Graphene Oxide-Delivered TIMP‑1 Enhances Skin Regeneration Over 40 Days
Abstract
This study validates the hypothesis that graphene oxide (GO) can serve as an effective vehicle for the sustained release of tissue inhibitor of metalloproteinase‑1 (TIMP‑1) to accelerate skin repair. GO’s morphology and thermal properties were characterized by scanning electron microscopy (SEM), atomic force microscopy (AFM), and thermogravimetric analysis (TGA). After loading TIMP‑1 onto GO, we compared the release kinetics of varying protein concentrations. Cytotoxicity was assessed by fibroblast cell‑cycle analysis and apoptosis assays. In vivo, we evaluated the impact of TIMP‑1‑GO on a rat excisional wound model. GO exhibited a maximum lateral dimension of 1,140 nm and a peak volume of 10,674.1 nm³. TIMP‑1 remained continuously released for at least 40 days from GO. Fibroblast proliferation and viability were unaffected by TIMP‑1‑GO (p > 0.05). Histological and immunohistochemical analyses revealed significantly enhanced collagen deposition and vascularization (p < 0.05). Our data demonstrate that GO enables controlled, long‑term delivery of TIMP‑1, markedly improving skin regeneration.
Background
Cutaneous injuries arise from trauma, diabetes, burns, or surgery, and traditional autografts or synthetic skin substitutes often face donor shortages, infection risks, and suboptimal integration.
TIMP‑1 binds matrix metalloproteinases (MMPs) to protect extracellular matrix (ECM) integrity and modulates growth factor turnover essential for wound healing. Dysregulation of the TIMP‑MMP axis contributes to impaired healing, keloids, and fibrosis. Therefore, precise spatial and temporal delivery of TIMP‑1 is critical for optimal re‑epithelialization.
Existing cytokine carriers—PLGA, chitosan, PLGA microspheres, and hydrogels—provide limited release profiles. Graphene oxide, a two‑dimensional carbon material with abundant hydrophobic π domains and ionizable edges, offers high loading capacity, aqueous dispersibility, and low intrinsic toxicity, making it a promising platform for protein delivery.
Here, we evaluate recombinant human TIMP‑1 loaded onto GO for controlled release and skin repair efficacy in vitro and in a rat wound model.
Materials and Methods
Cell Culture
Primary rat fibroblasts (Institute of Biochemistry and Cell Biology, CAS) were cultured in DMEM supplemented with 10% FBS at 37 °C, medium changed every 48 h.
GO Characterization
GO flakes (Chengdu Organic Chemicals Co.) were examined by SEM (Hitachi S3000N, 15 kV) and AFM (VEECO MultiMode). Size distribution was measured via dynamic light scattering (Zetasizer 3000 HSA). TGA/DSC (Pyris 1 TGA) assessed thermal stability from 25–1,100 °C at 10 °C/min.
TIMP‑1 Adsorption on GO
GO was labeled with DiI (red, Sigma) and incubated with FITC‑conjugated TIMP‑1 (Thermo Scientific) in PBS for 4 h at 4 °C (GO:TIMP‑1 = 1:1 w/w). Confocal microscopy (Olympus IX81) visualized adsorption; FTIR (Nicolet 5700) confirmed protein binding.
Release Kinetics of TIMP‑1 Protein
GO loaded with 10, 20, or 30 µg/ml TIMP‑1 was suspended in 1.5 ml PBS in 60‑mm dishes and incubated at 37 °C. Supernatants were collected at predetermined intervals, refreshed with PBS, and quantified by TIMP‑1 ELISA (R&D Systems).
GO Biocompatibility Assay
Fibroblasts were exposed to 20 µg/ml TIMP‑1‑GO and monitored over 6–72 h. Viability was measured with CCK‑8 (absorbance at 450 nm, n = 5 per group).
Flow Cytometric Characterization
Cells were stained with Hoechst 33258 and Annexin‑V‑FITC/PI, fixed, and analyzed on a BD FACSCanto II to assess cell‑cycle distribution and apoptosis.
In Vivo Experiment
All procedures adhered to NIH guidelines and Zhejiang University Ethics Committee approval. Four‑week‑old male Sprague–Dawley rats received 10 mm × 10 mm excisional wounds. Fourteen days post‑surgery, rats were randomized into four groups: PBS, GO, TIMP‑1, or TIMP‑1‑GO (1:1 v/w). Treatments were subcutaneously injected (1 ml per site, four sites, once weekly for two weeks). Animals were euthanized 28 days post‑surgery for histological analysis.
