Super Glue: Chemistry, Manufacturing, and Quality Control
Background
Super glue, formally known as cyanoacrylate adhesive, is a high‑strength, fast‑curing glue used across consumer, industrial, medical, and construction applications. It achieves its bonding power by forming a polymeric chain when exposed to moisture.
Adhesives can be grouped into five primary categories:
- Solvent glues – An adhesive base blended with volatile organic solvents (e.g., toluene) that evaporate, leaving a dry bond. These glues are flammable and work quickly.
- Water‑based glues – Use water as the solvent. They cure more slowly but are non‑flammable. Common examples include white glue and casein glue, which can be prepared from milk protein.
- Two‑part systems – Consist of a resin and a hardener (e.g., epoxy, resorcinol). Proper mixing is essential for optimal performance, especially on metal surfaces.
- Animal hide glues – Derived from hides, bones, or other animal parts. They are typically sold as powders or flakes that are mixed with water and heated.
- Cyanoacrylates (C.A.s) – The newest and strongest category, produced from synthetic polymers. They cure almost instantly in the presence of moisture and are used for everything from household repairs to medical sutures.
Cyanoacrylate adhesives were first discovered by Dr. Harry Coover and Dr. Fred Joyner at Kodak in 1951 during a refractometer experiment. Although the initial test ruined a costly instrument, it revealed a remarkably strong adhesive. Kodak launched Eastman 910 in 1958, the first commercial C.A. product. Today, numerous manufacturers offer a variety of formulations tailored to specific applications.
Although the exact polymerization mechanism is complex, the basic principle is that the monomer chains react with moisture to form a tightly cross‑linked network. This network generates a bond strength that can reach several thousand pounds per square inch (psi) of shear resistance, making C.A.s ideal for demanding tasks.
Raw Materials
The core ingredients for cyanoacrylate polymers include:
- Ethyl cyanoacetate – the primary monomer.
- Formaldehyde – a reactive agent that initiates polymerization.
- Nitrogen or other inert gases – to prevent unwanted reactions during processing.
- Free‑radical inhibitors and base scavengers – additives that suppress premature curing and remove impurities.
Other ancillary chemicals ensure product stability and performance.
Manufacturing Process
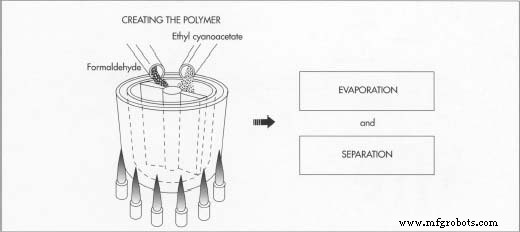
Creating the Polymer
- The process starts with ethyl cyanoacetate, which is mixed with formaldehyde in a glass‑lined kettle equipped with revolving blades. The mixture undergoes condensation, producing water that is subsequently evaporated by heating.
- Once water is removed, the remaining residue is the cyanoacrylate polymer. To keep the polymer from hardening prematurely, the kettle is purged with nitrogen.
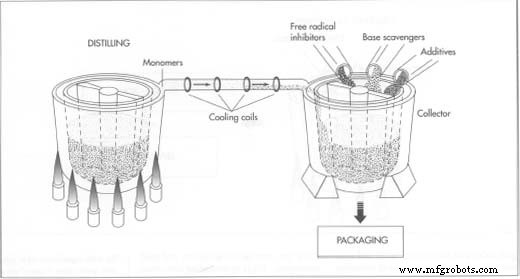
Separating Monomers
- Heating the polymer to approximately 305 °F (150 °C) causes thermal cracking, generating reactive monomers (ethyl or, with a different process, methyl cyanoacrylate). These monomers rise and are collected in a second vessel.
- They then pass through cooling coils, condensing into a liquid. Additional distillation steps may be employed for higher purity.
Preventing Premature Curing
During distillation, free‑radical inhibitors and base scavengers are added to the liquid monomer stream. The concentration of impurities is kept below parts‑per‑million levels to avoid visible precipitates, which would indicate contamination and require batch rejection.
Additives & Packaging
- Manufacturers can adjust viscosity (thick, medium, thin) and incorporate additives that enable bonding to porous or uneven surfaces.
- Once formulated, the adhesive is filled into tubes made of non‑reactive plastics such as polyethylene or, less commonly, aluminum. The tubes are sealed to protect the product from moisture until use.
Quality Control
Because the polymerization reaction is universal—every monomer present will bond—the manufacturing process must be meticulously controlled. Key checkpoints include:
- Verification of raw material purity and supplier compliance.
- Monitoring of mixing times, temperature profiles, and volume at each stage.
- In‑line testing of viscosity and reactivity.
- Final shear‑strength tests, which typically confirm values exceeding 10,000 psi.
Only products that meet these rigorous standards are shipped, ensuring that every bottle delivers the predictable, high‑performance bond expected from a cyanoacrylate adhesive.
Manufacturing process
- Diamond's Exceptional Role in Modern Medicine: From Surgical Instruments to Nanomedicine
- The History, Production, and Future of Glue: From Ancient Adhesives to Modern Innovations
- Mastering Java's super Keyword: Advanced Usage & Practical Examples
- DIY Low‑Cost Glove Controller & Propeller Car – Build Your Own Arduino Hand Controller
- DIY Hot Glue LED Matrix Lamp – Build Your Own Colorful Display
- DIY Automated Electronic Component Tester – Simple, Accurate, and Reliable
- Jokari Super 4 Plus Automatic Wire Stripper 20050 – Precision, Durability, and User‑Approved Performance
- UNS S32760 (Super Duplex 1.4501) – Premium Corrosion‑Resistant Alloy for Sour Service
- Super Invar® 32‑5: Ultra‑Low Thermal Expansion Alloy for Precision Engineering
- Elevate Your Game: 3D-Printed Shoes for the Super Bowl



