Rapid Electrospun SrTiO₃‑Modified Rutile TiO₂ Nanofibers: Enhanced UV‑Driven Photocatalytic Degradation of Methyl Orange
Abstract
We report a straightforward one‑step electrospinning synthesis of SrTiO₃‑modified rutile TiO₂ composite nanofibers. X‑ray diffraction, scanning and transmission electron microscopy confirm the successful formation of a SrTiO₃/TiO₂ heterojunction. Photocatalytic tests reveal that the SrTiO₃/TiO₂ (rutile) nanofibers degrade methyl orange (MO) under UV illumination at nearly twice the rate of pristine TiO₂ nanofibers, owing to their high crystallinity and efficient separation of photogenerated charge carriers.
Background
Titanium dioxide (TiO₂) is a benchmark semiconductor prized for its environmental benignity, chemical stability, and strong photo‑electric properties. It is widely applied in optics, photovoltaics, sensors, and wastewater remediation. While anatase TiO₂ generally shows superior photocatalytic activity compared to rutile, its wider band gap (3.2 eV) limits visible‑light absorption. Rutile TiO₂, with a slightly narrower band gap (3.0 eV) and greater thermodynamic stability, is more suitable for repeated use in pollutant degradation. Enhancing the photocatalytic efficiency of rutile TiO₂ remains a key research challenge. Strategies such as engineering high‑surface‑area morphologies (e.g., nanofibers, nanobelts, microflowers) and constructing heterostructures with other semiconductors (e.g., ZnO/TiO₂, CdS/ZnO, CeO₂/graphene) have proven effective. Strontium titanate (SrTiO₃) is especially attractive due to its thermal robustness and resistance to photocorrosion, and it has been employed in H₂ generation, NO removal, water splitting, and dye degradation. SrTiO₃/TiO₂ heterojunctions have been shown to promote charge‑carrier separation, boosting photocatalytic activity. However, reports on SrTiO₃‑modified rutile TiO₂ nanofibers are scarce, largely because conventional synthesis routes are cumbersome. Electrospinning offers a simple, scalable method to fabricate nanofiber composites with controllable composition and morphology. In this study, we present a single‑step electrospinning approach to prepare SrTiO₃/TiO₂ (rutile) nanofibers and investigate their photocatalytic performance and underlying mechanism.
Methods
Materials
Analytical‑grade acetic acid, N,N‑dimethylformamide (DMF, 99.5 %), tetra‑butyl titanate (TBT, 99.0 %), strontium acetate (99.97 %), and polyvinylpyrrolidone (PVP, M_w = 1.3 × 10⁶) were purchased from Shanghai Macklin Biochemical Co. Ltd.
Preparation of SrTiO₃/TiO₂ (rutile) Composite Nanofiber
The composite nanofibers were fabricated by direct electrospinning followed by calcination (Fig. 1). A 2.2 g PVP stock was dissolved in 8 mL DMF and 2 mL acetic acid, stirred for 8 h. Then 2 g TBT was added and stirred for 4 h. Strontium acetate was gradually introduced until the solution became transparent. The resulting sol‑gel was loaded into a glass syringe equipped with a 0.5 mm stainless‑steel needle and pumped at 0.6 mL h⁻¹. A 15 kV potential was applied with a 15 cm needle‑collector gap, at < 40 % relative humidity and 20 °C. The collected non‑woven mats were dried at 80 °C for 6 h, then calcined at 700 °C (5 °C min⁻¹) for 1 h to yield SrTiO₃/TiO₂ (rutile) nanofibers. Bare TiO₂ (rutile) and SrTiO₃ nanofibers were also prepared for comparison. SrTiO₃ loading levels of 1 wt %, 3 wt %, 5 wt %, and 10 wt % were denoted ST‑1, ST‑3, ST‑5, and ST‑10, respectively.
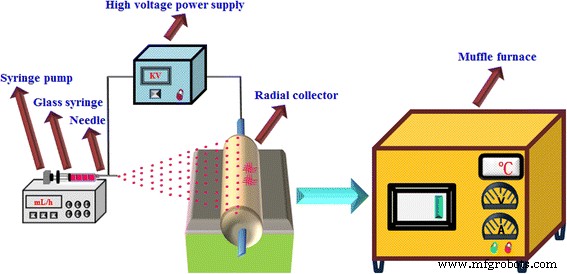
Schematic diagram of the preparation process of the photocatalyst.