Histological and Immunohistochemical Analysis
Tissues were fixed in 4% formalin, decalcified, dehydrated, and embedded in paraffin. Hematoxylin–eosin and Masson’s trichrome stained sections evaluated collagen architecture. CD34 immunostaining (Abcam) quantified neovascularization; sections were processed with DAB visualization and graded by three blinded observers.
Statistical Analysis
Data represent mean ± SD from three independent experiments. One‑way ANOVA with Student–Newman–Keuls post‑hoc test determined significance (p < 0.05 = significant; p < 0.01 = highly significant). Analyses were performed using SPSS 17.0.
Results
GO Characterization
AFM revealed 2‑D sheet‑like GO structures (Fig. 1a). SEM confirmed irregularly shaped flakes (Fig. 1b). Dynamic light scattering indicated a mode size of 1,140 nm and a volumetric peak at 10,674.1 nm³ (Fig. 2a).
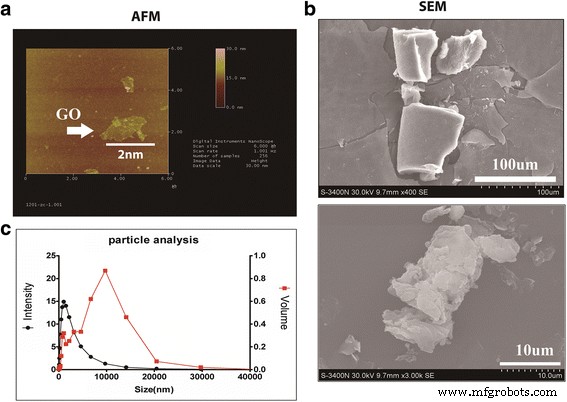
GO morphology: (a) AFM, (b) SEM, (c) size distribution.
TIMP‑1 Adsorption on GO
Confocal imaging confirmed 75 ± 1.2% TIMP‑1 loading onto GO after 4 h (Fig. 1c). FTIR spectra showed distinct peaks for TIMP‑1‑GO versus bare GO, confirming protein attachment. TGA curves overlapped, indicating thermal stability was retained (Fig. 2d).
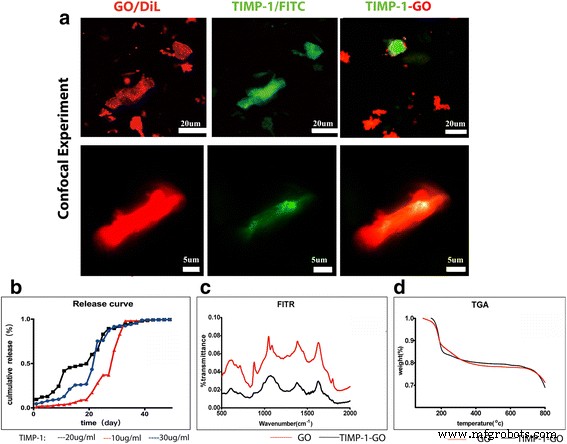
TIMP‑1‑GO characterization: (a) loading efficiency, (b) release profile, (c) FTIR, (d) TGA.
TIMP‑1 Release
Loading 2, 10, or 30 µg/ml TIMP‑1 onto GO produced sustained release. The 2 µg/ml dose reached 50% cumulative release faster than higher doses, yet all formulations maintained detectable TIMP‑1 for ~40 days (Fig. 2c).
Cell Proliferation and Viability on TIMP‑1‑GO
Fibroblast viability, cell‑cycle progression, and apoptosis rates were comparable across control, GO, TIMP‑1, and TIMP‑1‑GO groups (p > 0.05) (Fig. 3a‑c).
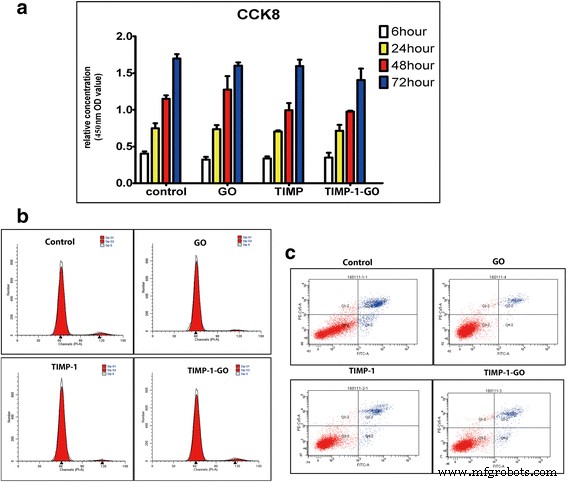
Effect of TIMP‑1‑GO on fibroblast function: (a) viability, (b) cell‑cycle, (c) apoptosis.
