Surface Defects in Ti‑Doped MgAl₂O₄ Nanophosphors and Their Role in White Emission
Abstract
Ti‑doped MgAl₂O₄ nanophosphors were prepared by a low‑temperature combustion method and exhibit a bright white emission. High‑resolution STEM imaging directly visualised extrinsic Schottky defects—Al vacancies (VAl′′′) and Ti4+ ions substituting Al (TiAl⁺) at the particle surface. Density‑functional theory calculations confirmed that the defect associate (TiAl⁺–VAl′′′)² is energetically stabilised, providing a clear microscopic explanation for the observed blue‑shifted white light in the nanoscale material.
Background
Transitioning from bulk to nanoscale materials profoundly alters mechanical, optical and electrical properties, largely due to size‑induced strain and the prevalence of non‑equilibrium surface structures [1–6]. In nanophosphors, quantum confinement and surface‑related defect chemistry can be harnessed to tailor emission characteristics [2,7–9]. The oxidation state of a dopant and its crystallographic site strongly influence phosphor performance; in nanoparticles, dopants frequently occupy surface sites that are inaccessible in bulk crystals, leading to distinct luminescent behaviour [8,9].
Pure MgAl₂O₄ contains an intrinsic Mg2+ vacancy (VMg′′) that gives rise to a 720 nm red emission. Ti‑doping replaces Ti4+ on Al sites, suppressing this red band and generating a blue emission from single crystals [10,11]. However, our earlier study of Ti‑doped micron‑sized MgAl₂O₄ powders heat‑treated at 1300 °C revealed a white emission, which we attributed to the simultaneous presence of Ti3+ and Ti4+ on both Al and Mg sites [12]. The present work visualises the extrinsic Schottky defects on the surface of Ti‑doped MgAl₂O₄ nanopowders and links them to the emission differences between nano‑ and micron‑scale samples.
Methods
The precursors Mg(NO3)2·6H2O (2.46 g), Al(NO3)3·9H2O (7.246 g) and urea (5.231 g) were dissolved in deionised water. Ti oxy‑acetyl‑acetonate (0.1 g) provided a 2 mol % Ti doping level. The mixture was stirred, evaporated on a hot plate, and then calcined at 500 °C for 1 h in air to produce the Ti‑doped MgAl₂O₄ nanophosphor.
Phase purity was confirmed by X‑ray diffraction (Rigaku XRD). Photoluminescence (PL) spectra were recorded on a PSI PL Darsa pro‑5000 spectrometer using 260 and 360 nm excitation from a Xe lamp. Morphology and size were examined by high‑resolution transmission electron microscopy (TEM, JEOL JEM‑2100F). Al vacancies and Ti dopants were visualised by high‑resolution scanning TEM (HR‑STEM, JEOL JEM‑2100F).
First‑principles density‑functional theory (DFT) calculations employed the Perdew–Burke–Ernzerhof GGA functional and PAW pseudopotentials in VASP [13–15]. A 500 eV plane‑wave cutoff and 10−5 eV self‑consistency criterion were used. Defect formation energies and binding energies were calculated for various dopant–vacancy configurations to assess their thermodynamic stability. Surface energies of the (100) plane were evaluated by constructing 4×1 supercells with a 2×1 vacuum slab, and the influence of Ti site position on surface formation energy was analysed.
Results and Discussion
Figure 1 shows the XRD pattern of the Ti‑doped MgAl₂O₄ nanophosphor, confirming phase formation at 500 °C. Broad diffraction peaks indicate sub‑20 nm crystallites (Figure 1b), in contrast to the highly crystalline micron‑sized powders from our previous study (prepared at 1300 °C).
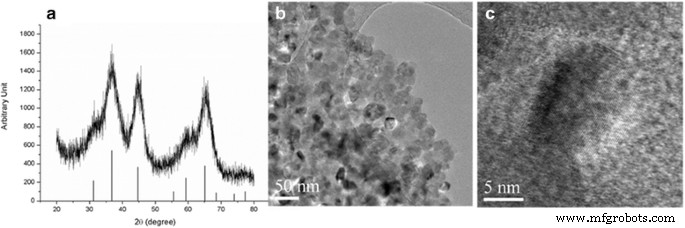
a XRD pattern of Ti‑doped nano MgAl₂O₄ (500 °C, 1 h) with reference peaks from JCPDS; b–c TEM images of the powder
Photoluminescence spectra (Figure 2) reveal white emission under 260 nm excitation for both nano (500 °C) and micron (1300 °C) samples. The nanopowder displays a slight blue shift, attributable to the higher proportion of Ti4+ occupying Al sites in the nanostructure, which suppresses red and green channels that are present in the micron‑scale material.