Characterization
Field‑emission scanning electron microscopy (FESEM, Hitachi S‑4800) coupled with energy‑dispersive X‑ray spectroscopy (EDS) examined surface morphology. Transmission electron microscopy (TEM, JEM‑2100, 200 kV) and high‑resolution TEM (HRTEM) revealed microstructure. X‑ray diffraction (Bruker D8‑Advance, Cu Kα, λ = 1.518 Å) identified crystalline phases. UV–vis diffuse reflectance spectra were recorded on a Hitachi U‑3900 spectrophotometer.
Measurement of Photocatalytic Activity
Photocatalytic performance was evaluated by degrading a 15 mg L⁻¹ methyl orange (MO) solution in a 50 mL quartz reactor. 30 mg of catalyst was dispersed, and the suspension was stirred in the dark for 30 min to reach adsorption–desorption equilibrium. Degradation was driven by a 25 W UV‑C mercury lamp (λ ≈ 254 nm). Aliquots (5 mL) were withdrawn every 10 min, centrifuged, and the residual MO concentration was measured at 464 nm using a spectrophotometer.
Results and discussion
Figure 2 presents XRD patterns of bare TiO₂ (rutile), bare SrTiO₃, and the SrTiO₃/TiO₂ (rutile) composites. Peaks at 2θ = 27.5°, 36.1°, 41.3°, and 54.4° correspond to the (110), (101), (111), and (211) planes of rutile TiO₂ (JCPDS 78‑1510). Peaks at 32.4°, 40.0°, 46.5°, and 57.8° match the (110), (111), (200), and (211) planes of cubic SrTiO₃ (JCPDS 84‑0443). The clear presence of both phases confirms successful formation of the SrTiO₃/TiO₂ heterojunction under 700 °C calcination, which enhances charge‑carrier mobility and photocatalytic activity.
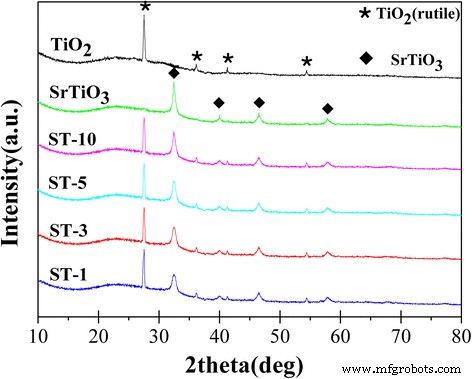
XRD patterns of the bare TiO₂ (rutile), bare SrTiO₃, ST‑10, ST‑5, ST‑3, and ST‑1.
FESEM images of the ST‑3 sample (Fig. 3) show that the as‑spun fibers have a smooth, continuous surface with a diameter of ~300 nm. After calcination at 700 °C, the diameter shrinks to ~200 nm, and the fibers become slender and rough, thereby increasing the specific surface area and providing more active sites for photocatalysis.

FESEM image of ST‑3. a As‑prepared ST‑3, inset: high‑magnification SEM (unsintered); (b)-(d) ST‑3 after sintering.
TEM and HRTEM analyses (Fig. 4) further confirm the coexistence of rutile TiO₂ and cubic SrTiO₃ phases. Lattice fringes with spacings of 0.324 nm (TiO₂ (110)) and 0.275 nm (SrTiO₃ (110)) are clearly resolved, evidencing the formation of an intimate heterojunction that facilitates efficient charge separation. Selected area electron diffraction (SAED) patterns and EDS spectra (Fig. 4c–d) corroborate the high crystallinity and elemental composition.
