- Optimizing Blast Furnace Design for Superior Performance and Longevity
- Why Traceability Matters: Boosting Quality, Safety, and Brand Trust
- UGIMA® 4542 H900 – Superior Machinability Stainless Steel
- AMPCO® 22 Forgings – Duplex Alloy with Exceptional Hardness, Compression & Wear Resistance
- UGIMA® 4542 – Premium Machinable Precipitation‑Hardened Stainless Steel
- POM (Polyoxymethylene) CNC Machining: Superior Properties & Applications