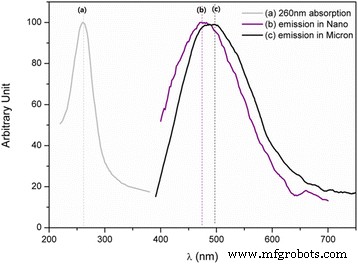
Photoluminescence of Ti‑doped MgAl₂O₄: a 260 nm excitation; b nano (500 °C); c micron (1300 °C)
HR‑STEM imaging (Figure 3) provides direct evidence of surface defects. In Figure 3a, a dark spot within the lattice indicates an Al vacancy (VAl′′′), while a brighter site (Figure 3c) confirms a Ti4+ dopant occupying an Al site. Fourier transformation of the lattice (Figure 3a inset) confirms the [001] orientation, supporting the assignment of the vacancy to the Al sublattice.
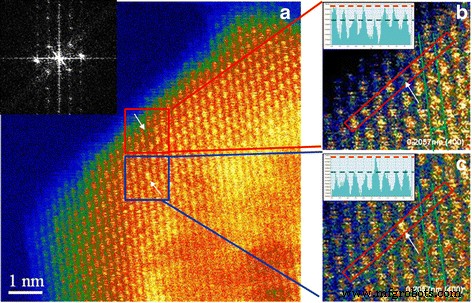
HR‑STEM images of Ti‑doped nano MgAl₂O₄: a Al vacancy and Ti4+ dopant; b magnified view of the vacancy;
DFT calculations (Figure 4) show that the surface energy decreases as Ti approaches the (100) surface, indicating a thermodynamic preference for surface localisation of Ti. Binding‑energy analysis (Figure 4b) demonstrates that the (TiAl⁺–VAl′′′)² associate is most stable when the defects are nearest neighbours, accounting for the blue emission characteristic of the nanopowder. The energetic hierarchy (≈4.5 eV for vacancy formation vs. ≈7.0 eV for O2− interstitials) [17,18] further supports the prevalence of these extrinsic Schottky defects on the nanocrystal surface.
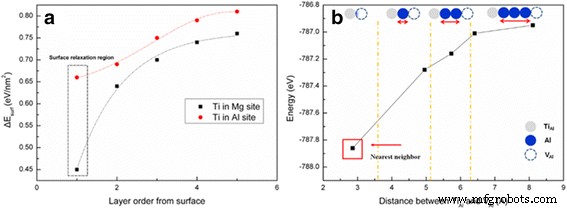
a Surface energy of Ti at Al vs. Mg sites; b Binding energy of (TiAl⁺–VAl′′′)² vs. separation distance
Collectively, the experimental and theoretical evidence demonstrates that the surface‑concentrated Ti4+–Al vacancy associates drive the blue‑shifted white emission of Ti‑doped MgAl₂O₄ nanophosphors, contrasting with the more balanced defect distribution in micron‑sized powders.
Conclusions
HR‑STEM imaging confirmed the presence of Ti4+ substituting Al and accompanying Al vacancies at the surface of Ti‑doped MgAl₂O₄ nanophosphors. The enhanced surface concentration of the (TiAl⁺–VAl′′′)² associate explains the observed blue shift of the white emission relative to micron‑scale material. While Ti4+ prefers Mg sites in bulk spinels, the low‑temperature combustion synthesis favours surface localisation on Al sites in the nanosystem, illustrating how processing conditions govern defect chemistry and luminescence.
Abbreviations
- Al nitrate:
Al(NO3)3·9H2O
- DFT:
Density functional theory
- HR‑STEM:
High‑resolution scanning TEM
- Mg nitrate:
Mg(NO3)2·6H2O
- PL:
Photoluminescence
- TEM:
Transmission electron microscopy
- Ti oxy‑acetyl‑acetonate:
C10H14O5Ti
- Urea:
CO(NH2)2
- VASP:
Vienna ab initio simulation package
- XRD:
X‑ray diffractometry
Nanomaterials
- Understanding and Preventing Surface Defects in Titanium Alloys
- Optimized Al₂O₃/MgO Emission Layers for Ultra‑High Gain Electron Multipliers
- Nanostructuring of Au/Ru(0001) Thin Films: Height Modulation and Superstructure Formation
- Boosting ZnO UV Emission with Silver Nanoparticle Arrays via Surface Plasmon Resonance
- Metal Surface Finishes: A Professional Guide to Optimal Application Techniques
- Mastering Surface Finish: A Comprehensive Guide to Measurement and Function
- Achieving Zero Defects: A Pragmatic Approach to Eliminating Failures
- In-Depth Tutorial: Understanding the Fundamentals of Electron Emission
- Mastering Surface Grinding: Key Principles for Superior Finish
- Understanding Welding Defects vs. Discontinuities: Key Differences and Quality Impact