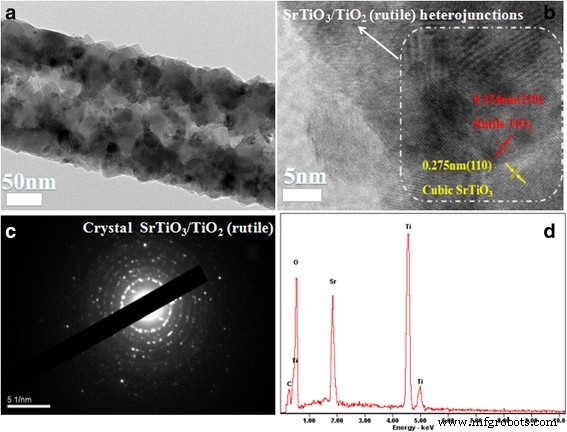
TEM image and EDS spectrum of ST‑3. a TEM image; (b) HRTEM of the rutile TiO₂ and SrTiO₃ interface; (c) SAED of ST‑3; (d) EDS of ST‑3.
The photocatalytic degradation of MO was evaluated for bare TiO₂ (rutile), bare SrTiO₃, and the SrTiO₃/TiO₂ composites (Fig. 5). After 40 min of UV irradiation, the degradation efficiencies were 62 % (ST‑1), 93 % (ST‑3), 79 % (ST‑5), 43 % (ST‑10), 47 % (bare TiO₂), and 44 % (bare SrTiO₃). The ST‑3 sample exhibits the highest activity, approximately twice that of bare TiO₂, demonstrating the benefit of an optimal SrTiO₃ loading. Excess SrTiO₃ (ST‑10) reduces performance because SrTiO₃ alone is a weaker photocatalyst; thus a balanced heterojunction is essential.
Photocatalytic activity of different samples. a Absorption spectra of ST‑3 during photocatalysis; b Degradation curves; c Recycling of ST‑3; d UV–vis spectra of products.
Recycling tests (Fig. 5c) show negligible loss of activity after five cycles, indicating robust structural stability and excellent cyclic performance.
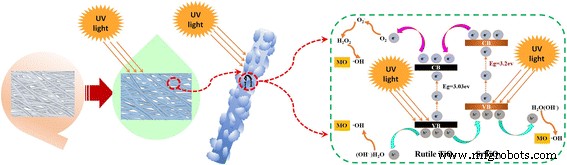
The mechanism underlying the enhanced photocatalysis is illustrated in Fig. 6. Under UV irradiation, both SrTiO₃ and rutile TiO₂ generate electron–hole pairs. Electrons in the SrTiO₃ conduction band transfer to the TiO₂ conduction band, while holes migrate to the SrTiO₃ valence band, effectively separating charges and extending their lifetimes. These charge carriers then react with dissolved oxygen and water to produce reactive oxygen species that oxidize MO molecules. The schematic (Fig. 6) and the reaction pathway (Eqs. 1–10) confirm this sequence of events.

Proposed mechanism for photocatalytic degradation of MO by SrTiO₃/TiO₂ (rutile) heterojunction.
Overall, the SrTiO₃/TiO₂ (rutile) composite nanofibers represent an inexpensive, scalable, and highly efficient photocatalyst for dye wastewater treatment.
Conclusions
We successfully fabricated SrTiO₃‑modified rutile TiO₂ nanofibers via a single‑step electrospinning process. The resulting heterojunction nanofibers exhibit high crystallinity and a three‑dimensional morphology that together elevate their photocatalytic performance, achieving nearly double the degradation rate of bare TiO₂ in UV‑driven methyl orange decomposition. This study demonstrates that simple electrospinning, combined with judicious heterojunction design, can produce robust photocatalysts suitable for large‑scale environmental applications.
Nanomaterials
- Top 10 Properties of Aluminium and Their Industrial Applications
- Aluminum 6061: Exceptional Corrosion Resistance & Versatile Applications
- Cellulose–POSS–Silica/Gold Core–Shell Hybrid Nanocomposites: One‑Step Sol‑Gel Synthesis and Multifunctional Properties
- Chromium‑Doped Titanium Dioxide: A Novel Colored Cool Pigment with High Near‑Infrared Reflectance
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Hierarchical Au@CdS‑CdS Nanoflowers: Stepwise Fabrication and Superior Photocatalytic Performance
- Hydrothermally Synthesized Nano-Copper Sulfide: Superior Adsorption of 17α-Ethynyl Estradiol
- Enhanced TiO₂ Catalysis and Antimicrobial Efficacy via Nitrogen and Carbon Nitride Co‑Doping
- Delrin (POM) Plastic: Properties, Grades, and Uses in CNC Machining