Efficacy of TIMP‑1‑GO in Excisional Skin Wound Model
After 28 days, TIMP‑1‑GO treatment yielded superior histological scores, increased collagen density, and enhanced hair follicle regeneration compared with PBS, GO, and TIMP‑1 alone (p < 0.05) (Fig. 4b‑c).
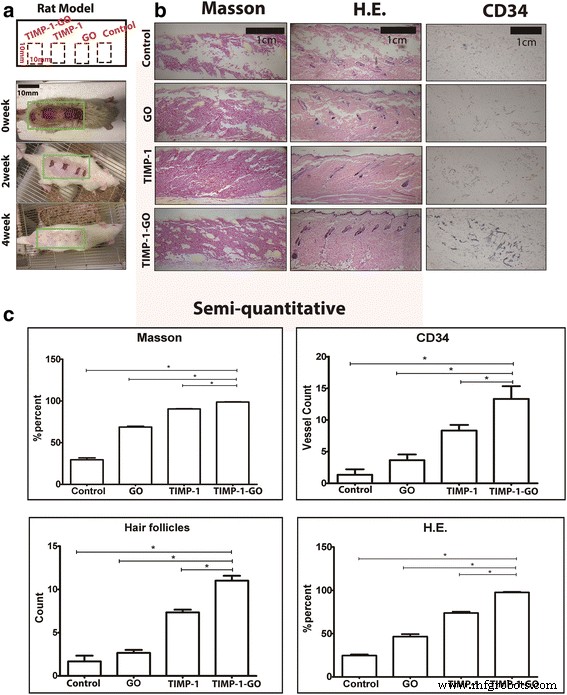
In vivo results: (a) experimental scheme, (b) histology/immunohistochemistry, (c) quantitative assessment.
Histologic and Immunohistochemical Analysis
Control wounds displayed fragmented collagen fibers at 4 weeks, whereas TIMP‑1‑GO‑treated wounds showed continuous, dense collagen and abundant CD34⁺ endothelial cells, confirming enhanced angiogenesis (p < 0.05) (Fig. 4c).
Discussion
Our findings establish GO as a safe, high‑capacity platform for the prolonged delivery of TIMP‑1, sustaining therapeutic protein release for up to 40 days. The sustained TIMP‑1 exposure supports balanced MMP activity, promotes ECM deposition, and facilitates neovascularization—key drivers of effective wound closure.
Compared with conventional carriers that release cytokines rapidly, GO’s π‑π stacking and electrostatic interactions afford a controlled, extended release profile. This mitigates the need for repeated dosing and reduces potential local side effects.
While high concentrations of carbon nanomaterials can elicit inflammation, our in vitro and in vivo data show no detectable GO deposition or adverse cellular responses at the tested dose (20 µg/ml). Nevertheless, long‑term safety studies remain essential for clinical translation.
Conclusion
Graphene oxide enables sustained, biocompatible delivery of recombinant TIMP‑1, enhancing collagen formation and angiogenesis in a rat skin wound model. This platform offers a promising strategy for chronic wound management and could inform the design of next‑generation biomaterial‑based therapeutics.
Nanomaterials
- Artificial Skin: Advanced Tissue Engineering for Burns, Wounds, and Future Regenerative Therapies
- Environment‑Responsive Metal–Organic Frameworks: Precision Drug Delivery for Tumor Therapy
- Zebrafish: A Real‑Time Model for Nanotechnology‑Enabled Brain Drug Delivery
- Nanotechnology: From In‑Vivo Imaging Systems to Controlled Drug Delivery
- Graphene Oxide–Silver Nanoparticle Nanocomposites: A Potent Antibacterial and Antifungal Agent
- Optimized Preparation and Physicochemical Characterization of Dual‑Drug Nanoliposomes Encapsulating Erlotinib and Doxorubicin
- High-Sensitivity Graphene Oxide Fluorescent Aptasensor for Rapid Detection of CCRF-CEM Leukemia Cells
- Graphene‑Based Materials for Bone Regeneration: Opportunities, Challenges, and Clinical Perspectives
- Achieving Near-Perfect Absorption in Graphene‑Metal Structures Using Plasmonic Interference
- Intelligent Nanoantenna‑Based Drug Delivery System for Targeted Cancer Therapy



